Purpose
Endometrial carcinoma is the most common gynecological cancer in developed countries [1]. Most women present with early-stage disease, and have a good prognosis. The standard of care includes surgery (radical hysterectomy with bilateral salpingo-oophorectomy) with or without evaluation of pelvic and/or para-aortic lymph nodes. Adjuvant treatment with vaginal-cuff brachytherapy (VCB), external beam radiotherapy (EBRT), and/or chemotherapy (CT) is recommended based on surgical stage and known risk factors for recurrence, such as age, nuclear grade, histological sub-type, depth of myometrial invasion, and lymphovascular space invasion (LVI) [2].
Vaginal-cuff brachytherapy is the standard adjuvant treatment in women with high-intermediate risk endometrial cancer [3], and is frequently used in combination with pelvic EBRT and/or chemotherapy in patients with high-risk disease, who are at an increased risk of relapse and cancer-related death [4]. Nowadays, high-dose-rate (HDR) brachytherapy is used to treat 96% of patients who need brachytherapy [5]. Main advantages of HDR include outpatient delivery and decreased radiation exposure to patients and healthcare providers.
The most commonly used applicator is a single-channel vaginal cylinder with iridium-192 (192Ir) source. In the past few years, cobalt-60 (60Co) sources have become more popular [6] due to, in part, a longer half-life of 60Co (5.27 years) compared to 192Ir (74 days), requiring less frequent source replacement (3 months intervals vs. 5 years). Differences in their emission spectrum results in TG43 radial functions behaving differently, and being higher in 192Ir up to about 20 cm in water [7, 8]. This potential advantage for organs at risk (OARs) dosimetry for 60Co source is greatly diminished by the impact of inverse square of distance effect. In both sources, electrons from β– decay are absorbed by the core and the capsule [9]. Finally, clinical plans are very similar in both cases [8, 10, 11]. Higher energy of 1.25 MeV of 60Co compared to 0.397 MeV of 192Ir has historically raised concerns about the efficacy, toxicity, and quality of life of patients treated with 60Co HDR brachytherapy [7].
The main objective of this study was to report treatment dosimetry parameters, and evaluate treatment outcomes and toxicity of patients with endometrial cancer treated with 60Co source in HDR brachytherapy. In addition, this study intended to compare our results with those reported in literature.
Material and methods
This was a retrospective study to report VCB dosimetry parameters and outcomes of all patients with localized endometrial cancer treated with adjuvant HDR vaginal brachytherapy (VBT) with a 60Co source at our institution from January 2011 to February 2017. We identified a total of 93 patients with FIGO stage IA-IIIC2 endometrial cancer, all of which underwent surgical staging with a total hysterectomy, bilateral salpingo-oophorectomy with or without pelvic and/or para-aortic lymph node assessment, and/or peritoneal cytology. External beam radiation therapy was administered in all patients with high-risk factors (stage III and stage IA-III non-endometrioid with myometrial invasion) and in most patients with high-intermediate risk factors (substantial lymphovascular space invasion, stage II and/or stage IBG3 endometrioid). Adjuvant chemotherapy was administered in patients with high-risk histology, stage III, and some patients with cumulative individual high-risk factors (deep myometrial invasion, high grade and/or substantial LVSI).
2 mm CT-based brachytherapy simulation was applied in all patients in each application for treatment planning. Foley catheter was used to empty the bladder, and then it was filled with 50 cc of saline. Additionally, 30 cc of contrast were used in the rectum for contouring. A catheter to decompress a gas-filled rectum was employed when needed. Contouring of the rectum (including sigmoid volume) and bladder followed the GEC-ESTRO guidelines [12].
All patients received HDR VBT using a single-channel vaginal cylinder. The most frequently used dose-fractionation schedules were 21 Gy in three weekly fractions of 7 Gy (exclusive VCB) or 11 Gy in two fractions of 5.5 Gy after pelvic EBRT (range, 46-50.4 Gy). Dose was prescribed to a depth of 5 mm from the applicator surface, and target included the upper third of the vagina; a 3.5 cm treatment length was used in most patients (83.9%). Diameter size of the vaginal cylinder ranged between 2.5 and 3.5 cm. Clinical dosimetry was calculated using HDRPlus (Eckert & Ziegler) and Sagiplan (HDRPlus evolution) software in all patients. All patients were treated using a Multisource HDR Unit (Eckert & Ziegler).
Dosimetry variables analyzed included EBRT prescription dose, maximal dose (Dmax), dose to the most exposed 2cc (D2cc), 1cc (D1cc), and 0.1cc (D0.1cc) for both the rectum and bladder as well as the distance from cylinder surface to the 150% and 200% isodose line (100% = prescription dose) in vaginal mucosa (lateral and from the top of applicator). Equivalent total doses in 2 Gy per fraction (EQD2) were calculated using the following equation: EQD2 = nd (d + α/β)/(2 Gy + α/β), where n is the number of fractions and d is the dose (Gy) per fraction (assuming α/β = 10 Gy for tumor, and α/β = 3 Gy for late normal tissue damage).
After the treatment, all patients were assessed by their radiation oncologist. Follow-up included pelvic examination 4-6 weeks after treatment, every 3-6 months for the first 2 years, and then every 6-12 months until death.
Clinical outcomes analyzed included local recurrence (LR), regional recurrence (RR), distant metastasis (DM), overall survival (OS), and progression-free survival (PFS). OS and PFS were obtained from the day of surgery. For patients who were lost to follow-up, data were censored at the time of last follow-up. For toxicity analysis, physician-assessed genitourinary, rectal and gynecologic toxicities were retrospectively collected from available follow-up notes according to the National Cancer Institute common terminology criteria for adverse events (CTCAE), version 3.0. Acute toxicity was defined as symptoms during ≤ 6 months after VBT, and chronic toxicity as > 6 months after VBT.
All data were analyzed using SPSS statistical analysis and compared using χ2 test and Fisher’s exact test. Clinical outcomes were analyzed using Kaplan-Meier, and log-rank test was applied to assess differences between groups. Toxicity evaluation was assessed using SPSS and MATLAB software for possible cross-correlation within dosimetric parameters by using Pearson r test and stepwise multivariate linear regression.
Results
A total of 93 patients with endometrial cancer, who received adjuvant VBT were identified eligible for inclusion. Mean age at diagnosis was 66 years (range, 45-85 years). The most common histological sub-type was endometrioid adenocarcinoma (61.3%), followed by papillary-serous carcinoma (11.8%) and clear cell carcinoma/ uterine carcinosarcoma (7.5% each group). There were 71% patients with FIGO stage I (35.5% IA and 35.5% IB), 11.8% with stage II, and 17.2% with stage III. Baseline patients’ characteristics are shown in Table 1. All patients were treated with radical surgery (hysterectomy and bilateral oophorectomy) plus VBT. Lymph node assessment was performed in 70 patients (75.3%). Adjuvant EBRT (range, 46-50.4 Gy) was performed in 50 patients (53.8%), and 36 patients (38.7%) also received adjuvant chemotherapy. Treatments’ characteristics are listed in Table 2.
Table 1
Patients’ and tumors’ baseline characteristics
Table 2
Treatment characteristics
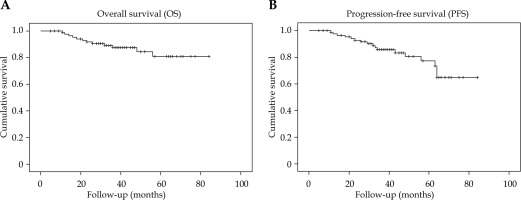
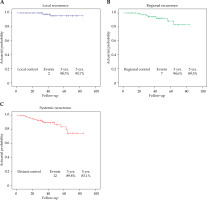
The median follow-up time was 39 months (range, 5-84 months). The 3-year OS rate was 87.5% and PFS was 85.5%. The 5-year OS and PFS rates were 81% and 77.2%, respectively. Survival rates are shown in Figure 1. The relapse patterns were analyzed and vaginal recurrence was seen in 2 patients, 7 patients experienced regional recurrence, and 12 patients developed distant metastasis. The 3- and 5-year estimates for LR, and RR are summarized in Figure 2.
Fig. 2
Actuarial Kaplan-Meier estimates for local control (A), regional control (B), and distant metastasis control (C). Absolute number of events and actuarial estimates for outcomes at 3 and 5 years are indicated
Dosimetric parameters for organs at risk (rectum and bladder) and distance from the applicator surface to V150% and V200% isodose lines were calculated, and are outlined in Table 3. Acute and chronic radiation toxicity data were available for 91 patients (Table 4). The most common acute toxicity was vaginal mucositis, which was reported in 67.8% of patients, and only 8.9% of patients experienced ≥ grade 2 mucositis. The most common G1 chronic toxicities were vaginal stenosis (18.7%) and vaginal dryness (7.8%), and the most common G2 toxicity was vaginal stenosis (5.5%). There were no grade 4 toxicities; only two patients developed G3 chronic toxicities: one patient displayed complete obliteration of the vagina and the other reported severe dyspareunia, both of them receiving EBRT + VBT. Actuarial 3-year G2 and G3 vaginal toxicity were 7.7% and 2.2%, respectively.
Table 3
Dosimetric parameters
Table 4
Acute and chronic toxicities
After excluding 13 patients with partially missing dosimetry data, Pearson correlation was used to test whether there was statistically linear relationship between dosimetric parameters and toxicity (CTCAE). Despite the lack of ≥ G1 toxicity, we found hints of correlation between vaginal stenosis and bladder Dmax and D0.1cc, between vaginal fistula and rectum D2cc and D1cc, and between vaginal mucositis and total prescribed dose. All correlations were found to present above 15% correlation index. Additionally, a stepwise correlation analysis was performed with below 5% significance level.
Discussion
Vaginal vault brachytherapy is used in high-intermediate or high-risk endometrial cancer alone or in combination with EBRT. Iridium-192 sources have been widely used for a long time, although 60Co source application has increased in the last few years. Several researchers have published their results and experience using 60Co HDR brachytherapy, mostly in cervical cancer, with dosimetric parameters, local control, and toxicities comparable to those reported using 192Ir [13, 14]. Differences can be found between cobalt and iridium in maximum doses in the upper vaginal mucosa due to significant differences in the anisotropy factor in the longitudinal axis of the source [15, 16]. After literature review, we were not able to find a publication about clinical outcomes in endometrial cancer treated with 60Co HDR brachytherapy source, although toxicity has been reported in some series [15, 16].
Vagina is the most common site of recurrence after surgery in endometrial cancer. Both VBT and EBRT ± VBT are good options to decrease the risk of local recurrence from 18-26% to 5-6% [3, 17, 18]. Vaginal failure after VBT ranges from 0 to 3.1% [17]. In the present study, VBT using 60Co source showed to be effective in keeping low-risk (2.2%) of vaginal recurrence, with rates of local control similar to those reported in literature using 192Ir HDR source or low-dose rate (LDR) brachytherapy.
Dosimetric parameters to OARs were similar to those reported in other papers. Mean rectum D2cc in our study was 88.1%, within the ranges of 88-88.3% described in other publications [15, 16]. Mean bladder D2cc was 79%, in line with 84.2% reported by Tormo et al., but higher comparing to 55% reported by Mobit et al. This is probably due to differences in simulation protocol (full vs. empty bladder) and contouring (full bladder vs. catheter balloon) [15, 16].
The main risk of toxicity with VBT is limited to proximal vaginal, resulting in atrophy, stenosis, and/or reduced length. Since the assessment of vaginal toxicity is difficult, we decided to report acute mucositis and evaluate mucosal side effects in terms of vaginal stenosis, dryness, and dyspareunia, although quality of life on sexual activity was not analyzed. Grade ≥ 3 toxicity reported in literature is very low (range, 0-5.2%) [19], and our results are in line with these data, with reported ≥ grade 3 toxicity of 2.2%. Although low acute vaginal side effects were seen with 60Co, reported ≥ grade 1 vaginal mucositis was 65.6% (65.1% in patients treated with exclusive VBT) compared to 36.6% reported in other studies [3]. This may be partially due because in our series, we reported acute toxicity at last day of brachytherapy, while in PORTEC-2 trial, patients were assessed 2-4 weeks after end of treatment [3]. HDR brachytherapy with 60Co is well-tolerated and toxicity is low, with only two patients developing grade 3 complications.
We were able to find hints of correlation between vaginal stenosis, fistula, and mucositis with some dosimetric parameters, although the correlation analysis lacks statistical power due low number of ≥ grade 1 toxicity cases in the sample. Further data may be required if we want to prove any actual correlation.
The present study has some limitations. First, patient data were collected retrospectively, and this limits the strength of our clinical outcomes. In addition, toxicity was not systematically evaluated, which may affect our toxicity results. Finally, we were not able to report baseline characteristics or quality of life questionnaires, because these data were not regularly recorded.