Introduction
Coeliac disease (CD) is an autoimmune disorder of the small intestine triggered by ingested gluten from barley, rye and wheat. The disorder occurs in genetically predisposed people – class II human leukocyte antigens (HLA), such as HLA-DQ2 or, much less commonly, HLA-DQ8, are expressed in the majority of patients with CD. It is estimated to affect 1 in 100 people worldwide. The disease has both intestinal and extraintestinal manifestations. The intestinal symptoms include abdominal pain, diarrhoea and other nonspecific abdominal symptoms. The manifestations outside the gastrointestinal tract include rashes (e.g. dermatitis herpetiformis), arthralgias, neurologic and psychiatric symptoms, fatigue, infertility, weight loss, iron deficiency anaemia, osteoporosis or osteomalacia due to calcium and vitamin D malabsorption.
Coeliac disease can be associated with other autoimmune conditions, such as type 1 diabetes, autoimmune thyroiditis and hepatitis, Sjögren’s syndrome and IgA nephropathy (IgAN) [1]. Individuals with CD suffer a 3-fold increased risk of future IgAN [2] along with an increased risk of subsequent end-stage renal disease [3].
Atypical form of CD presenting with a few or even no gastrointestinal symptoms or signs is largely responsible for the increased prevalence of CD today.
Case report
We describe a case of a 24-year-old man with atypical form of CD for whom the diagnosis started with renal disorder.
The patient (measuring 164 cm, BMI 20) was admitted to the Department of Internal Medicine because of peripheral oedema, headache and elevation of blood pressure (140/90 mmHg). The symptoms had appeared a few days earlier. In laboratory analysis, microcytic anaemia, hypoalbuminaemia and elevation of hepatic enzymes were found. Urinary protein loss was 6 g/day. The diagnosis of nephrotic syndrome was established and coexistence with CD was also suspected. In fact, immunoglobulin (Ig) A and IgG antibodies against endomysium and against gliadin were detected in the serum of the patient and the endoscopic biopsy of the duodenum revealed markedly increased numbers of intraepithelial lymphocytes (60-70 per 100) along with crypt hyperplasia and moderate mostly lymphocytic infiltrates present in the lamina propria. The villi exhibited a leaf-like appearance with moderate blunting in height (subtotal villous atrophy) (Fig. 1A). These histopathological features were classified as stage 3B according to the modified Marsh-Oberhuber classification [4].
Fig. 1
Duodenal biopsy shows loss of normal villous architecture. There is extensive atrophy present with almost complete absence of villi along with marked intraepithelial lymphocytosis (insert). Moderate degree of chronic inflammation is seen in the lamina propria (H&E, 100×, 400× insert)
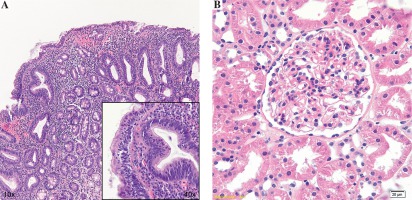
Percutaneous kidney biopsy was also performed. Immunofluorescence microscopy showed mesangial granular IgA and C3 deposits in glomeruli. Class I IgAN with a focal increase in mesangial cells was revealed by light microscopy (G I according to the modified H. S. Lee system); according to the Oxford classification of IgAN M0, S0, E0, T0 predictors of outcome were found [5] (Fig. 1B). Electron microscopy was not performed.
Gluten-free diet, ACE inhibitor according to KDIGO Guidelines [6] and oral iron were introduced to the patient. Gradual remission of the nephrotic syndrome was observed. Clinical and laboratory characteristics of the patient before and after 6 months of therapy are shown in Table 1.
Table 1
Clinical and laboratory parameters of the patient at the onset and after 6 months of therapy
Discussion
This case presents atypical CD associated with nephrotic syndrome due to IgAN. Two similar cases were previously described in the literature [7, 8]; both presented nephrotic syndrome. Moreover in one, in contrast to our patient, renal function was markedly affected at presentation. The pathogenic link between CD and IgAN involves increased permeability of the mucosal barrier to pathogens that can stimulate the production and further deposition of pathogenic IgA in the glomerular mesangium [9]. Moreover, patients with CD overexpress the transferrin receptor on enterocytes, which acts as an IgA receptor, and the transferrin receptor is associated with transglutaminase 2 in CD and IgAN [10]. Recently, Kiryluk et al. [11] performed a genome-wide association study (GWAS) of IgAN and, besides nine previously reported associations, identified six new signals. Most of them were associated with either risk of inflammatory bowel disease or maintenance of the intestinal epithelial barrier, activation of mucosal IgA production and defence against intracellular pathogens. These findings could explain the genetic susceptibility of some individuals with CD to develop IgAN. On the other hand, a recent serologic study by Moeller et al. [12] did not demonstrate a significant role of CD in IgAN. In fact, in comparison with healthy subjects, there was no statistically significant increase in IgA or IgG antibody titres to gliadin in IgAN patients. Moreover, patients with IgAN and unaffected controls did not differ in the level of antibodies to deamidated gliadin and TG2, CD-specific serologic markers. A large epidemiological study from Sweden also demonstrated that only 0.026% of CD patients develop IgAN [13].
In our patient, introduction of a gluten-free diet and ACE inhibitor resulted in complete remission of nephropathy and improvement of clinical and laboratory disorders of CD. Koivuviita et al. [8] reported a case of a male patient with nephrotic syndrome and IgAN in whom the treatment with prednisone and immunosuppressive drugs was ineffective. Also in this case introduction of a gluten-free diet after the diagnosis of CD was established resulted in complete remission of kidney disease.
In conclusion, there may be a small group of patients with IgAN coexisting with CD in whom a gluten-free diet seems to be the treatment of choice for the resolution of kidney disease.


