Purpose
Cervical cancer is the fourth most common cancer among women in terms of incidence and mortality [1]. Majority of patients present in locally advanced stage, and need external beam radiotherapy (EBRT) and brachytherapy (BT) as well as concurrent chemotherapy. Brachytherapy plays an important role in the treatment of cervical cancer. Low-dose-rate (LDR) brachytherapy is replaced by high-dose-rate (HDR) brachytherapy, which is more convenient for patient, with no risk of radiation exposure to the staff and most importantly, with the advantage of dose optimization [2]. Most of the institutes follow similar dose prescription pattern for LDR brachytherapy; however, there is a wide range of discrepancy in HDR brachytherapy dose prescription. In the literature, we have observed different institutions following different HDR brachytherapy schedules such as 5.5 Gy × 5 fractions, 6 Gy × 4 fractions, 7 Gy × 3 fractions, and 9 Gy × 2 fractions, probably because of the availability of the resources and logistic problems. In our institute, we follow 6.5 Gy × 4 fractions for two consecutive days, after 45 Gy EBRT. Since the local control and toxicities are determined by the dose per fraction, it is important to know the EQD2 for different dose prescription schedules for HDR brachytherapy. EQD2 or equieffective dose is also referred as equivalent dose in 2 Gy fractions [1,3,4,5,6].
The aim of this work is to investigate different HDR brachytherapy dose schedules of EQD2 for high-risk clinical target volume (CTVHR) and organs at risk (OARs), and also comparing dose schedules with available literature.
Material and methods
In this retrospective study, cervical cancer patients with stage IB2 to IIB treated from January 2017 to December 2017 were included. Fifty patients who underwent intracavitary brachytherapy (ICBT) were included, and patients who underwent combination of ICBT with interstitial needles were excluded from the study.
Procedure
Around 10-15 days after the completion of EBRT, ICBT applicators (uterine tandem with flange and two vaginal ovoids) were inserted under spinal anesthesia or mild sedation. To ensure the emptiness of the rectum, sodium phosphate enema was given two hours before the procedure. Computed tomography (CT) scan simulation without intravenous contrast was completed after the insertion of applicators. During CT simulation, bladder was filled with 50 ml of normal saline and rectum was filled with 20 ml of rectal contrast. Axial CT slices of 3 mm thickness were taken from the upper border of third lumbar vertebra to the middle of shaft of the femur. The CT images were transferred to treatment planning system (HDR Plus, Bebig, Eckert and Ziegler, Germany) compliant with the Task Group 43 (TG-43) algorithm.
Planning and evaluation
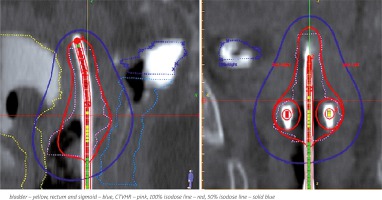
The OARs such as bladder, rectum, sigmoid, and CTVHR were contoured according to the guidelines published by Viswanathan et al. [7]. CTVHR was defined based on the findings of examination under anesthesia and CT scan image. Intermediate-risk clinical target volume (CTVIR) was created by giving 10 mm margin to CTVHR. Applicators were digitized using the applicator library and verified with digitally reconstructed radiograph (DRR) before activating the dwell positions. At that point, CTVHR surface control points were generated and dose prescribed to CTVHR. The dose-volume histogram (DVH) parameters were evaluated and isodose reshaper tool was used to maintain the pear shape dose distribution with maximum possible D90 (dose received by 90% of the CTVHR) and restrict the dose to OARs within the permissible limits. Figure 1 shows the dose distribution of intracavitary brachytherapy. Even though the dose was prescribed to CTVHR, we also calculated the dose received by point A, which was marked 2 cm superior to external os and 2 cm perpendicular to the uterine tandem.
Dosimetric details
Four different plans were generated for each patient, with dose prescriptions of 5.5 Gy × 5 fractions (plan A), 6.5 Gy × 4 fractions (plan B), 7 Gy × 4 fractions (plan C), and 9 Gy × 2 fractions (plan D) to CTVHR. The EQD2 for 0.1 cm3 and 2 cm3 of bladder, rectum, sigmoid, point A, and D90 were calculated for all plans, using the formula BED = nd (1 + d/α/β) and EQD2 = BED/[1 + (2/α/β)], where n is the number of fractions and d is the dose per fraction. The α/β ratio was considered as 3 for normal tissues and 10 for the tumor [8]. The values were tabulated and analyzed by the statistical software SPSS version 18.0 (SPSS Inc., IBM, Chicago, USA). One-way ANOVA test was used to compare the values. The values were considered to be statistically significant when p ≤ 0.05.
Results
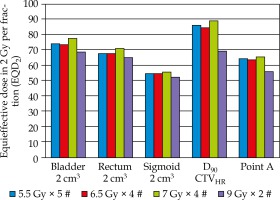
A total of two hundred plans were generated for different dose prescriptions. The mean EQD2 for OARs, D90 CTVHR, and point A is summarized in Tables 1 and 2, and Figure 2. The mean CTVHR volume was 47.12 ±13.8 cm3. We have observed that plan D delivered lower EQD2 to 0.1 cm3 and 2 cm3 of bladder compared to other plans (p = 0.001). There was no difference in the mean EQD2 for 0.1 cm3 and 2 cm3 of bladder for plan A, B, and C (p = 0.8, p = 0.1, and p = 0.07). Plan C delivered higher EQD2 to 0.1 cm3 and 2 cm3 of rectum compared to other plans (p = 0.001). There was no significant difference in the mean EQD2 for 0.1 cm3 of rectum for plan A, B, and D (p = 0.9, p = 0.3, and p = 0.2). The mean EQD2 for 2 cm3 of rectum was lower for plan D compared to plan A and B (p = 0.001, p = 0.002). There was no significant difference in the mean EQD2 for 0.1 cm3 and 2 cm3 of sigmoid for all the plans. The mean EQD2 of D90 CTVHR and EQD2 of point A was similar for plan A, B, C, but plan D delivered lower EQD2 to D90 CTVHR and point A compared to other plans (p = 0.0001).
Table 1
The total EQD2 (EBRT + BT) of organ at risks for different dose prescriptions (mean and standard deviation)
Table 2
The total EQD2 (EBRT + BT) of D90 CTVHR and point A for different prescriptions (mean and standard deviation)
| Plan (dose × fractions) | D90 CTV HR (Gy10) | Point A (Gy10) |
|---|---|---|
| Plan A (5.5 Gy × 5) | 85.9 ±3 | 64.36 ±6 |
| Plan B (6.5 Gy × 4) | 84.67 ±2 | 63.49 ±6 |
| Plan C (7 Gy × 4) | 89.03 ±2 | 65.24 ±7 |
| Plan D (9 Gy × 2) | 69.16 ±4 | 55.86 ±5 |
Discussion
This retrospective dosimetric study was conducted to compare 6.5 Gy × 4 fractions of ICBT that we follow with the other fractionation schedules available in the literature. An attempt was made to compare the EQD2 of D90 CTVHR and OARs. In the literature, we found different dose schedules like 5 Gy × 5 fractions, 6 Gy × 4 fractions, 7 Gy × 4 fractions, 7.5 Gy × 3 fractions, and 9 Gy × 2 fractions being applied in practice [9,10,11,12,13,14,15,16]. The choice of fractionation schedule of a particular oncology center mainly depends on the number of patients, availability of the staff, cost effectiveness, etc. At our center, we follow single application of ICBT with four fractions of 6.5 Gy, with 6-hour gap between the fractions, on two consecutive days, two weeks after the completion of EBRT of 45 Gy in 25 fractions by 3DCRT technique, along with concurrent chemotherapy. Image-guided intensity modulated external beam radiochemotherapy and MRI-based adaptive brachytherapy in locally advanced cervical cancer (EMBRACEII) study recommends MRI-based brachytherapy for better delineation of OARs and CTVHR [7,9]. In our institute, we practice CT-based brachytherapy because of limitation of the resources. We have summarized the EQD2 of OARs and D90 CTVHR of different studies in Table 3. In these studies, the EQD2 for 2 cm3 of bladder, 2 cm3 of rectum, and 2 cm3 of sigmoid are in the range of 79.7-88.2 Gy, 57.5-82.8 Gy, and 61-73.9 Gy, respectively. According to the ICRU 89 and the American Brachytherapy Society (ABS), the recommended EQD2 for 2 cm3 of bladder and rectum is ≤ 90 Gy and ≤ 75 Gy, respectively, and it is also recommended to achieve 85 Gy EQD2 for D90 CTVHR [3,4]. In the present study, the range of EQD2 for 2 cm3 of bladder, 2 cm3 of rectum, and 2 cm3 of sigmoid is 68.58-77.27 Gy, 64.82-71.11 Gy, and 52.14-55.44 Gy, respectively. Although volume-based prescription is the standard practice, most of the centers still follow the X-ray-based point A prescription. Ghosh et al. in their study based on X-ray-based planning observed higher incidence of late toxicities of bladder and rectum in patients treated with 9 Gy × 2 fractions compared to 7 Gy × 3 fractions [16]. Mazeron et al. reported increased grade 3 and higher rectal complications for dose > 75 Gy for 2 cm3 of rectum [17]. Georg et al. reported in their study that there is an increased probability of grade 3 rectal toxicities for dose greater than 88 Gy and 76 Gy for 0.1 cm3 and 2 cm3 of the rectum, respectively [18]. In a retrospective study by Manir et al. on correlation of rectal toxicity and dose, it was recommended to restrict the dose between 75 to 81 Gy for 0.1 cm3 of the rectum and between 64 to 69 Gy for 2 cm3 of the rectum to avoid grade 3 proctitis [19]. In our study, we have observed EQD2 in the range of 80 to 89 Gy for 0.1 cm3 of the rectum and 65 to 71 Gy for 2 cm3 of the rectum by all the four plans. All the dose fractionation schedules were in compliance with the ICRU 89 recommendation for OARs. The EQD2 of D90 CTVHR in different studies are in the range of 73.82-93.1 Gy. Studies have shown better local control with EQD2 of D90 CTVHR in the range of 80 to 90 Gy [3,5]. Tanderup et al. in their study reported better local control rate with D90 CTVHR ≥ 85 Gy [20]. Dimopoulos et al. reported lower incidence of local recurrence and better local control in patients who received more than 87 Gy EQD2 for CTVHR [21]. In the present study, the mean EQD2 of D90 CTVHR of plan A, B, C, and D were 85.9 Gy, 84.67 Gy, 89.03 Gy, and 69.19 Gy, respectively. Even though we have prescribed the dose to CTVHR, we calculated the dose received by point A. The EMBRACE II study aimed at achieving 65 Gy EQD2 to point A [5]. In our study, mean EQD2 of point A with plan A, B, C, and D were 64.36 Gy, 63.49 Gy, 65.24 Gy, and 55.86 Gy, respectively. Overall, plans A, B, and C were better in terms of EQD2 for D90 CTVHR and point A. We are in the process of comparing our clinical data with different institutions practicing different dose fractionation schedule of HDR ICBT for cervical cancer. The limitation of the present study is that we have used CT-based planning, which might have overestimated the CTVHR volume as reported by few studies [12,22]. In future, we plan to adopt MRI-based brachytherapy for cervical cancer.
Table 3
Equieffective dose in 2 Gy in available literatures
| Literature | Year of study | No. of patients | EBRT dose (Gy) | BT dose × fractions | Bladder 2 cm3 (Gy3) | Rectum 2 cm3 (Gy3) | Sigmoid 2 cm3 (Gy3) | D90 CTVHR (Gy10) |
|---|---|---|---|---|---|---|---|---|
| Pötter et al. [9] | 2011 | 156 | 50.4 | 7 Gy × 4 | 86 | 65 | 64 | 93 |
| Jamalludin et al. [10] | 2016 | A | 48.6 | 7 Gy × 4 | 80.8 | 81.7 | 73.9 | – |
| B | 45.4 | 7.5 Gy × 3 | 80.4 | 82.8 | 65.9 | – | ||
| Kirisits et al. [11] | 2005 | 22 | 45 | 7 Gy × 4 | 83 | 64 | 63 | 87 |
| Beriwal et al. [12] | 2011 | 44 | 45 | 5-6 Gy × 4 | 79.7 | 57.5 | 66.8 | 83.3 |
| Tharavichitkul et al. [13] | 2013 | 47 | 45 | 6.5-7 Gy × 4 | 88.2 | 69.6 | 72 | 93.1 |
| Nomden et al. [14] | 2013 | 46 | 45 | 7 Gy × 4 | 83 | 66 | 61 | 84 |
| Wang et al. [15] | 2016 | 42 | 46 | 5 Gy × 5 | 86 | 72.9 | 73.27 | 73.82 |
| Present study | 50 | 45 | 5.5 Gy × 5 | 74.06 | 67.43 | 54.56 | 85.9 | |
| 45 | 6.5 Gy × 4 | 73.75 | 67.57 | 54.43 | 84.67 | |||
| 45 | 7 Gy × 4 | 77.27 | 71.11 | 55.44 | 89.03 | |||
| 45 | 9 Gy × 2 | 68.58 | 64.82 | 52.14 | 69.16 |
Conclusions
EQD2 of bladder, rectum, sigmoid, D90 CTVHR, and point A is similar with dose fractionation schedule such as 5.5 Gy × 5 fractions, 6.5 Gy × 4 fractions, and 7 Gy × 4 fractions. EQD2 of 9 Gy × 2 fractions was significantly unfavorable compared to other schedules. Since it is a dosimetric study, further clinical studies are suggested to analyze the clinical outcome.