Purpose
Low-dose-rate (LDR) brachytherapy for oral cancer present a high local tumor control rate while preserving oral function and maintaining a high quality of life [1-4]. One of the radiation sources used in LDR brachytherapy is gold-198 (198Au) grain, a tiny radio-active source measuring 2.5 mm in length and 0.8 mm in diameter, with minimal invasive implantation. 198Au grain has a physical half-life of 2.7 days and gamma energy of 0.412 MeV. In patients with tongue cancer, it has been reported that there were fewer adverse events after treatment with 198Au grain than with iridium-192 (192Ir) in LDR brachytherapy [5].
Most oral lesions treated with 198Au grain brachytherapy, in our hospital, are superficial and thin, and the grains are implanted just below the mucosal epithelium; hence, a grain may fall out of the tissue relatively early following implantation. According to the guidelines of Japan’s Ministry of Health, Labor, and Welfare, a patient must be admitted to a radiation-controlled room for at least three days after implantation of 198Au grains in case of grain displacement [6]. Early displacement of radio-active sources from the oral cavity may expose medical staff to radiation as well as patient’s family or others surrounding the patient, and cause radio-active contamination. In addition, effects of such early displacement on treatment outcomes may occur. Therefore, it is necessary to prevent the displacement of radio-active source after implantation. This study aimed to investigate the status of displacement of 198Au grains in patients with oral cancers as well as the factors and countermeasures for the displacement of 198Au grains.
Material and methods
A total of 34 patients with oral cancer, who were treated with 198Au grain brachytherapy alone in our hospital between 2013 and 2019 were enrolled in this study. Baseline characteristics of the 34 patients are shown in Table 1. The distribution of primary lesions included the tongue (n = 22; 4 carcinomas in situ, 10 T1, 4 T2, 1 T3, 1 rT1, and 2 rT2), floor of the mouth (n = 7; 1 carcinoma in situ, 4 T1, and 2 T2), buccal mucosa (n = 4; 1 carcinoma in situ, 1 T1, and 2 T2), and upper lip (n = 1; 1 carcinoma in situ), and were classified according to the 2017 Union for International Cancer Control TNM classification [7]. Depending on the type of tumor growth, tumors were classified into the following three types: superficial, exophytic, and infiltrative. A tumor with superficial development grew on the outermost surface of the mucosa. For a tumor with exophytic growth, most of the tumor was located on the surface of the mucosa, and grew from the surface of mucosa outward. For a tumor with infiltrative growth, most of the tumor was located beneath the surface of the mucosa, with or without ulceration. None of the patients had cervical lymph node or distant metastasis at the start of 198Au grain brachytherapy.
Table 1
Baseline characteristics
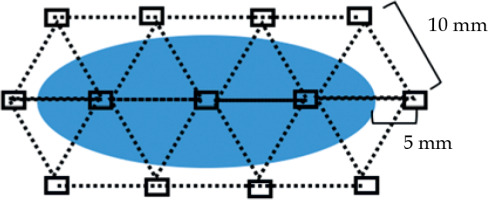
The technique for 198Au grain implantation was as follows: the longest diameter of the lesion, which was equivalent to Lugol’s staining-free area, was identified, and the grains were placed at equal intervals of 10 mm. They were placed approximately 5 mm behind the lesion’s posterior margin and approximately 5 mm ahead of the lesion’s anterior margin. Next, the grains were arranged at approximately 10 mm above and below radiation sources of the initial arrangement. Each grain was positioned at the apex of an equilateral triangle (Fig. 1). Based on this arrangement, additional grains were placed according to the lesion’s shape.
Fig. 1
Arrangement of 198Au grains. The oval region shows the mucosal area without Lugol’s staining, and squares represent 198Au grains. Each dotted line indicates an approximately 10 mm distance between grains
198Au grains were implanted in the treatment room next to patient’s radiation-controlled room. Implantation of 198Au grains in the oral cavity was confirmed using X-ray before the patient was allowed back to the radiation-controlled room. Subsequently, the treatment room was surveyed to ascertain that no displacement of 198Au grains occurred. Thereafter, radiographs were typically taken once or twice daily in the treatment room to confirm the presence of 198Au grains in the oral cavity. Each time, a radiation survey was conducted in the treatment and patient rooms to exclude the displacement of 198Au grain. In case grain displacement from the oral cavity was confirmed, we tried to locate it using a radiation survey meter in the patient’s room and treatment room. If the displaced 198Au grain was not detected in the room, postero-anterior chest and abdominal X-rays were performed to identify possible ingestion or aspiration of 198Au grain or its’ hidden presence in the clothes or skin.
After 198Au grain implantation, all patients always wore a removable spacer made of dental silicon impression materials, including a lead plate with a 4 mm thickness. It was only removed at mealtimes and tooth brushing. A filter was placed in the sink outlet to prevent 198Au grains from being flushed down the drain during when teeth brushing. When a patient had difficulties swallowing saliva while wearing the spacer, the saliva was removed by spitting it into a tissue or using a suction tube. The saliva and tissues were stored in the patient’s room until the patient left the hospital.
The patient could leave the isolation area according to the criteria prescribed by Japan’s Ministry of Health, Labor, and Welfare. According to the criteria, residual radio-activity in the body should not exceed 700 MBq, equivalent dose-rate per centimeter at a point 1 m away from the patient’s body surface should not exceed 40.3 μSv/h, and the patient must stay in the hospital for at least three days in case of 198Au grain displacement [6].
Permanent dose of 198Au grain brachytherapy was calculated using an X-ray image taken 24 hours after 198Au grain implantation. First, the treatment area was calculated based on X-ray image using the following formula:
According to the Manchester system (see Table 2) [8], radio-activity required to deliver 10 Gy to therapeutic area was calculated. This value was regarded as A. Next, permanent total dose was calculated as follows [9]:
Table 2
Manchester system table [8]
| Area (cm2) | 226Ra (mg hr.) | 198Au (mCi [MBq]) |
|---|---|---|
| 1 | 72 | 2.70 (100) |
| 2 | 103 | 3.86 (143) |
| 3 | 128 | 4.80 (178) |
| 4 | 150 | 5.62 (208) |
| 5 | 171 | 6.41 (237) |
| 6 | 188 | 7.05 (261) |
| 7 | 204 | 7.65 (283) |
| 8 | 219 | 8.21 (304) |
| 9 | 235 | 8.81(326) |
| 10 | 250 | 9.38 (347) |
with activity/grain representing the MBq value at 198Au grain implantation, 37 representing 1 mCi = 37 MBq, and 10 representing 10 Gy.
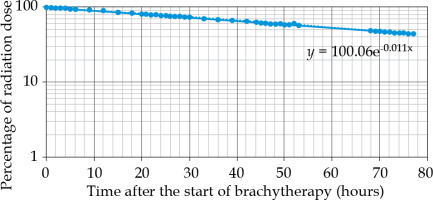
Reduction rate in the radiation dose of a 198Au grain was estimated (see Fig. 2). Location of the displaced grain was classified as either inside or outside of gross tumor volume (GTV). We defined GTV as the region that was unstained with Lugol’s solution.
Number and location of the displacement of 198Au grains, primary lesion, growth type, and treatment results were evaluated. Moreover, exposure dose for medical personnel was determined using a semi-conductor pocket dosimeter attached to the chest of the medical personnel during the patient’s hospitalization.
The study was approved by the (anonymized for review) University Ethics Committee (registration number anonymized for review), and was conducted in accordance with the Declaration of Helsinki. According to the University Ethics Committee guidelines, we obtained opt-out informed consent.
Results
Of the 34 patients included in the study, 4 (11.8%) had at least one displacement of 198Au grain during hospitalization. In terms of the number of radiation sources, 367 grains were implanted in 34 patients, and 5 grains fell out of the oral cavity, resulting in a displacement rate of 1.4%.
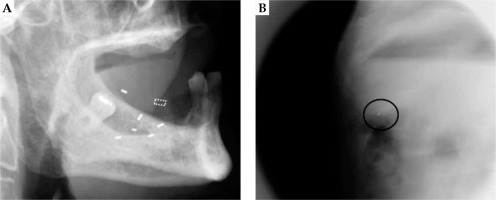
The characteristics of the patients and displaced grains are shown in Tables 3 and 4. The displaced grains were located on the tongue (n = 4) and upper lip (n = 1; Fig. 3). Four of the five displaced grains were located outside GTV and one inside GTV (case number 2). There was no displacement of the grains implanted in the floor of the mouth or the buccal mucosa. Three of the four patients underwent re-implantation with a 198Au grain at the same location under local anesthesia. In these three cases, 198Au grains were found on the floor in the patient’s room and were detected using radiation survey meters. Figure 4 shows a representative case of 198Au grain re-implantation (case number 4). In this patient, seven 198Au grains were implanted for carcinoma in situ of the upper lip. One of the seven grains was displaced approximately 3 days after implantation (Fig. 4D). The patient’s room was surveyed, and the displaced grain was found on the floor. After confirming the location of the displaced grain by radiography, it was disinfected and re-implanted in the same location (Fig. 4F).
Fig. 3
Location of displaced 198Au grains. A-D) The cases 1-4, respectively. Squares show positions of 198Au grains, and the circle indicates the missing 198Au grain. Solid and dotted circles represent the positions of the first and second displaced 198Au grains (B)
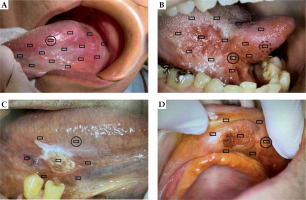
Fig. 4
Case number 4. A-C) Day 0, 1, and 2, respectively, after 198Au grain implantation. Day 0 represents the implantation day. D) Day 3 after 198Au grain implantation. The dotted square shows the estimated location of the displaced grain. E) Using a radiation survey meter, the displaced grain (arrow) was found on the floor of the patient’s room. F) After re-implantation. The displaced grain was implanted in the same location (arrow)
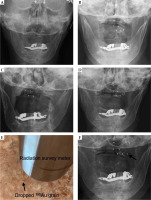
Table 3
Patient characteristics
Table 4
Characteristics of displaced radio-active sources and exposure dose for medical personnel
One patient (case number 3) swallowed a grain, and it was detected in the abdomen (probably the stomach) on radiography (Fig. 5). In this case, the grain was not removed for re-implantation. Follow-up observation was conducted during hospitalization while monitoring the position of the swallowed grain on radiographs. We expected the 198Au grain to be passed out with fecal matter; hence, the patient’s feces were stored in a container and surveyed or radiographed to confirm the discharge of the grain. However, during the hospitalization period, the 198Au grain was not passed in the stool. At the next hospital visit, an abdominal X-ray did not detect the 198Au grain. Therefore, we assumed that it was spontaneously excreted in the stool following hospital discharge. The patient had no gastro-intestinal symptoms. In case number 3, the estimated time from the implantation of 198Au grains to the displacement ranged from 0 to 23 hours. The estimated maximum dose reduction rate was 100% per grain displacement.
Fig. 5
Case number 3. A) The dotted square indicates the estimated location of the displaced grain. B) The 198Au grain (circle) was detected in the stomach
Of the four patients with 198Au grain displacement, two were recurrence-free as well as without cervical lymph node and distant metastases during their respective follow-up periods (case numbers 1 and 4). One patient (case number 2) with tongue cancer had local recurrence and cervical lymph node metastasis at 8 and 38 months following 198Au grain brachytherapy, respectively. A recurrent tongue lesion developed in the area adjacent to the posterior margin of the primary lesion. The local recurrent tongue lesion and cervical lymph node metastasis were treated surgically and controlled during follow-up. Another patient with tongue cancer (case number 3) died 20 months after 198Au grain brachytherapy due to multiple distant metastases. In contrast, 2 of the 30 patients without 198Au grain displacement experienced local recurrence (6.7%).
Discussion
In the present study, we reviewed the current status of displacement of 198Au grains in patients with oral cancers, and evaluated the factors and countermeasures for it. Four of the 34 patients (11.8%) had at least one displaced 198Au grain during hospitalization. In terms of the number of radiation sources, 367 grains were implanted in 34 patients, and 5 grains fell out of the oral cavity, resulting in a displacement rate of 1.4%.
There are no reports on 198Au grain displacement in oral cancer; however, seed migration and displacement in the treatment of prostate cancer have been mentioned. In prostate brachytherapy using 125I seeds, displacement of seeds occurs in 6.0-30.4% of patients and 0.13-0.84% of total implanted seeds [10-15]. In the current study, the displacement of seeds occurred in 11.8% of the patients and 1.4% of the total implanted seeds. Displacement rates of radio-active sources in previous studies were similar to those observed in this study. As brachytherapy for prostate cancer uses many radio-active sources, a dose reduction due to displacement of a few seeds may have insignificant influence on clinical outcome [14]. In contrast, as radio-active sources in 198Au grain brachytherapy in oral cancers are arranged in proportion to the size and shape of the lesion [9, 16, 17], the treatment effect may vary when one grain deviates from the scheduled position or is displaced. This may lead to recurrence depending on the location or timing of the displaced grain. However, if the possible 198Au grain displacement was to be compensated by increasing the number of grains, the implantation of a larger number of grains may unnecessarily increase radiation exposure to surrounding normal tissues, and increase severity of complications. Therefore, implantation techniques should be re-considered to reduce displacement of 198Au grains.
An early 198Au grain displacement after implantation may be due to backflow in implantation route. To prevent this, the applicator insertion point should be approximately more than 10 mm away from the placement positions of the grains. The oral mucosal epithelium is thin, non-keratinized stratified squamous, and, unlike other gastro-intestinal mucosae, characterized by the absence of a muscularis mucosae [18]. A 198Au grain arranged at the periphery of a lesion is located just below the mucosal epithelium (the lamina propria and submucosa, which are relatively sparse tissues) [19]. Moreover, the tongue and lips are highly mobile, and the mucosal epithelium is easily injured by contact with the teeth or spacer, and is fragile due to influence of 198Au grain implantation. Therefore, it can be considered that a 198Au grain located just below the mucosal epithelium may destroy the mucosal epithelium when the tongue or lip moves, and might fall-off from the mucosal surface. All cases of grain displacement occurred in either patients with tongue or lip cancers in the present study, and 4 out of 5 dropped-out grains were placed in the periphery of a lesion. To prevent this, the number of insertion points should be as small as possible, so that injury to the mucosal epithelium is reduced. Also, re-insertion should be performed from the same point. The position of an implanted 198Au grain should be established to be deeper until it reaches the muscle layer. However, attention should be paid to the notion that the 198Au grain is not located in such a deep part as to lower the mucosal surface dose. As differences in displacement rates have been reported with years of experience, therapeutic experience is also a factor in preventing source dropouts [12], and sufficient training using a phantom is required before 198Au grain implantation in a patient.
Four of the five displaced 198Au grains were found on the floor of the patient’s room. 198Au grain that had fallen out of the lesion into the oral cavity might have been discharged into the tissue paper along with saliva, and then dropped onto the floor. It is also considered that the patient with lip cancer spontaneously discharged the 198Au grain from the upper lip, and it fell directly onto the floor. As a countermeasure, and for early detection of displaced 198Au grains, we consider instructing the patient to carefully examine the tissue paper when spitting saliva and to check whether 198Au grain was included in the saliva.
The exposure dose to the medical personnel at the time of implantation was similar to that reported in previous studies, except for case 1 [19, 20]. The potential reason for higher exposure dose in case 1 was the higher number of radiation sources and longer time of 198Au grain implantation, as X-rays were taken to confirm the number and position of 198Au grain implants. Since the exposure doses to medical personnel are expected to increase due to frequent monitoring of 198Au grains during hospitalization, healthcare providers should plan essential procedures early to reduce the exposure dose. Time required for checking the 198Au grain implants should be shortened, and the working distance between the healthcare provider and the patient should be increased. In addition, exposure doses for medical personnel can be reduced by allowing workers to work in shifts; hence, the number of staff should be increased. Among the four cases with 198Au grain displacement, three displaced grains were found on the floor of the patient’s room, and if the displacement had not been noticed, medical personnel and cleaners could have been unnecessarily exposed to radiation.
In the groups with and without displacement of 198Au grains, 1 of 4 patients (25%) and 2 of 30 patients (6.7%), respectively, had local recurrence of the tumor during the follow-up period. Only four patients had 198Au grain displacement in this study, but one of them developed a recurrence. In this case, the displacement of the 198Au grain occurred twice, and one of the grains was located inside GTV; therefore, the dose reduction due to the 198Au grain displacement might have led to local recurrence. The other three cases, in which the location of the displaced 198Au grain was outside GTV had no local recurrence. It is possible that the position of the displaced radio-active source affected local control. In case 2, two 198Au grains were displaced during hospitalization and the rates of estimated dose reductions were 28% and 17% in the first and second times, respectively. Local recurrence occurred after 38 months. The estimated total dose after the loss of two grains was 65 Gy, the lowest among the four cases, which may be a reason for a recurrence. However, in the case of T1 or T2 oral cancers with LDR brachytherapy alone, the radiation dose required is 60-70 Gy [21], and it was considered that the dose was not always insufficient in case 2. In addition, this patient experienced a recurrence after surgery. Recurrence after definitive treatment of head and neck cancers has been associated with a poor prognosis [22-24]. Overall salvage cure rate was reported as 21% for recurrent squamous cell carcinoma of the oral cavity [22], and two-year disease-free survival rate after definitive resection of recurrent oral cancer was 47% [23]. The more advanced the cancer stage at the time of recurrence, the poorer the prognosis: 73% in stage I, 67% in stage II, 33% in stage III, and 22% in stage IV [23]. Moreover, two- and five-year local control rates after re-irradiation with brachytherapy for recurrent oral cancers ranged from 53% to 72% [25, 26] and from 52% to 69% [26, 27], respectively. In this study, case number 2 had a high probability of recurrence due to insufficient dosing. In this patient, the total radiation dose without the displacement of 198Au grain was 76 Gy, which was reduced to 65 Gy after grain displacement, suggesting that higher doses were needed for local control.
This study has several limitations. First, no statistical analysis was performed because of the low number of cases with 198Au grain displacement. However, we recognize that 198Au grain displacement is a significant concern because it affects not only the treatment outcome, but also exposure to medical personnel. Future studies enrolling more patients with 198Au grain displacement should evaluate the consequences on treatment outcomes and determine methods to prevent displacement. This requires regular X-ray and survey meter checks to determine whether 198Au grain has fallen off after implantation, and detailed records of medical personnel’s exposure dose. Second, radiation doses were calculated using plain radiographs; thus, only the dose delivered to the entire lesion could be calculated. We were not able not calculate the specific dose reduction for the lesion part with the displaced 198Au grain. In the future, to examine treatment effect of missing radiation source, the dose of displaced 198Au grain should be calculated in addition to the irradiation dose of the whole lesion. In prostate brachytherapy, the number of displaced seeds was reported to be relatively smaller when inserted by a radiation oncologist with long experience [12]. Although the experience of radiation oncologist might influence the displacement rate of 198Au grains, we did not investigate this aspect in our study. Since the 198Au grains for a single-patient were implanted by several practitioners, identifying the one who implanted the displaced grain was impossible. In the future, association of experience of practitioners involved in brachytherapy and number of seeds displaced should be examined.
Conclusions
The displacement of 198Au grains occurred in 4 out of 34 enrolled patients (11.8%), and only 5 out of the 367 implanted grains (1.4%) were displaced. Of the four patients with 198Au grain displacement, one had two displaced grains and experienced local recurrence. When performing brachytherapy with 198Au grains for superficial lesions on the tongue or lips with significant mobility, care should be taken to avoid grain displacement. A displacement of 198Au grains causes radio-active contamination of the patient’s room, which unnecessarily exposes medical personnel to radiation. Furthermore, displacement of 198Au grains may result in radiation underdosing and consequent tumor recurrence. Therefore, frequent monitoring of the radio-active implant is necessary for early detection of implant displacement.