Purpose
Eyelid carcinoma is a rare but highly visible tumor, thus usually detected at early stages. Malignant eyelid neoplasms account for less than 3% of all skin cancers in the head and neck area [1]. A persistent, slow-growing lesion that does not respond to topical treatments, requires biopsy by an ophthalmologist. Most cases are basal cell carcinomas (BCC), while squamous cell carcinomas (SCC) or sebaceous carcinomas are occurring less commonly [2]. First treatment option for eyelid carcinoma is an excision with clear margins, aiming at preserving eyelid function. Mohs surgery or wide excision are the primary treatments, achieving local control rates of 95-98% [3]. However, these options can lead to several adverse effects, particularly in cosmetic and functional aspects [4].
The most effective alternative to surgery is brachytherapy, as it delivers a curative dose of radiation while protecting nearby tissues, particularly the eyeball, cornea, and lens. Due to the anatomical location of the tumor, external beam radiotherapy (EBRT) fails to deliver conformal dose distributions, although electron beam irradiation is used rarely [5] despite the associated risk of side effects, such as cataract.
Brachytherapy (BT), also known as interventional radiotherapy (IRT), has been used for decades, recently evolving from low-dose-rate (LDR) approach with radioactive iridium wires, to high-dose-rate (HDR) using an iridium-192 afterloader with dose optimization and modulation [6]. It consists of inserting one or more plastic tubes, which cover the affected eyelid to deliver radiation dose in short sessions, without the need to isolate the patient in a shielded room. Even though the literature on LDR-BT have demonstrated its efficacy, this system is no longer used [7]. Cases treated with pulsed-dose-rate radiotherapy (PDR) or HDR-BT are small series, and there is no clear evidence on usefulness of these techniques [8]. However, most of publications show very good local control results, with limited toxicity and function preservation [9]. The most widely used is the interstitial technique, while superficial or contact BT studies have also been published [10].
In this article, we reviewed the published literature and described the interstitial implant technique in detail. BT is one of the best ways to treat eyelid carcinoma, as a complement to surgery in cases with involved or very close surgical margins, as the sole initial treatment, or in cases with a post-surgery recurrence. The purpose of this review was to promote the utilization of this technique as an addition to ocular surgery, with close collaboration among radiation oncologists, ophthalmologists, and dermatologists.
Interstitial brachytherapy implant technique in the eyelid
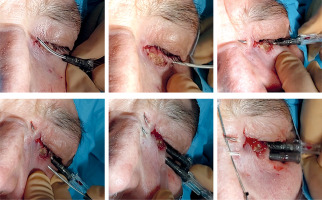
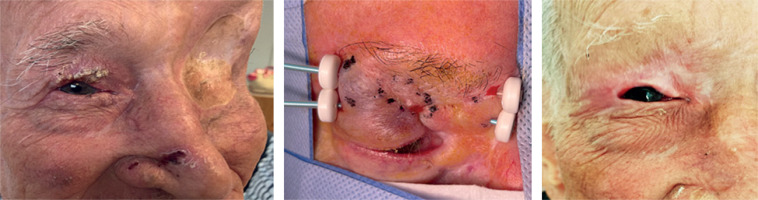
The procedure is short and simple: one to three parallel plastic tubes are inserted horizontally to cover the tumor or surgical scar. Local anesthesia with lidocaine or truncal anesthesia with Ultracaine™ injected into the infra-orbital nerve is required (Figure 1). General anesthesia is not needed as well as antibiotics administration. Antibiotic eye drops, corticosteroids drops, or ointment are applied to the eye to reduce irritative symptoms.
Fig. 1
Acantholytic SCC, 14 × 9 mm, 80-year-old woman. Truncal anesthesia with Ultracaine™ injected into the infra-orbital nerve
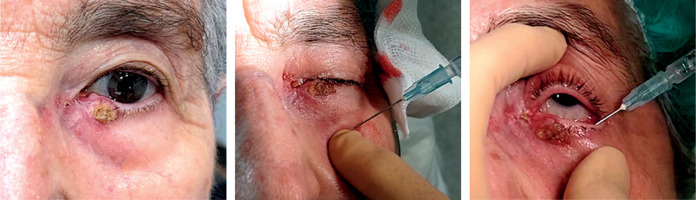
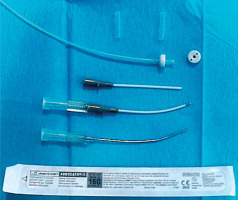
For this treatment, hollow, open needles are used, and they can be replaced with plastic tubes, as in other IRT sites. It is practical to use a 4-5 cm long Abbocath™, which can be slightly curved to fit the shape of the eyelid (Figure 2). Clinical target volume (CTV) is defined based on visible tumor or scar, leaving a margin of 5-10 mm on each side for the needle insertion and exit points. The procedure starts from the outer side, following the eyelash line, ensuring to direct the needle outward and avoid damaging the eyeball. For small lesions or linear scars, a 3-4 cm length is sufficient. In a small or microscopic lesion, a single needle may be appropriate, but in the case of macroscopic lesion or larger CTV, a second parallel needle of the same length is inserted, spaced 7-10 mm caudally (Figure 3).
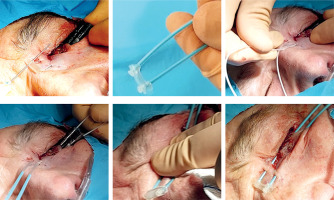
In the next step, a 6F plastic tube (2 mm thick) is inserted, with a thin guidewire and a button at the end, from the tip of the open needle. The needle is carefully withdrawn until the plastic tube remains in place. Another button is added to the outer end to secure the plastic tube, as is done in other locations, such as breast BT. The remaining needles are also replaced with plastic tubes. The button can be changed before insertion with a plastic guidewire that holds it in place for greater comfort (Figure 4). In a larger tumor, a third needle may be required, forming a triangle with two needles inserted previously, using a small template with pre-drilled holes to secure the system. However, this is usually not necessary as larger tumors should be removed, and it is preferable to perform BT in the scar if the margins are compromised.
Planning and prescription
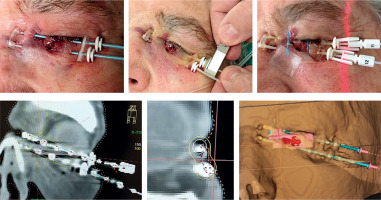
A planning CT scan is performed with thin 1.5 mm sections to visualize the horizontal tubes. Radiopaque markers should be placed on the edges of CTV. The tubes are cut to the same length and numbered (Figure 5). Gross tumor volume (GTV) is drawn on CT images if visible, and the CTV as marked on the skin without additional margin. The eyelid is few mm thick. Organs at risk (OARs), such as the lens and eyeball, should be contoured.
Fig. 5
Fixation of the buttons, tube cutting, and CT planning with radiopaque markers. Isodoses and 3D reconstruction
When using a single tube, the dose is prescribed remotely, 3-5 mm from the source, optimizing the positions to cover the entire length of the CTV. If two plastic tubes are used, the dose is prescribed at A points located between them, considering 100% isodose. Therefore, the prescribed isodose is 85% or 80% to cover the eyelid thickness. In the case of a macroscopic tumor, the same isodose is prescribed, and the curves are manually adapted to cover the GTV.
Interstitial implants are treated in 8 to 14 sessions, twice daily, with an interval of 6-8 hours between each one. The dose per fraction varies between 3 and 4.5 Gy. In the case of macroscopic tumor, total doses of 4-4.5 Gy × 9-10 sessions [11, 12], 3.5 Gy × 12-14 sessions [13], or 3 Gy × 14 sessions are administered. In the cases of post-operative BT without macroscopic tumor, the scar is treated with 4 Gy × 8-9 sessions, 3.5 Gy × 9-10 sessions, or 3 Gy × 10-12 sessions, depending on the thickness of CTV. Higher doses per fraction, 6.5 Gy × 6 sessions in six days, have been described, but in such a sensitive area, it seems more advisable to use lower doses per session [14]. As for biological equivalent dose (BED), following linear quadratic model with α/β = 10 for tumor, the BED should be between 50 and 66 Gy with tumor present, and 40 to 56 Gy for post-operative cases. It is important to define the distance at which the dose is prescribed, since the actual dose administered in proximity to the catheter is different when the dose is prescribed at 3 mm instead of 5 mm.
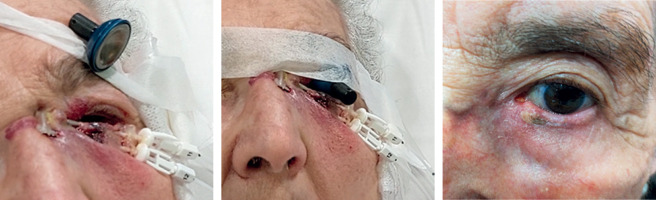
For the treatment session, anesthetic eye drops are placed in the eye, so that a lead eye shield (0.5-1 mm thick) can be placed, resting it on the inner conjunctiva and also covering the upper eyelid [15]. A special eye shield, such as that used for electrons in external beam radiotherapy (EBRT), can be employed (Figure 6). The treatment lasts for less than one minute. Between sessions, antibiotic and dexamethasone eye drops are prescribed three or four times a day. Oral antibiotics are not required. On the last day, removal of the tubes is simple by cutting and gently stretching them, applying pressure at the ends to avoid bleeding.
Results
Using LDR-BT at doses of 60 Gy for BCC and 70 Gy for SCC, local control was achieved in 96% [16]. Toxicity was reported in 18%, more frequently when treating post-surgery recurrences, but late toxicity ≥ G3 was observed in 3% of cases only [17]. Published HDR cases demonstrated a control rate of around 94% [18, 19]. In 17 cases with a mean follow-up of 24 months, using 4 Gy in 10 fractions prescribed at 3 mm, no visual complications were seen [11]. Also, a review of six publications on HDR showed 95% control and good functional and cosmetic results [20]. In 58 patients with lower eyelid BCC, 4 Gy in 9 fractions were administered over 5 days in post-operative cases and up to 40 Gy in 10 fractions in cases with tumor, local control with 95% and 100%, respectively, was achieved at 44 months [12]. Eight patients developed grade 3 cataracts during follow-up, and cosmetic results were excellent or very good in 93% of the cases. In a study, higher doses per fraction were used, i.e., 5 Gy in 9 sessions over 5 days, up to a total dose of 45 Gy [13]. Whereas in 20 patients, a dose of 6.5 Gy was used once a day for 6 sessions with a 40-month follow-up, and 5-year control rates of 90% in SCC and 50% in BCC were achieved [14].
The most common toxicities are acute conjunctivitis or keratoconjunctivitis. The patient should be warned that eyelid swelling can be most severe between the second and fourth week after implantation, so they should continue using eye drops for 2 to 3 weeks. After six weeks, the eyelid is fully recovered, with epilation of the lower eyelashes (Figure 6).
Late side effects may include linear atrophy of the eyelid skin, depigmentation, and epilation of the eyelashes. Epiphora, ectropion, and dry eye are uncommon, but the risk increases with large implants and higher doses per fraction [13]. This is not the case when only the lower eyelid is affected. If eye protection is used to shield the cornea during each session, the development of cataract over time is rare. In addition, upper eyelid carcinomas are not a contraindication for the treatment when accurate shielding is used (Figure 7).
Discussion
Brachytherapy offers a clear dosimetric advantage over EBRT due to the rapid fall-off of dose curves and protection of nearby OARs. It is a minimally invasive, quick, and simple procedure that can be performed in collaboration with the ophthalmologist at the start of the treatment. It is done in the same way as other interstitial implants, such as the lip, skin, or breast. If a system, e.g., Oncosmart™ is available, the patient does not require hospitalization after implantation and can be treated on an outpatient bases.
Surgery remains the first treatment option for eyelid tumors if eyelid retraction with sequelae can be avoided, as large tumors are more difficult to be adequately covered with plastic tubes. Each case should be discussed in a skin tumor committee before deciding on the best treatment, and the option of surgery and post-operative BT should be considered if necessary due to risk factors for a relapse. Because this is such a visible area, it should be closely monitored and BT delayed until an initial recurrence is observed, which can be easily covered with one or two plastic tubes. However, if available, BT should be used as an adjuvant treatment rather than delayed until a recurrence occurs.
Dosimetry is important, as variations in prescription at a distance of 5 mm or 3 mm from the source provide the same nominal doses, but actual doses are different. Therefore, it should not be prescribed at more than 5 mm to avoid late toxicity. With two tubes, the isodose chosen is the one that covers the thickness of the CTV between them. Consequently, the 90% isodose is not usually employed, rather a more peripheral curve, such as 85% or 80%, assuming that the dose around the tube will be higher when using a more peripheral isodose. The dose per fraction is determined based on individual case, age, and condition. Also, if an implant is performed on Monday and the first session is given immediately that same day, it may be advisable to perform 9 or 10 sessions, removing the implant on Friday, and leaving the tubes in place for only four nights. This requires choosing a dose per fraction of 3.5 to 4.5 Gy. The higher the fraction dose, the greater the risk of late toxicity. If a dose of 3 Gy or 2.5 Gy is chosen, the total treatment time will be longer and extended till the weekend.
Irradiated volume is very limited; in a study among 17 patients, the median V100 was 2.38 cc [11]. The irradiated volume depends on the size of tumor and the number of catheters, while there is no maximum volume recommended, ensuring that the surrounding tissues are protected. Publications on HDR-BT for eyelid carcinomas are scarce and present small number of cases, thus, a standard regimen cannot be recommended. We described different options to support decision-making in each case [11-14, 18-20]. Instead of HDR-BT, PDR-BT can also be used, but very few cases have been described.
In more extensive cases involving the inner corner of the eye, BT can also cover the area, but it is advisable to leave a catheter in the tear duct by the ophthalmologist for several weeks to avoid stenosis with permanent epiphora. The upper eyelid is a more complex area, but BT is also possible on a case-by-case basis. Good results have been published in advanced eyelid tumors with involvement of periocular tissues using multiple plastic tubes [21].
In this review and description of the technique, we focused on the most common cases, in which the tumor is at early stage and a simple implant possible, ensuring protecting the eyeball and cornea. With further experience, other more complex cases can be addressed. BT allows dose adaptation to very limited volumes, and with HDR optimization, it achieves better distribution than LDR, which has already clearly demonstrated its efficacy.
Conclusions
BT/IRT is one of the best options for the treatment of lower eyelid carcinomas, whether post-operatively or exclusively. It delivers a curative dose in a very limited volume, with excellent protection of the remaining ocular structures. It is a simple and minimally invasive technique, and should be routinely implemented in all centers with brachytherapy units.