Purpose
Rectal injury sometimes occurs after radical prostate cancer treatments, such as prostatectomy, external beam radiation therapy (EBRT), and brachytherapy, with a recto-urethral fistula as a rare but profoundly life-altering sequela [1]. In radiation therapy, the dose to the anterior rectum is often a limiting factor, but that dose may be reduced by increasing the separation between the prostate and rectum, either by the presence of perirectal fat or by the injection of some spacer material. Hyaluronic acid injections to decrease radiation-induced rectal toxicity were first reported by Prada et al. in patients undergoing combined modality external beam and high-dose-rate (HDR) brachytherapy [2]. A subsequent clinical trial of a commercial product, the SpaceOAR™ System (Boston Scientific, Marlborough, MA, USA), confirmed significant and durable improvements in bowel quality of life (QoL), with lesser improvements in urinary and sexual QoL after use of hydrogel spacer during intensity-modulated radiation therapy (IMRT) [3, 4]. Rectal dose reduction caused by increasing the rectum-prostate separation is also important in salvage brachytherapy of previously irradiated patients [5, 6].
After permanent seed, low-dose-rate (LDR) brachytherapy implants, hydrogel spacer insertion has been shown to significantly reduce the most crucial rectal dosimetry parameters and to have minimal effect on target volume dosimetry [7]. The spacer material is usually injected before beginning EBRT or after completion of brachytherapy implant. Higher risk patients have better biochemical survival outcomes if they are treated with combined modalities of external beam radiation therapy and brachytherapy [8]. However, it remains unclear whether hydrogel injection is equally effective when it precedes a course of moderate external beam radiation followed by brachytherapy, or when hydrogel is placed immediately after completion of brachytherapy implant. Asymmetry in hydrogel distribution was found to adversely affect rectal dosimetry in prostate IMRT, and it is expected that displacement of the relatively flat posterior border of the prostate to the one that may be tilted superiorly or inferiorly and/or left vs. right, would adversely affect brachytherapy dosimetry [9, 10]. An asymmetrical SpaceOAR™ placement prior to the brachytherapy procedure makes treatment planning and placement of brachytherapy seed implants more difficult and complex due to distortion of the relatively flat posterior prostate. Even with an ideal, symmetric placement of hydrogel, increasing the prostate-rectum separation by about 5 mm makes execution of a template-guided seed implant more difficult, because more of the anterior prostate may be obstructed by pubic arch interference.
Increasing the time gap between hydrogel spacer placement and brachytherapy implant also increases the amount of hydrogel resorbed. MRI imaging at 12 weeks post-SpaceOAR showed only modest absorption based on differences in rectum-prostate separation compared to the initial separation, but patients imaged just one week later presented with markedly reduced separation [11]. Although the time gap to brachytherapy should not exceed 12 weeks, post-implant rectal dosimetry may be a sensitive indicator of relative loss of rectum-prostate separation. That separation will continue to decrease, as the brachytherapy radionuclide decays – half the total prescribed brachytherapy dose is delivered in one half-life after the implant, and 90% of the prescribed dose is delivered in 56 days for 103Pd (200 days for 125I).
This work retrospectively compared the brachytherapy dosimetry of 348 patients, 174 consecutive patients injected with hydrogel SpaceOAR™ vs. a reference cohort of 174 contemporaneous patients without hydrogel injection. In addition, the dosimetry of two SpaceOAR™ sub-groups were compared, including 83 patients who had the hydrogel applied before EBRT and brachytherapy vs. 91 patients who had the hydrogel placed at the conclusion of brachytherapy implant.
Material and methods
The study population consisted of 348 consecutive prostate patients who received a low-dose-rate permanent seed brachytherapy implant between November 2016 and July 2020. This population was divided into two cohorts: 174 men who were injected with SpaceOAR™ and received brachytherapy between January 2019 and July 2020, and 174 patients not injected with hydrogel who had brachytherapy implants after November 2016. Ninety one of 174 SpaceOAR patients had the hydrogel placed immediately after the brachytherapy implant, and the remaining 83 patients had the hydrogel injected in 57.9 ±16.1 days (mean ± standard deviation) and median = 63 days (interquartile range, 50-69 days) prior to brachytherapy implant. In that pre-brachytherapy time period, these patients received a risk-group-specific moderate dose of EBRT as either 2.0 Gy fractions delivering a total of 20 Gy, or 1.8 Gy fractions delivering either 45 Gy or 50.4 Gy.
The SpaceOAR™ hydrogel was injected transperineally under ultrasound guidance after hydro-dissection of Denonvilliers’ fascia with sterile saline, following the approach of Pinkawa et al. and the manufacturer’s recommendations [12]. For patients prescribed a moderate dose of EBRT, the hydrogel was injected concurrently with implantation of three Varian Calypso® radiofrequency tracking beacons (Varian Medical Systems, Palo Alto, CA, USA) about one week prior to EBRT treatment planning [13]. All the 348 patients in the study were planned and implanted with 103Pd seeds, and had post-implant imaging and dosimetry performed within 24 hours according to the American College of Radiology practice guidelines [14]. Between the SpaceOAR™ and non-SpaceOAR™ patients (Table 1), there was no significant difference between the planning or evaluation target volumes, or the number of seeds implanted. The SpaceOAR™ patients had a statistically higher Gleason score, which, according to our protocols, led to a greater prescribed EBRT dose and a lower brachytherapy prescribed dose (Table 2). Our chosen seed strength was proportional to the brachytherapy prescribed dose and to a lesser extent to the prostate volume. Therefore, the lower brachytherapy prescribed dose for SpaceOAR™ patients led to a lower mean individual seed strength.
Table 1
SpaceOAR vs. no SpaceOAR, continuous variables
Table 2
SpaceOAR vs. no SpaceOAR, categorical variables
Results
At post-implant dosimetry, the rectal mean dose, rectal maximum dose, and rectal wall V50 were all statistically significantly lower in the SpaceOAR™ patients (Table 1). The minimum dose covering 90% of the planned target volume (PTV D90) was also statistically significantly lower in the SpaceOAR™ patients, 121.1% ±10.0% vs. 123.4% ±8.7% in the patients with no hydrogel spacer. The clinical effect of this small dosimetric difference was unclear, and all patients, with or without SpaceOAR™, met our criteria for acceptable dosimetry, namely, D90 > 100% of the prescribed dose and V100 > 90% of the target volume. These data are summarized graphically in Figure 1, and the stark differences in rectal max. dosimetry are illustrated in the dose distribution histograms of Figure 2.
Fig. 1
Mean dosimetric parameters of patients implanted with SpaceOAR hydrogel (sOAR, n = 174) and a reference cohort of patients with no SpaceOAR (no sOAR, n = 174). The dosimetry difference between the two patient groups was statistically significant (p ≤ 0.05) for prostate target volume D90, rectal average dose, and rectal maximum dose
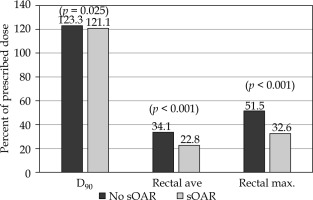
Fig. 2
Histograms of rectal maximum dose as a percentage of the prescribed dose (%Rx) for the two equal-sized cohorts: patients without SpaceOAR (A, reference) and patients implanted with SpaceOAR hydrogel (B). The latter group is more tightly skewed toward low doses
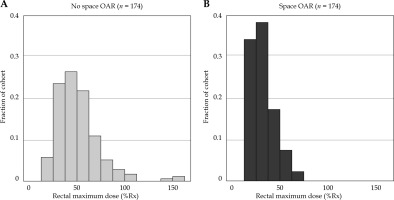
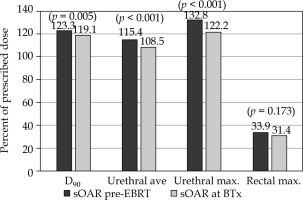
Table 3 compares the sub-groups of SpaceOAR™ patients, i.e., those injected with the hydrogel before external beam and brachytherapy with those injected with the hydrogel just after the brachytherapy implant. There was no significant difference in rectal dosimetry parameters between the sub-groups. However, in the patients injected with SpaceOAR™ at the completion of brachytherapy, PTV parameters D90 and Vpd% as well as urethral dosimetry parameters were statistically significantly lower than in those patients injected with SpaceOAR™ prior to any radiation therapy. The bar graphs of Figure 3 place these dosimetric differences in a context: the differences are statistically significant, but their clinical significance is uncertain.
Table 3
Patients who received SpaceOAR prior to any radiation therapy compared to those who received SpaceOAR on the day of brachytherapy seed implant
Fig. 3
Mean dosimetric parameters of SpaceOAR subgroups: patients implanted with SpaceOAR hydrogel before any radiation therapy (sOAR pre-EBRT, n = 83) and patients implanted with SpaceOAR hydrogel after completion of a brachytherapy implant (sOAR at BTx, n = 91). The dosimetry difference between the two patient groups was statistically significant (p ≤ 0.05) for the prostate target volume D90, urethral average dose, and urethral maximum dose, but they were not significantly different for any rectal dosimetry parameters
It is noteworthy that in the sub-group injected with SpaceOAR™ prior to any radiation therapy, the PTV D90 and V100 means were nearly identical with the reference cohort not injected with the hydrogel. Table 4 details the differences between the non-hydrogel reference group and the two SpaceOAR™ sub-groups. PTV and urethral dosimetry parameters of the sub-group receiving Space-OAR™ prior to EBRT were not significantly different from the reference cohort. However, the sub-group receiving SpaceOAR at the time of brachytherapy had statistically significantly lower mean PTV and urethral dosimetry than those patients not receiving any hydrogel. Both the SpaceOAR™ sub-groups (prior to and after brachytherapy) had significantly lower mean and maximum rectal doses than the non-hydrogel reference cohort.
Table 4
Patients who received no SpaceOAR (reference group) compared to those who received SpaceOAR prior to any radiation therapy and compared to those who received SpaceOAR on the day of brachytherapy seed implant
Because the injected hydrogel was not always a uniform 5 mm spacer between the prostate and rectum, treatment plans sometimes placed seeds within the gel rather than within the prostate tissue. Our protocols require expansion of the prostate by about 5 mm in all directions except posteriorly to create the PTV, which has a planning goal of V100 > 99.9%. This has always entailed a large number of seeds outside the prostate and some outside the PTV. However, planned placement of seeds posterior to the PTV is an unusual feature of some SpaceOAR™ EBRT patients. Adding about 5 mm of rectal spacing necessarily raises the prostate, thus increasing the likelihood of pubic arch interference. This was addressed by adjusting the patient’s leg position to open the pelvis or to insert the implant needles on an upward trajectory to get behind the pubic arch. This occurred in the SpaceOAR™ and non-SpaceOAR™ cohorts, and the intra-operative dosimetry was unremarkable. Since the seed placement produced satisfactory dosimetry in all cases, it was not noteworthy, and therefore our documentation was insufficient to determine if arch interference was more common in EBRT sub-groups.
Discussion
For LDR prostate brachytherapy treatments, the dose to the rectum in those patients with injected spacer material is considerably less than in non-spacer control groups. This is consistent with the results reported by Kahn et al. [15] for 125I brachytherapy and by Taggar et al. [7] for 103Pd brachytherapy. We did find one rectal dose parameter that was not significantly different between the SpaceOAR™ and non-SpaceOAR™ cohorts: the rectal V110. Because of the rapid dose fall-off with distance for 103Pd seeds, the rectal volume receiving 110% of the prescribed prostate dose approached zero, and the median for both cohorts was zero cm3.
In their 125I brachytherapy study, Kahn and colleagues found no significant difference between prostate dosimetry parameters in patients with or without hydrogel spacer, but the 103Pd brachytherapy study of Taggar and colleagues did find a significantly higher prostate V150 and V200 in hydrogel group. This is contrary to our finding of a significantly lower D90 in the hydrogel group and no significant difference in the other PTV indices. All patients in the Taggar study had the hydrogel injected at the completion of brachytherapy implant; therefore, the proper comparison group in our study was the Space-OAR™ at brachytherapy sub-group. The patients in this sub-group relative to the non-hydrogel cohort had statistically significantly cooler PTV D90, V100, V150, V200, and the prostatic urethra dosimetry parameters. Some of the difference between our target volume and urethral dosimetry and that of the Taggar group may be due to implant approach. Creating a PTV by adding 5 to 7 mm to the prostate makes the PTV about twice the volume of the prostate. Our PTV V100 goal of > 99% resulted in placing a large fraction of the seeds outside the prostate, and only a small number interior to the prostate and very few within 10 mm of the urethra.
The difference between the SpaceOAR™ sub-groups in terms of PTV and urethral dosimetry may be multifactorial. The patients injected with hydrogel at the completion of brachytherapy experience not only prostatic edema from implant trauma and radiation, but also distortion from the freshly placed hydrogel and edema due to infiltration into the prostate of the saline solution, creating the initial space for the hydrogel. In this work, we did not measure the dimensions of the prostate post-implant, so the hypothesis of distortion and additional edema caused by the hydrogel must await a future study.
In higher risk patients, we have always performed EBRT first followed by brachytherapy implant within one week of EBRT completion. The reverse sequence requires a time gap of one to two half-lives of the radionuclide to reduce the overlay of brachytherapy and EBRT doses. This gap adds to the radiobiological uncertainty of the total biological effective dose (BED) due to poorly characterized repair and repopulation rates. However, this sequence should be considered in the case of patients with marginal or actual pubic arch interference at the initial prostate volume study, because of the increased technical difficulty of implant after the hydrogel spacer, which raises the prostate.
Limitations of this study include its retrospective design. Prostate brachytherapy involves dynamic processes of radioactive decay, the inception and patient variable resolution of edema due to implant trauma and radiation, the resorption of implanted hydrogel, and the possible migration and loss of implanted seeds. Sequential post-implant dosimetry studies as part of a clinical trial would have provided more granular temporal information, but we chose to use day 0 dosimetry, which is a protocol we have used for all our prostate brachytherapy patients. The AAPM Task Group 137 recommends post-implant imaging on either day 0 or at the nominal optimal dosimetry time for the radionuclide being used [16]. Some of our patients on clinical trials had additional imaging studies, but none of those were part of this study. Although some EBRT patients were implanted in the time period when the spacer is being absorbed, the spacer has already served its primary purpose of protecting the rectum from EBRT.
Conclusions
In a study population treated with LDR prostate brachytherapy, the hydrogel SpaceOAR injected between the prostate and rectal wall to increase the separation between the prostate and rectum, significantly reduced rectal dosimetry values compared to the reference group without spacer injection. The rectal dose sparing effect was similar in the sub-group of patients injected with the hydrogel prior to EBRT (2-10 weeks prior to the brachytherapy implant) and the sub-group patients injected with the hydrogel at the conclusion of the brachytherapy implant.



