Purpose
Endometrial cancer is the second most frequent gynecological malignancy worldwide, and its prevalence is more common among high-income countries [1]. Most of these cases are diagnosed at an early-stage due to post-menopausal vaginal bleeding as an early warning sign [2]. Total abdominal hysterectomy and bilateral salpingo-oophorectomy with or without lymphadenectomy is the mainstay of management. The type of surgery, especially in terms of lymphadenectomy, varies depending on the tumor size, grade, and depth of myometrial invasion of the tumor [2,3]. Post-operative radiotherapy (RT), either external beam RT (EBRT) and/or vaginal brachytherapy (VBT), is recommended for patients with early-stage disease based on the presence of adverse risk features [4,5,6]. The results of multiple randomized trials demonstrated a locoregional control benefit with adjuvant RT over observation alone, but its effect on survival is controversial [7,8].
In this study, we aimed to investigate a long-term survival outcomes and prognostic factors affecting survival in patients with early-stage endometrial cancer.
Material and methods
Patients diagnosed with non-metastatic uterine adenocarcinoma who underwent total hysterectomy and bilateral salpingo-oophorectomy between 2006 and 2016 at two institutions were analyzed. The patients were restaged according to the 2009 International Federation of Gynecology and Obstetrics (FIGO) staging system, and data of 311 patients with stage I-II endometrial cancer who received ± adjuvant treatment were included and retrospectively reviewed. The patients’ files, electronic information system, RT records, and telephone interviews were used to collect the data. The collected and analyzed details included age at diagnosis, date of surgery, type of surgery, inclusion of lymph node dissection, histology, grade, depth of myometrial invasion, presence of lymphovascular space invasion (LVSI), type of adjuvant chemotherapy, type of adjuvant radiation, dose of radiation, dates of radiation, date of recurrence, type of recurrence (if applicable), date of last follow-up, and date of death.
All the patients underwent primary surgical treatment, as total abdominal hysterectomy and bilateral salpingo-oophorectomy ± bilateral pelvic paraaortic lymph node dissection. Post-operative RT either EBRT and/or VBT were suggested to patients with adverse risk factors. Patients with high-grade histologies (non-endometrioid and endometrioid grade 3) and stages beyond Ib usually received both the treatments, patients who presented one major risk factor usually obtained either VBT or EBRT, and patients with minor risk factors received no further therapy. Starting from late 2008, the patients were treated with 3D pelvic EBRT, but prior to 2008, a 2D box technique was performed. The linear accelerators with 6 MV or 15-18 MV energies were used at standard fractionation (180-200 cGy) to a total dose of 4,500-5,040 cGy. The anterior/posterior pelvic field borders were L4-L5 vertebra intersection superiorly, below the obturator foramen inferiorly, and 1.5-2 cm lateral to the widest portion of bony pelvis laterally. The lateral field borders were posterior of S3 and anterior part of pubic symphysis.
Vaginal brachytherapy was applied either as boost therapy following EBRT or as a sole treatment modality. Total VBT dose was 10-25 Gy/2-5 fractions prescribed to vaginal surface or mucosa, defined as 5 mm depth from the applicator surface depending on a boost occurrence. Adjuvant chemotherapy (carboplatin and paclitaxel) was administered to patients with high-grade non-endo-metroid histology.
The primary endpoints of the present study included disease-free survival (DFS) and disease-specific survival (DSS). Although, all the recurrence sites were noted separately, since we acquired various information with phone calls and we could not confirm certain old hospital data, we chose DFS as the endpoint. Moreover, the majority of patients with this diagnosis were senior with comorbid diseases, therefore we used DSS in order to present the association between variables and survival without interference. The diagnosis date was accepted as the onset point for disease-free and disease-specific survivals. The endpoint for DSS was the last control date for living patients. Patients who died were divided into two groups: as a decease due to the disease or not. Patients who died due to the causes other than endometrial carcinoma were excluded in the DSS analysis. The endpoint for DFS was the first event date for recurrence and distant metastasis, the last control date for patients who have not relapsed.
Adult patients with FIGO stages I-II endometrial cancer who were curatively treated with all accessible information were included in the study. Patients who received palliative RT, had missing files, and follow-up information were excluded from the study.
Statistical analysis
Statistical analyses were conducted using IBM SPSS Statistics version 20 (SPSS Inc., Chicago, IL, USA), and non-parametric tests were used to assess variables. The categorical demographic characteristics of enrolled patients were calculated with χ2 and Fisher’s exact tests. Spearman’s rank correlation test was utilized for univariate correlation analysis. Mann-Whitney U test was conducted for two groups of independent statistical analysis and Kruskal-Wallis test was performed for 3 or more independent group analyzes. After Bonferroni correction, the significance was evaluated by post-hoc analysis. The variables studied included the patient age, stage, degree of myometrial invasion, biggest diameter of tumor, tumor grade, lymphovascular invasion, lower uterine segment invasion, and adjuvant treatment. Kaplan-Meier test estimated the survival and with log-rank test were used for univariate analysis. Multivariate Cox regression analysis was performed to determine the effect of variables on survival. The endpoints included progression free survival (PFS) and DSS. All the study outcomes were measured from the date of surgery to address a potential lead time bias. P-values of less than 0.05 were considered statistically significant, and all statistical tests performed were 2-sided.
Results
The data of 311 patients from 2 institutions, with FIGO stage I-II endometrial cancer who underwent curative treatment were retrospectively evaluated. The median age was 60 (range, 33-85) years. All of the patients had surgery, 74 (24%) received no further treatment, 4 (1%) obtained chemotherapy only, 234 (75%) received RT, and 24 (7%) received both treatments. The pathology of 277 (89.4%) patients were endometrioid adenocarcinoma, the rest were non-endometrioid type. According to the FIGO staging system, 148 (47.9%), 108 (34.9%), and 57 (17.2%) of the patients were stage Ia, Ib, and II, respectively. Radiotherapy was applied to 106 (45.8%) of the patients as only VBT, and 128 (54.2%) of the patients as both EBRT and VBT. Patients and diseases characteristics are summarized in Table 1.
Table 1
Patients and disease characteristics
The median follow-up time was 102 (range, 3-205) months. During this period, 68 (21.9%) patients died, among whom 41 (62.8%) died due to disease (n = 28) or treatment-related (n = 13) causes, 24 (33.8%) due to other reasons, and 3 (4.4%) due to unknown causes. Three patients had second primary, with two having breast carcinoma and one bladder carcinoma. During the follow-up time, 26 (8.3%) patients experienced a recurrence, with 15 (6.4%) patients receiving RT and 11 (14.2%) no additional RT.
Results of disease-specific survival
The median DSS was 101 (range, 1-205) months. The 2-year, 5-year, and 10-year DSS were 93.6%, 86.8%, and 82.2%, respectively.
Among the aforementioned variables, the age, LVSI, grade, stage, and adjuvant RT were found to have significant relationship with DSS. The age below 65 years, LVSI, grade 3, stage II disease, and no adjuvant RT were the negative prognostic factors of DSS (Figure 1). Multivariate analysis also confirmed the importance of these variables (Table 2).
Table 2
Detailed analysis of disease-specific survival (DSS) according to the factors significantly affecting DSS
No significant difference in DSS was noted between the patients receiving either EBRT or VBT (median DSS, 100 (range, 4-142) months vs. 102 (range, 10-205) months).
Results of disease-free survival
The median DFS was 99 (range, 1-205) months. The 2-year, 5-year, and 10-year DFS were 86%, 76%, and 74.3%, respectively.
Disease-free survival was not significantly influenced by cervical stromal involvement (p = 0.19), pathological subtype (p = 0.34), the use of chemotherapy (p = 0.16), tumor size (p = 0.61), advanced age (p = 0.43), grade (p = 0.14), and stage (p = 0.34).
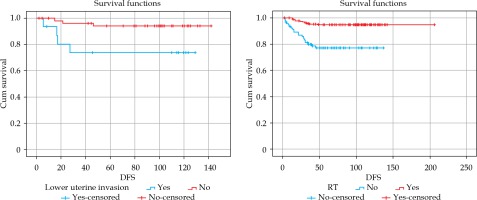
A significant correlation was found between lower uterine invasion and DFS (p = 0.015, hazard ratio [HR] = 0.19, CI: 0.04-0.85) (Figure 2). The 5-year and 10-year DFS were 73.7% and 73.5% vs. 94.2% and 93.7% in patients with lower uterine invasion vs. no invasion, respectively.
Similarly, the correlation between adjuvant RT and DFS was significant (p = 0.017, HR = 4.2, CI: 2.0-8.8) (Figure 2). The 5-year and 10-year DFS were 95% and 94% vs. 77.3% and 76% in patients with RT vs. without RT, respectively. In Table 3, the values of median (range) DFS were indicated according to the factors, which significantly affected DFS.
Discussion
In this study, we presented the 10-year survival results of 311 patients with early-stage endometrial cancer from two centers, a rather heterogeneous group with different approaches. All patients underwent surgery, 74 (24%) received no further treatment, 4 (1%) obtained chemotherapy only, 234 (75%) received RT, and 24 (7%) received both treatments. The 5-year and 10-year DFS were 76% and 74.3%, respectively. In multivariate analysis, lower uterine segment invasion positivity and no adjuvant RT were determined as independent unfavorable prognostic factors for DFS. The 5-year and 10-year DSS were 86.8% and 82.2%, respectively. For DSS, the high-grade, LVSI positivity, stage II, ≥ 65 age, and no adjuvant RT were found to be the independent unfavorable prognostic markers. As a result of the present study, adjuvant RT in early-stage endometrial cancer is a significant independent prognostic factor for both disease-free and disease-specific survival.
Adjuvant RT has a well-defined role in early-stage endometrial carcinoma; post-operative EBRT reduces isolated relapses from 14-12% to 4-3%, absolute benefit of nearly 10% in stage I patients [4,5]. In our cohort, comprised of stage I and II, 6.4% of patients who received RT experienced recurrence, a quite comparable result. Patients with stage I, grade 1, or grade 2 with < 50% myometrial invasion and endometrioid histology without other risk factors, are generally considered low-risk, and expected recurrence rate is < 5% [9]. Unexpectedly, the rate of recurrence rate was quite high in our patients without adjuvant RT. We presume that some of the patients with risk factors already necessitating adjuvant treatment did not receive RT, either because they were not offered or the pathology underscored grade or degree of myometrial invasion; this emphasize the importance of central pathology review in randomized trials.
The advantage of chemoradiotherapy over RT alone was not demonstrated in early-stage endometrial cancer [10]. In the recently published PORTEC-3 multicenter phase 3 study, the benefits of adjuvant chemotherapy and pelvic RT in high-risk endometrial cancer patients were examined. Patients were randomized to pelvic RT and chemotherapy (2 cycles of cisplatin concurrent with RT, and 4 cycles of carboplatin and paclitaxel following RT) and pelvic RT only (48.6 Gy) arms. In stage I-II patients, the advantage of survival was not achieved with adjuvant chemotherapy. In a subgroup analysis, adjuvant chemotherapy increased failure-free survival (FFS) only in stage III disease. This study demonstrated that pelvic control was achieved only by RT in early-stage endometrial cancer patients. In a review for evaluating adjuvant chemoradiotherapy versus RT alone in early-stage endometrial cancer patients, the superiority of adjuvant chemoradiotherapy over RT alone was not demonstrated in overall survival (OS) and FFS [11]. In the GOG 249 phase 3 study [12], 601 patients with high-risk early-stage endometrial cancer were randomized to pelvic RT, with 3 cycles of paclitaxel/carboplatin, and VBT arms. The primary endpoint was recurrence-free survival. However, the superiority of VBT plus chemotherapy over pelvic RT could not be revealed. The authors propounded that pelvic RT was the most effective and optimal adjuvant treatment option in early-stage endometrial cancer [12]. In a recent study, chemotherapy was applied, only in 9% of patients, which did not impact survival parameters.
Lin et al. [13] examined the role of adjuvant RT in 337 stage I endometrial cancer patients from a single center. 5-year OS and 5-year loco-regional recurrence-free survival (LR-RFS) were 96.3%, 97.2% for the group without RT, and 91.6%, 97.1% for the group with RT, respectively (p = 0.06 for OS and p = 0.956 for LR-RFS). In a multivariate analysis, > 60 years of age and > 50% myometrial invasion were significant prognostic factors for OS. They revealed pre-operative CA-125 level, > 60 years of age, LVSI, and adjuvant RT as independent prognostic factors for LR-RFS. In the conclusion of the study, adjuvant RT was an independent predictor for LR-RFS. However, the authors failed to demonstrate its effect on OS. Similarly, in our study, the advanced age, absence of adjuvant RT, and LVSI positivity were found to be independent prognostic factors for DSS. In two other similar retrospective studies, the presence of LVSI has been denoted to be associated with vaginal relapse and poor OS [14,15].
Yılmaz et al. [16] investigated the impact of adjuvant RT on survival in patients with early-stage high-grade endometrial cancer. Adjuvant RT was administrated to 57% of patients as pelvic RT (average dose of 46 Gy) and the rest as VBT (average dose of 21.5 Gy). There was no significant relationship between adjuvant RT and overall and DFS. Elevated serum CA-125 level and histological grade were independent predictors for DFS. In contrast, in the present study, the adjuvant RT was an independent prognostic factor for both disease-specific and disease-free survival. We indicated that the absence of adjuvant RT and lower uterine segment invasion were negative prognostic factors for DFS.
Due to the concerns about potential toxicity of EBRT, the PORTEC-2 trial was designed to illustrate if VBT would be sufficient instead of EBRT as an adjuvant RT in early-stage endometrial cancer [6]. This trial was non-inferior study comparing EBRT and VBT alone. Vaginal recurrence was 0.9% and 2% in the VBT alone and EBRT arms, respectively, and this difference was not statistically significant. Furthermore, there was no statistical difference between the two arms at 5-year OS rates (82% vs. 86%). Vaginal brachytherapy has been indicated to cause less morbidity than EBRT, while providing similar vaginal tumor control. Kim et al. [17] reported the treatment results of post-operative VBT and/or EBRT in patients with stage I endometrial cancer. The 5-year DFS was 88% in the EBRT group and 96% in the VBT group. Similarly, the 5-year OS was 94% in the EBRT group and 96% in the VBT group. The results were not significant due to low number of patients and events. However, they argued that VBT may be preferred in selected patients due to lower than EBRT toxicity and similar treatment outcomes [17]. In our series, we also demonstrated no survival difference between patients receiving EBRT and VBT.
In the present study, the main goal was to report long-term survival results of patients with early-stage endometrial cancer. Data presented the results of 15-year follow-up, and these results are the conclusion of the actual treatment executed without any randomization as in phase 3 studies, which reflects the real-world data, and makes the current study valuable. Moreover, we reported 10 years of survival in this study, whereas many studies in the literature reported 5 years of survival [4,5,10]. We submitted the results of DFS similar to FFS: the 5-year DFS was 76% and the 10-year DFS was 74%. In the literature, 5-year DFS rates for patients with early-stage endometrial cancer ranged from 75% to 88% [16,17,18].
This study has some limitations. It was retrospective in nature involving a heterogeneous group of patients. Failure to separate the treatments according to the risk groups restricts the study. Moreover, differentiating the site of recurrence could help to understand the results better. This study evaluated data of patients from a period of 15 years, and potential variation/evolution in standards of management can be a disadvantage while interpreting results.
Conclusions
At 15 years of follow-up, the adjuvant RT has been shown to be the significant independent prognostic factor for both DSS and DFS. Radiotherapy should remain the essential part of the treatment in the management of early-stage endometrial cancer.
The abstract of current study was presented as an oral presentation in the Second Black Sea Gynecology and Obstetrics Congress.