Introduction
In 1996, for the first time in Ukraine, an educational program in pediatric immunology started at the Department of Pediatric Infectious Diseases and Pediatric Immunology, Shupyk NMAPE (Kyiv, Ukraine). The immunologists of the department focused their efforts on the detection of children with primary immunodeficiency diseases (PIDs). Back then, there was a lack of data on the presence and number of PIDs in Ukraine. In order to improve the quality of health care for children with congenital immune pathologies, the Kyiv Children’s Immunological Centre was established in 1999 at the initiative of Professor L. Chernyshova. Owing to this step, pediatric PID patients got access to the appropriate health care. Since its creation, the Kyiv Children’s Immunological Centre has been working as a national reference center providing assistance in the diagnosis and treatment of PID patients from all regions of Ukraine. The department employees have been advising children from Kyiv and other cities and regions of Ukraine.
To overcome the major problem related to the low awareness and alertness of primary care physicians in managing children with PID, within the J Project, an educational campaign was launched for physicians under the guidance of Professor Laszlo Marodi in March 2004 [1]. The first Ukrainian J Project meeting was held in Kyiv. Pediatric immunologists from various countries – participants of the J Project – attended the event.
At that time, there were already pediatricians who specialized in pediatric immunology at the Department of Pediatric Infectious Diseases and Immunology, Shupyk NMAPE. Pediatric immunologists worked in all central cities of Ukrainian regions. Furthermore, other reference centers (Lviv, Kharkiv) and regional centers were established.
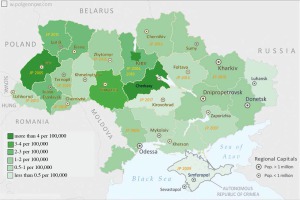
Annually, the J Project covered a new region in Ukraine, engaging hundreds of clinicians. Meetings were held in various regions of the country. Since 2004, 16 J Project Ukraine meetings have been held, covering more than half of the country’s regions (Fig. 1). Educational meetings have been conducted mainly in regions with a lower number of PID patients. They have involved lectures, workshops and case presentations for primary care physicians on early PID detection. Overall, several hundred physicians attended each J Project meeting, including pediatricians, general practitioners, and other health professionals. Educational materials on warning signs and early PID diagnosis have been distributed among physicians. Within the programs of all those meetings, there have been set up advanced educational sessions for pediatric immunologists.
Fig. 1
J Project meetings map in Ukraine and the incidence of primary immunodeficiency disorders (PIDs) in different regions
Representatives from different countries, including Hungary, Poland, Macedonia, Italy, Czech Republic, Turkey, Belarus, Russia, Latvia, Moldova and Greece, have taken part in the Ukrainian J Project meetings.
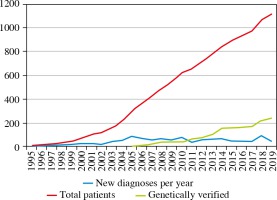
Due to the spread of the J Project activity in many regions of the country, the diagnosis of PIDs has been improved significantly. In contrast to 1997, when the first two patients were found to have Bruton’s disease, more than 1,000 patients were diagnosed with PID in 2020 (Fig. 2). In 2003, the National Registry for Primary Immunodeficiencies was implemented in Ukraine.
The J Project promoted the creation of a network of doctors, biologists and scientists in Eastern and Central Europe for better cooperation in health care and research into PID. Such fruitful cooperation of pediatric immunologists from various countries within J Project enabled us to diagnose rare PIDs and confirm them with genetic diagnosis.
Most J Project countries, including Ukraine, do not yet have their own genetic diagnosis and still require support from Western Europe and USA centers. One of the goals of the J Project, which is defined by the Antalya Declaration (http://www.jprojectnetwork.com/Page/AboutJProject/Antalyadeclaration), is free access to genetic testing for all patients. Debrecen PID Center led by Professor Laszlo Marodi had never abandoned any specimen sent for genetic diagnosis until 2015, when the laboratory was unreasonably closed.
Prevalence and incidence of primary immunodeficiency disease
The prevalence of PID in Ukraine was estimated by calculation of the number of living PID patients per 100,000 inhabitants. We used the data collected for the PID registry. Prevalence calculations were based on Ukrainian population data sourced from the National Statistics Committee of Ukraine. The incidence of PID was calculated using data on the year of birth of patients and the number of live births in the respective year.
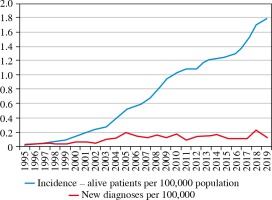
To date, the overall prevalence of PID in Ukraine is 1.78 cases/100,000 inhabitants (in 2014, the prevalence was 1.22/100,000, in 2017 it was 1.46/100,000). Estimates of PID prevalence do not include the patients with transient hypogammaglobulinemia of infancy and benign childhood neutropenia (Fig. 3).
The incidence of PIDs cases calculated over an 18-year period (2001-2019), is 10.0 per 100,000 live births. When comparing the data before and after initiating the J Project in Ukraine, we found striking differences in values with the incidence of PID being 5.0/100,000 children born alive by 2004, and 9.9/100,000 children born alive between 2004 and 2019.
National primary immunodeficiency disease registry
The National Registry for Primary Immunodeficiencies was established in 2015 and the data on all patients with PID diagnosed in 27 regions of Ukraine between 1996 and 2014 were incorporated. The information on patients from Crimea and Sevastopol collected until 2013 was also included. All pediatric immunologists experienced in the care of PID patients were invited to collaborate with the Kyiv Children’s Immunological Centre. The registry is regularly updated. Most PID patients are monitored at the Department of Pediatric Infectious Diseases and Immunology. The registry also includes the information on PID patients registered at other immunological centers of Ukraine (Lviv, Kharkiv) [2].
We collected the data in paper format, including demographics and key medical data (date of PID diagnosis, date of symptom onset, main clinical features, laboratory tests) and created the database. PIDs were classified according to the International Union of Immunological Societies’ (IUIS) criteria [3]. The patients were diagnosed with PID pursuant to the guidelines of the European Society for Immunodeficiencies (ESID) [4].
The registry database was established and hosted by the Kyiv Children’s Immunological Centre, which functions as a reference center. The research was carried out in accordance with the principles of the Helsinki Declaration. The study protocol was approved by the Ethics Committee of Shupyk NMAPE and local ethics committees of participating centers. Informed consent was obtained from patients or parents prior to inclusion in the registry.
At the beginning of 2020, the total number of patients who were entered into the Ukrainian PID Registry was 1,123 [684 (60.9%) males, 439 (39.1%) females]. Of these, there were 953 live patients in the registry, 136 (12.1%) patients died and 34 patients were lost to follow-up. Of the living patients, children (n = 680 of 953) outnumbered adults, 273 adult patients were aged > 18 years. Among the deceased patients, 125 died in childhood, 11 patients died aged > 18 years.
The PID Registry included 187 children with transient hypogammaglobulinemia of infancy and benign childhood neutropenia, whose manifestations disappeared with age. Excluding the patients with the above transient conditions, 766 living PID patients are being followed up; among them 257 patients are aged > 18 years.
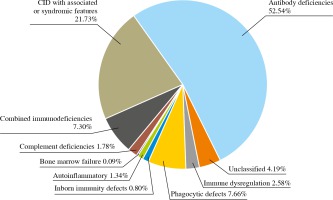
In general, the distribution of the main PIDs in Ukraine is similar to the structure in European countries (Fig. 4). Compared to the European registry, there is the same pattern with regard to the proportion of antibody deficiencies (54% ESID data), combined immunodeficiencies with syndromic features (18%) and combined immunodeficiencies (7%) [5, 6]. The Ukrainian registry presents 64 specified nosological PID entities. Antibody deficiencies are the most common subcategory of PID [590/1,123 (52.54%)], followed by combined immunodeficiencies (CID) with associated or syndromic features [244/1,123 (21.73%)], CID [(82/1,123 (7.3%)], congenital defects of phagocyte number and function [86/1,123 (7.66%)], diseases of immune dysregulation [29/1,123 (2.54%)], complement deficiencies [20/1,123 (1.78%)] and autoinflammatory disorders [20/1,123 (1.34%)].
Fig. 4
Distribution of primary immunodeficiency disorder (PID) groups in primary immunodeficiency registry in Ukraine
The most common nosological entities are minor antibody deficiencies: selective IgA deficiency – 206 (18.3%) and transient hypogammaglobulinemia of infancy – 220 (19.5%). Among the severe immunodeficiencies, the most represented are X-linked agammaglobulinemia – 66 (5.8%), chronic granulomatous disease (CGD) – 28 (2.5%) and CID with syndromic features, including syndrome DiGeorge – 71 (6.3%), Nijmegen syndrome – 53 (4.7%), ataxia-telangiectasia – 51 (4.5%), and Wiskott-Aldrich syndrome (WAS) – 29 (2.6%). Severe combined immunodeficiencies (SCID) account for 63 cases (5.6%).
To date, PID has been genetically confirmed in 300 patients in Ukraine, who constitute 26.7% of all patients in the PID registry (Table 1).
Table 1
Genetic defects of 300 primary immunodeficiency disorder (PID) patients in Ukraine
[i] CD40LG – CD40 ligand gene, MALT1 – mucosa-associated lymphoid tissue lymphoma translocation protein 1 gene, RAG1 – recombination activating 1 gene, DOCK8 – dedicator of cytokinesis 8 gene, IL7R – IL7 receptor gene, ADA – adenosine deaminase gene, IL-2RG – interleukin 2 receptor subunit gamma gene, LIG4 – DNA ligase 4 gene, DCLRE1C – DNA cross-link repair 1C gene, WAS – Wiskott-Aldrich syndrome gene, ATM – ataxia telangiectasia mutated gene, NBS1 – Nijmegen breakage syndrome 1 gene, STAT3 – signal transducer and activator of transcription 3 gene, Del 22q11.2 – chromosome 22q11.2 deletion syndrome, SPINK5 – serine peptidase inhibitor kazal type 5 gene, PMRP – POP1 homolog, ribonuclease P/MRP subunit gene, KMT2D – lysine methyltransferase 2D gene, BTK – Bruton tyrosine kinase gene, PIK3CD – phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit delta gene, PIK3R1 – phosphoinositide-3-kinase regulatory subunit 1 gene, TNFRSF13B – TNF receptor superfamily member 13B gene, IGLL1 – immunoglobulin lambda like polypeptide 1 gene, NFKB2 – nuclear factor kappa B subunit 2 gene, AICDA – activation induced cytidine deaminase gene, SH2D1A – SH2 domain containing 1A gene, TNFRSF6 – tumor necrosis factor receptor superfamily, member 6 gene, CXCR4 – C-X-C motif chemokine receptor 4 gene, CTLA4 – cytotoxic T-lymphocyte associated protein 4 gene, AIRE – autoimmune regulator gene, FOXP3 – forkhead box P3 gene, RAB27A – RAB27A, member RAS oncogene family gene, LRBA – LPS responsive beige-like anchor protein gene, CYBB – cytochrome b-245 beta chain gene, CYBA – cytochrome b-245 alpha chain gene, FERMT3 – fermitin family member 3 gene, HAX1 – HCLS1 associated protein X-1 gene, ELANE – elastase, neutrophil expressed gene, SBDS – SBDS, ribosome maturation factor gene, IL-12R – interleukin 12 receptor subunit beta 1 gene, ITGB2 – integrin subunit beta 2 gene, RAC2 – Rac family small GTPase 2 gene, GATA2 – GATA binding protein 2 gene, IRF8 – interferon regulatory factor 8 gene, STAT1 – signal transducer and activator of transcription 1 gene, SMARCAL1 – SWI/SNF related, matrix associated, actin dependent regulator of chromatin, subfamily a like 1 gene, CTSC – cathepsin C gene, NOD2 – nucleotide binding oligomerization domain containing 2 gene, MVK – mevalonate kinase gene, ADA2 – adenosine deaminase 2 gene, NLRP3 – NLR family pyrin domain containing 3 gene, MEVF – MEFV innate immunity regulator, pyrin gene, NLRP12 – NLR family pyrin domain containing 12 gene, NFAT5 – nuclear factor of activated T cells 5 gene, TNFRSF1A – TNF receptor superfamily member 1A gene, TINF2 – TERF1 interacting nuclear factor 2 gene, TCN2 – transcobalamin 2 gene, FANCC – FA complementation group C gene, RTEL1 – regulator of telomere elongation helicase 1 gene, TERT – telomerase reverse transcriptase gene
Education campaign
Despite the progress in PID diagnosis in Ukraine over the last 15 years, we have encountered the challenges related to the recognition of PIDs, especially some novel immunodeficiency groups (defects of immune dysregulation, autoinflammatory disorders, defects of innate and intrinsic immunity).
In 2017, the Ukrainian Association of Pediatric Immunology initiated the series of clinical conferences named RAPID (Rapid Diagnostics of PID) with the main aim of improving diagnosis in regions through on-site patient counseling.
In 2018, the J Project meetings in Ukraine changed the format from focusing only on primary care physicians to the involvement of health professionals of other subspecialties. A meeting with hematologists was held in 2018 to increase the number of identified patients with immune defects and cytopenias that had been virtually undiagnosed before. This was also facilitated by the expansion of genetic diagnosis through commercially available panel sequencing (Invitae Laboratory, USA). Collaboration with hematologists has also triggered the development of bone marrow transplantation in Ukraine. As of today, a registry of bone marrow donors in Ukraine has been established and it is constantly being updated (https://ubmdr.org).
Improved PID diagnosis and treatment in Ukraine contributed to the increased number of patients surviving to adulthood. This posed new challenges to the immunological community concerning the continuity in the management of patients between pediatric and adult specialists involved in PID management. For the first time, the J Project Meeting in Ternopil (2019) brought together our country’s pediatric and adult immunologists. A consensus was reached regarding the joint actions to be taken and implementation of the joint patient registry. For the first time, an adult patient with autoinflammatory disorder (ADA2 deficiency) was referred for transplantation.
We believe that focusing on the interdisciplinary collaboration is a promising direction for the further development of the J Project in Ukraine. Obviously, severe cases are most concentrated among doctors of different specialties (pulmonologists, rheumatologists, hematologists, etc.). Therefore, it is important to increase the alertness among primary care physicians, but also among doctors of other specialties who are not sufficiently aware of PID issues today. In the future, J Project meetings together with pulmonologists, rheumatologists, cardiologists, and allergists are planned.
Collaboration in scientific research
The J Project has a close professional partnership with the ESID. Thanks to the J Project, pediatric immunologists from Ukraine have entered the European medical space [7, 8]. Cooperation in the field of PID was established with colleagues from Poland, Hungary, Belarus, and Iran [9, 10].
The J Project has also gradually transformed from an educational campaign for physicians to a collaborative research program aimed at identifying the mutational spectrum of genes associated with PID and analyzing unique genotype-phenotypic relationships in Eastern and Central Europe. Thus, in 2011, two groups of researchers from different countries (one group included L. Chernyshova, A. Bondarenko, and V. Chernyshov from Ukraine) described a new variant of chronic mucocutaneous candidiasis (CMCC) caused by gain-of-function mutations in the gene STAT1 [11-14].
As of today, within the framework of the Cooperation Agreement, a scientific project is jointly being implemented with the Republican Centre of Pediatric Oncology, Hematology and Immunology of Belarus – “The genetic landscape of SCID in Eastern Europe” – according to the grant of the scientific program CHILDREN (Jeffrey Modell Foundation, JMF) [15, 16].
The support in genetic diagnosis enabled us to diagnose several PIDs and study the clinical features and prevalence of immunodeficiencies in the Ukraine. The results of these scientific studies were summarized in research theses and articles, presented at conferences [17-21]. Given that PIDs are rare diseases, we can make reasonable conclusions only based on collecting, summarizing and analyzing data from different countries. The results of multicenter studies of patients with various PIDs are important for the development of novel methods of treatment. Such results are presented in publications of scientific teams combining efforts of scientists from different countries [22-26].
Progress with treatment of primary immunodeficiency disease
The J Project has spread and achieved a significant success not only in the number of diagnosed patients, but also in arranging their treatment. In 2011, the National Program of Replacement Therapy with Intravenous Immunoglobulin for Children with Predominant Antibody Deficiencies was approved. In 2019, a similar program was approved for adult patients. To date, all 194 patients with primary immunodeficiencies requiring replacement therapy with immunoglobulins are provided with medications. Most patients receive intravenous immunoglobulin, but in 2019, 16.1% of children and 26.2% of adults switched to subcutaneous immunoglobulin.
Hematopoietic stem cell transplantation (HSCT) was performed in 40 PID patients out of 156 children potentially needing this treatment. It was successful for 30 children. In the National Clinic OCHMATDYT, HSCT was performed successfully from family donors to six children with PIDs [WAS – 3, CGD – 1, hyper-IgE syndrome (mutation of DOCK8) – 1, and CMCC (STAT1 GOF mutation) – 1]. Most patients with PID were transplanted in clinics of other countries [27]. Among other treatments for patients with PID are sirolimus for children with autoimmune lymphoproliferative syndrome (ALPS) and activated PI3K delta syndrome (APDS), colony-stimulating factor for patients with neutropenia, abatacept for patients with cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) haplo-insufficiency, and some children with PID receive antibacterial and antifungal prophylaxis. Children with autoinflammatory disorders are treated with interleukin 1 (IL-1) antagonists, anti-IL-1β antibodies, colchicine and monoclonal antibodies against tumor-necrosis factor. The therapy with C1-inhibitor for patients with hereditary angioedema will start soon.
Significant support and success in achieving the J Project core goals have been reached through the activity of the non-governmental organization Rare Immune Diseases, which was founded in 2014 and represents the interests of the patient community. Progress in the J Project contributed to an increase in the number of identified patients and their integration, and introduction of other countries’ experience in patient communities. In our country, the patient and professional communities are in close friendships, helping and supporting each other in achieving common goals.
Development of regional immunological centers (Lviv, Ternopil)
The Western Ukrainian Center for Pediatric Immunology in Lviv led by Professor L. Kostyuchenko, has been working hosted by the multidisciplinary children hospital Western Ukrainian Specialized Pediatric Medical Centre since 2001. In 2019, the center joined the Pediatric Rheumatology Service and was renamed as the Western Ukrainian Clinic of Pediatric Immunology and Rheumatology, which has opened new opportunities for successful detection and treatment of immunodeficiencies of the group of autoinflammatory syndromes.
The database of patients with PIDs numbers over 300 patients and is one of the largest in Ukraine. In cooperation with the Institute of Hereditary Pathology of the National Academy of Medical Sciences of Ukraine, genetic diagnosis of Nijmegen syndrome was established and a database of patients and their families was created. Prenatal diagnosis was carried out in high-risk families with Nijmegen syndrome. The Western Ukrainian Clinic of Pediatric Immunology and Rheumatology actively cooperates with European centers which solve similar problems, including issues of genetic diagnostics of immunodeficiencies and their treatment.
With successful operation of the J Project in Ukraine, one more new center has been established recently. To improve PID detection in the Ternopil region, the enthusiastic group lead by Prof. O. Boyarchuk launched the project entitled “Implementation of the model of combined physician education and public awareness of the infrastructure to diagnose primary immunodeficiency diseases in children” in 2016. The project received support from the Jeffrey Modell Foundation. The major aim of this project was to improve PID detection in children at early stages through implementation of the methodology combining education of the physicians, increased public awareness, immunological testing, and effective medical support at various stages of PIDs. Education of primary care physicians and other specialists on early PID detection in every district of the Ternopil region raised the awareness of PIDs and improved early diagnosis. Within the first 2 years of the program the number of detected PIDs increased almost threefold, and in comparison to the two-year period before the initiation of the awareness campaign (2015-2016), it was almost fivefold higher (2 cases/year before vs. 9 cases/year during the awareness campaign) [28].
Collaboration with other Ukrainian pediatric immunology centers and colleagues from Poland, the USA (Yale University Pediatric Genomics Discovery Program, Dr. Carrie Lucas), Belarus, and Greece was very fruitful in PID diagnosis. Currently, there are 50 patients in the Ternopil Regional Registry. They represent seven groups of PID; 36 of the above patients are alive, the diagnosis has been confirmed with genetic testing in 21 individuals (42%), and 7 of them have been referred to adult immunologist follow-up care.
During the last three years, a number of research papers reflecting very productive work of the Kyiv Children’s Immunological Centre’s team have been published [29-35]. The center continues the implementation of the J Project principles in the regional centers and conducts targeted screening of children at risk of PID. The group is enthusiastic to start a pilot study in Ukraine on the neonatal screening of PIDs through quantifying T-cell receptor excision circles (TRECs) and kappa-deleting recombination excision circles (KRECs) to identify T- and B-cell lymphopenia.
Collaboration between the countries involved in the J Project and other countries in the field of molecular diagnosis of PID is vital. On the other hand, we should develop on-site molecular genetic laboratories and update the existing laboratories in Ukraine.
In conclusion, the future perspectives of the J Project in Ukraine include the development of professional multidisciplinary cooperation in management of patients with PIDs, change of formats of measures on raising the awareness of PIDs among the medical fraternity (consulting help to doctors in regions for active identification of patients with PIDs, expanding the audience of health workers, the dissemination of information on PIDs among the population), and conducting educational activities among nurses. It is critically important to improve PID diagnoses, including genetic diagnosis, and ensure access to all necessary treatment for PID patients. We should direct all our efforts towards the development of a bone marrow donor registry and HSCT in Ukraine.