Purpose
Cervical cancer is a growing public health problem in the low- and middle-income countries, and it is the third most common cancer among women worldwide [1]. Standard treatment of locally advanced cervical cancer is chemoradiation. Generally, radiotherapy is delivered as external beam irradiation and intracavitary brachytherapy. Brachytherapy (BRT) implicates application of a radioactive source in the vicinity of tumor to deliver high-dose to the tumor, while sparing normal surrounding tissues [2,3]. Traditionally, brachytherapy was performed by applying a radiation dose to a geometrical point A, without considering individual tumor size or anatomy of surrounding organs [4]. However, gynecological (GYN) GEC-ESTRO working group introduced recommendations for image-guided brachytherapy in cervical cancer to improve treatment planning and decrease organ at risk (OAR) complications [5]. The rectum is a possible site of side effects due to its proximity to the cervix [6]. Previous studies have demonstrated that late rectal complications are best quantified from the dose to 2cc (D2cc) of rectum [7]. Filling volume and distention of rectum, tandem angle, and treatment planning method are reportedly the factors altering D2cc of rectum [8,9].
Computerized tomography (CT) scanning advances allow target volume and OAR delineation to generate dose-volume histograms (DVH). Although administering dose to the point A has been the standard method for decades, recent improvements in customized dose distribution and optimization based on delineated contours might be the optimal solution for brachytherapy treatment planning [10,11].
Different studies have attempted to reduce rectal volume or distention by using enema, gas removal, or rectal retractor with unpromising results [8,9], but does optimization planning could overcome rectal dose-volume correlation. The aim of present study was to compare target volume coverage and dose delivered to the rectum as well as to evaluate the effect of rectal volume on its DVH in two different treatment planning approaches. Moreover, our study utilizes a stepwise approach, from the use of standard point A planning to the optimization based on the GYN GEC-ESTRO recommendations in our institution.
Material and methods
Study population
This was a dosimetric study including locoregionally advanced cervical cancer patients, who received high-dose-rate (HDR) brachytherapy boost as a component of definitive radiotherapy in the Interventional Radiotherapy Ward of Ahvaz Golestan Hospital between March 2016 and January 2018. Patients who met the eligibility criteria were included into the study.
Patients’ eligibility criteria consisted of a histologically confirmed diagnosis of cervical squamous cell carcinoma stage IIB-IVA, no evidence of distant metastases, no history of abdominal or pelvic RT, 0-2 points in the Eastern Cooperative Oncology Group performance status, and no known history of inflammatory bowel disease.
Patients were clinically staged according to the International Federation of Gynecology and Obstetrics (FIGO) criteria [12]. FIGO stage was determined by clinical examination and basic radiographic studies, such as intravenous urography and X-ray examination. All patients subsequently received further imaging after diagnosis, and those findings were used to stage each patient according to the AJCC criteria. Computed tomography and magnetic resonance imaging (MRI) were used to determine local tumor extension, lymph node involvement, and distant disease. Periaortic lymph nodes were considered the sites of possible distant metastases.
Inclusion criteria
Planning CT scan of all eligible patients were reviewed using treatment planning software (HDR Plus v3.7.0.0; Eckert & Ziegler BEBIG GmbH, Germany), and criteria for adequate implant placement were assessed as described in the American Brachytherapy Society (ABS) guidelines [1,13]:
The tandem should bisect the ovoids.
The ovoids should not be displaced inferiorly from the cervical stop and should be as symmetrical as possible (should overlap one another).
The tandem should be approximately one-half to one-third the distance between the symphysis and the sacral promontory.
The superior tip of tandem should be located below the sacral promontory within the pelvis.
Radio-opaque packing would be visible on radiographic images and should be placed anterior and posterior to the ovoids, with no packing visible superior to the ovoids. Superior packing represents an unwanted inferior displacement of the applicator.
Delineation of volumes of interest
To decrease intra- and interobserver variation in delineation of OAR and target volumes, all volumes were re-contoured by the same physician, based on the following criteria. Quality assurance was performed by at least one physicist.
Delineation criteria
Rectum was contoured from rectosigmoid junction superiorly to the ischial tuberosity inferiorly. The sigmoid was contoured from rectosigmoid junction to inferior border of the sacroiliac joint. The maximum rectal distension was measured as the largest anterior-posterior diameter of the rectum opposite to the applicator from the tip up to the cervical collar. The entire outer bladder wall was segmented [8].
High-risk clinical target volume (HR-CTV) was contoured based on the American Brachytherapy Society task group: the width of cervix and any parametrial extension was contoured, the superior border of cervix consisted of at least 3 cm height from the cervical collar [1].
Radiotherapy procedure included external beam radiation therapy (EBRT), followed by intracavitary brachytherapy course. All patients received concomitant weekly cisplatin during EBRT. EBRT and brachytherapy courses were administered in the outpatient setting. Initially, the whole pelvis was treated with 6/18 VM photons with a 3D conformal four-field box technique up to a 45-50.4 Gy in 22 to 28 fractions. Five HDR brachytherapy fractions were administered twice a week, using a 60Co remote afterloading technique.
Treatment planning
Two different methods of treatment planning and dose calculation were performed: volume-based optimization planning and Manchester method [4,14].
Manchester method or dose prescription to point A
Planning is conducted on applicator geometry without modifications in loading based on tumor volume and organ at risk doses. This method is generally accepted technique of brachytherapy for decades, which was considered in this work for comparison as a standard method.
Volume-based planning method
Planning is conducted as intensity modulated optimization with constraints as follows: HR-CTV D90: 100 ±10%, D2cc of the rectum and sigmoid < 75 Gy, and bladder D2cc < 90 Gy. All parameters were calculated as EQD2 using an ABS worksheet.
Other reported parameters for HR-CTV were V100, V150, V200, mean and maximum dose, and HR-CTV volume. Mean and maximum dose, volume, D1cc, and D0.1cc of rectum and bladder were described, with all data reported as percentages. Moreover, dose to point A (left and right) was defined as a reference. All mentioned parameters were calculated and also reported in Manchester method.
Results are presented as mean (± standard deviation) or as median (interquartile range [iqr]) for non-normal data. Normality was assessed using the Kolmogorov-Smirnoff test. DVH parameters (D0.1cc, D1cc, and D2cc) were assessed using paired and unpaired T-tests. Correlation among the variables was evaluated using the Spearman’s or Pearson’s coefficient tests. For all statistical analyses, the statistically significant value was set as 0.05 (p-value ≤ 0.05).
Results
In total, sixty-five eligible patients were included into the study. The mean age of participants was 46 years old, and almost 70 percent were stage IIB. All patients received 45-50.4 Gy of EBRT and 5 fractions of HDR brachytherapy, with a dose of 5.5 Gy prescribed for each BRT fraction. The mean rectal and HR-CTV volumes were 52.19 ±2.4 cc and 40.87 ±1.36 cc, respectively. The results of the comparison of dose-volume parameters between the point A and optimized plan are presented in Table 1. In the optimized plan, the mean dose to 90% of HR-CTV was significantly higher, whereas the D2cc, D0.1cc, D1cc, and maximum dose to rectum and sigmoid were significantly lower. The volume of HR-CTV receiving 100% of the prescribed dose was higher in optimized plan, but the difference was not statistically significant.
Table 1
The DVH parameters in optimized and point A plans. Data are presented as mean ± SD
The images of two different planning methods of a sample case are as follow. Figures 1 and 2 show dose distribution using optimized plan and point A plan, respectively.
Fig. 1
Dose distribution using optimized plan
Blue dots – bladder, pink dots – HR-CTV, yellow dots – sigmoid, brown dots – rectum, HRCTV – high-risk clinical target volume
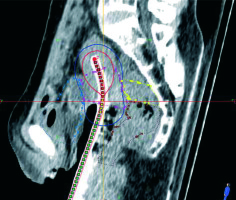
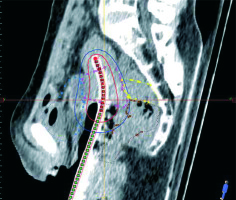
Fig. 2
Dose distribution using point A plan
Blue dots – bladder, pink dots – HR-CTV, yellow dots – sigmoid, brown dots – rectum, HR-CTV – high-risk clinical target volume
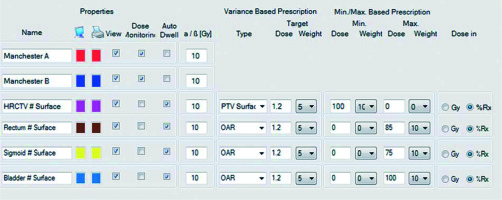
The optimized plan, using inverse planning method, provided the required dose distribution based on simultaneous HR-CTV coverage and normal tissue protection. It began with the description of dose constrains in the optimization window (Figure 3). Then, based on DVH parameters, various weights were changed to provide best dose distribution.
On the other hand, in the point A method, 100% of dose was prescribed to the point A. The DVH parameters of the above sample case using two different planning methods are compared in Table 2.
Table 2
Comparison of dose volume histogram parameters in optimized and point A planning methods
| Rectum D2cc (Gy/%) | Sigmoid D2cc (Gy/%) | Bladder D2cc (Gy/%) | HR-CTV D90 (Gy/%) | Lt Point A (%) | Rt Point A (%) | |
|---|---|---|---|---|---|---|
| Optimized plan | 4/72.9 | 4.2/76.9 | 4.7/86 | 5.5/100.4 | 108.7 | 104.5 |
| Point A plan | 5.2/93.8 | 4.9/89.1 | 7.1/130 | 6/109 | 103 | 100 |
There was a significant direct correlation between the total rectal volume and its dosimetric parameters in the optimized plan, but not in the point A plan. HR-CTV volume showed a significant indirect correlation with D90, V100, V150, and V200 in the point A plan, but no correlation was observed in the optimized plan (Table 3).
Table 3
The correlation between rectum, sigmoid, and HR-CTV volume and DVH parameters
The mean dose to point A was 96.83% in the optimized plan. The maximum recto-sigmoid anterior-posterior diameter was 3.93 cm, which showed a direct significant correlation with D2cc, D1cc, and D0.1cc of rectum in the optimized plan (p = 0.001, p = 0.002, and p = 0.002, respectively). No statistically significant correlation was observed between this parameter and dosimetric values in the point A plan.
Discussion
Our study suggests that not only D2cc of rectum and sigmoid was lower in the optimized plan compared to the point A plan, but also rectal volume showed a significantly direct relation with D2cc and other DVH parameters during HDR brachytherapy using the optimized plan (p = 0.012). This correlation was not observed for the point A plan (p = 0.051). Therefore, it could be hypothesized that during dose prescription to point A, reducing rectal volume does not impact total rectal dose and subsequently locoregional tumour control (LRCs), but in the dose optimizing plan, smaller rectal volume results in lower rectal dose and LRCs. Previous studies have investigated both dose-volume dependency of rectum and effectiveness of various volume reduction methods (gas removal, enemas, rectal retractors, etc.) [15,16,17].
The 2cc dose reduction of organ at risk using an optimization plan was demonstrated by Paul et al. They compared volume-based HDR brachytherapy plans to the point A-based plan. They revealed that volume-based plan resulted in a reduction of total dose and every HDR brachytherapy fraction dose to 0.1cc, 1.0cc, and 2cc of the OAR, 6-12%, and 8-37%, respectively [18]. Tang et al. showed that inverse planning simulation annealing (IPSA) optimization was not superior to graphical optimization regarding target coverage. Target values of D90, V100, V150, and V200 were comparable in these two planning methods (p > 0.05). Although, D2cc and D1cc of rectum and bladder were significantly lower in the IPSA method [19].
Sheybani et al. in a retrospective study showed that larger rectal volume results in higher absorbed doses of this organ. They reported a statistically significant correlation between total rectal volume, and V74% (p < 0.0001) and V50% (p < 0.0001); however, they did not find any correlation between filling volume and D2cc % [9].
Sabater et al. evaluated whether the rectal volume reduction related to gas removal results in lower doses to organ at risk during vaginal brachytherapy. They re-segmented and re-planned 14 pairs of CT scans. The only difference between CTs was the presence or lack of gas in the rectum. The first CT showed a basal status and the second indicated a status after gas removal using a tube. The study showed a significant rectal volume reduction in the second round of CTs (p = 0.0052), indicating a significant reduction in all studied rectal DVH parameters, except D25% and D50%. Our finding supports this dose-volume dependency of rectum [16].
The present study showed that rectal DVH parameters are significantly associated with its maximum recto-sigmoid anterior-posterior diameter. These results support the findings of Lim et al., who showed a significant direct relation of rectal distention with D2cc delivered in HDR brachytherapy of locally advanced cervical cancer [8].
Geometrical dose prescription by point A planning has been accepted as a standard brachytherapy method for decades. In this technique, the prescription point is determined based on applicator position, irrespective of the target coverage. Consequently, an increase or decrease in target volume could result in under- or overestimated dosage of the target.
Our findings showed that D90, V100, V150, and V200 of HR-CTV were significantly correlated with HR-CTV volume in the point A planning (p = 0.00).
Dose optimization based on anatomical constrains using inverse planning system would overcome this limitation. Our findings showed that target volume (HR-CTV) alterations could not interfere with D90, V100, V150, and V200 of HR-CTV using the optimized plan (p = 0.30, p = 0.35, p = 0.98, and p = 0.96, respectively). This means that the optimization resulted in better HR-CTV coverage with proper homogeneity.
Our study compared the optimized planning approach with the traditional point A planning as a standard method. The findings showed a good coverage of point A using anatomical optimization plan, with the mean dose at point A as over 96% of prescription dose.