Purpose
Secondary lung tumors (or lung metastases) are common lung malignant lesions, with a frequency of 30-55% of all cancer patients [1,2]. Primary cancers, which have the potential to metastasize to the lungs include bladder cancer, colon cancer, breast cancer, prostate cancer, sarcoma, Wilms tumor, and neuroblastoma [1,2].
There are currently no uniform guidelines in clinical practice for addressing small pulmonary metastases. Surgery of lung metastases is indicated only in highly selected cases, in whom a curative intent is possible without damaging lung function [2,3,4,5]. Unfortunately, lung metastases are rarely solitary, and multiple metastatic tumors in the lungs often occur at the same time. In these cases, systemic treatments or radiation therapy are more often indicated [6,7,8,9], but the use of stereotactic body radiotherapy (SBRT) is also limited due to radiation lung damage [10]. On the other hand, iodine-125 (125I) seed implantation (ISI) treatment can be administered repeatedly, since the local control rate is good and the treatment can prolong the survival of patients with lung metastasis [11,12]. Especially in the case where lung metastasis is very small, and detection and treatment occur in a timely manner, it is easy to achieve a complete response (CR) [13]. Because early-detected metastases are small, the amount of seeds used is small, and radiation-induced lung injury is minimal [13].
Direct needle puncture can be challenging due to the relatively small size of the lesions and their non-stationary positioning due to lung movement during respiration. Computed tomography (CT)-guided transthoracic needle biopsy is recommended for tissue biopsy of pulmonary lesions < 2 cm [14]. A meta-analysis found that the rate of major complications is low in a CT-guided lung biopsy, but the risk increases with lesion size decreasing [15,16,17,18].
Accurate puncture can reduce trauma to lung tissue and accelerate patient recovery. In our clinical work, we found that the use of a local anesthesia syringe as a guide improves the accuracy of the puncture method. In our hospital, 2% lidocaine is used for local anesthesia, with a 5-ml injection dose before CT-ISI is performed. After local anesthesia, the 5-ml syringe is not pulled out immediately but left at the site of local anesthesia. The small lesion in the lung is then co-located with this body surface marker. This is not only helpful to select the best puncture point, but also can provide a guide for the direction of the puncture needle. When larger syringes (such as 10 and 15 ml) are retained at the site of local anesthesia, the direction of the needle tip will be deviated due to gravity, which cannot provide an accurate guide for the implantation direction of the puncture needle.
The aim of the present study was to present our experience with the technique of using a local anesthesia 5-ml syringe as a guide for CT-ISI puncture. This could provide a simple method to improve the outcomes of CT-ISI of small lung lesions and avoid complications.
Material and methods
Study design and patients
This was a retrospective study of patients with small metastatic tumors in lungs treated with CT-ISI between December 2013 and March 2018 at the Department of Interventional Radiology of the Affiliated Hospital of the University. This retrospective study was approved by the ethics committee of the Affiliated Hospital of the University. The use of patient information was approved by our institutional review board, which waived a requirement for patient informed consent.
The inclusion criteria were: 1. Highly suspected diagnosis of metastatic tumors in the lung based on patients’ history of cancer and on clinical, imaging, and/or pathological characteristics of the lesions; 2. CT-ISI performed; 3. The largest diameter of lesions < 2.5 cm; and 4. Complete clinical data.
The exclusion criteria were: 1. Emphysema or a large lung bubble near the metastasis; 2. Unstable cardiorespiratory function, blood coagulation disorder, or active infection; or 3. Eastern Cooperative Oncology Group performance status (ECOG PS) ≥ 3.
Grouping
The patients were divided into two groups: a group where a 5-ml syringe was used as a guide during CT-ISI and a group, where this technique was not applied.
CT-ISI
The radioactivity of 125I seeds (CIAE-6711; Chinese Atomic Science Institution, Beijing, China) used in this study was 2.59 × 107 Bq. The ISI treatment plan system (TPS; KL-SIRPS-3D) and gun-type implantation device were from the Institute of Medical Science and Technology (Beijing, China). Disposable 5-ml syringes and 18-G puncture needles (Cook Medical, Bloomington, IN, USA) were applied for local infiltration anesthesia and puncture. Lung lesions were scanned by CT at 3-mm intervals, using a 64-slice CT scanner (Siemens, Germany) for intraoperative and post-operative follow-up.
One radiation physicist and two interventional radiologists generated treatment plan (TP) for each pulmonary metastasis using TPS, in order to determine the number of seeds to be implanted and the ideal implantation location. The planning target volume was defined as 0.5 cm of expansion external to the gross tumor volume. The prescribed dose was 135 Gy, which was chosen based on our previous studies [12,13]. Local anesthesia was administered with a 5-ml syringe (2% lidocaine; Yimin, Yichang, China) at the body surface location marker (Figures 1A,B, 2A, and 3A). An 18-G needle was then punctured into the pulmonary metastasis with or without the 5-ml syringe as a guide (Figures 1C-F, 2B,C, and 3B-D) and the 125I seeds were implanted (Figures 2D and 3E). As in our previous study, the seeds were placed 0.5-0.8 cm apart in line with the TPS to the best possible extent. According to the manufacturer’s instructions, the seed radiotherapy treatment radius of 125I is about 1 cm. Thus, for lesions ≤ 1 cm, the entire lesion can be fully included in the treatment range, even if the lesion is only punctured at the edge. For larger lesions, if the puncture location cannot fully cover the lesion, the puncture is considered failure and must be performed again, after adjusting the position or path of the puncture needle. Post-implantation dose verification was performed to verify the therapeutic dose according to the TPS and assessment of peripheral tissue (bronchial and vascular) damage. A D90 (dose contains 90% of the target volume) value of > 135 Gy at the last implantation was regarded as successful.
Fig. 1
Treatment of pancreatic cancer lung metastasis by 125I seed implantation, using a 5-ml syringe as a guide. According to the body surface localization of the 5-ml syringe used for local anesthesia and after the computed tomography (CT) scan, the 5-ml syringe was found to be near the lung metastasis (diameter of 1 cm) in three layers (layer thickness of 3 mm). Then, according to the syringe as the selection, an 18-G needle was used to access the lesion at first try to deliver the seeds. A, B) The fine white arrow indicates the body surface marker. The broad arrow shows the lung metastasis, C, D) The fine white arrow indicates the 5-ml syringe used as a guide, E, F) The broad arrow shows the 18-G needle punctured into the lung metastasis
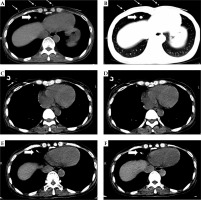
Fig. 2
A 60-year-old male underwent regular chemotherapy when pulmonary metastases were found 3 years after surgery for gastric adenocarcinoma. Then, he underwent repeated local seed implantation. A) The body surface marker. B) A 5-ml syringe was left at the site of local anesthesia. C) An 18-G needle was punctured into the pulmonary metastasis. D) The implantation was completed
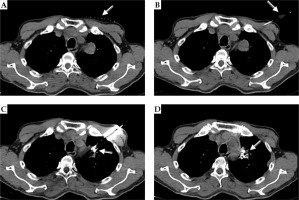
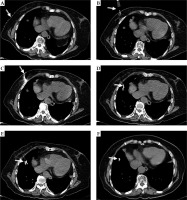
Fig. 3
A 70-year-old female underwent regular chemotherapy when pulmonary metastases were found 2 years after surgery for rectal carcinoma. She underwent local seeds implantation due to recurrent pulmonary metastases. A) The body surface marker. B) A 5-ml syringe was left as a guide after local anesthesia. C, D) An 18-G needle was punctured into the lesion. E) Seeds were implanted. F) The lesion disappeared and only a seed remained at 1 month after the implantation
Follow-up and evaluation of clinical effectiveness
Patients were monitored using continuous electrocardiogram for 6 hours after the implantation, with appropriate symptomatic treatment for chest tightness, chest pain, blood in sputum, vomiting, fever, etc. Generally, the patients were discharged within 2 days after implantation. The final follow-up was on March 31st, 2018. The follow-up time was calculated from the first-ever treatment of pulmonary metastases with CT-ISI to the last follow-up or death. Chest CT enhancement examinations were used to evaluate the maximum diameter of the metastatic pulmonary lesions (Figure 3F), to screen for new lesions, and to assess radiation-induced lung injury at 1, 3, 6, and 12 months after CT-ISI. The local control rate of pulmonary metastases was calculated independently by three radiologists, according to the modified response evaluation criteria in solid tumors (m-RECIST) [19]. The response rate (RR) was determined by the sum of the complete response (CR) and partial response (PR) rates. Radiation lung injury was evaluated according to the National Cancer Institute’s Common Terminology Criteria for Adverse Events, version 4.0 (CTCAE 4.0) [20].
Statistical analysis
Continuous variables were expressed as means ± standard deviations (SD) and were analyzed using the Student’s t-test. Categorical variables were presented as frequencies and percentages and were analyzed using the χ2 test or Fisher’s exact test, as appropriate. Two-sided p-values < 0.05 were considered statistically significant. GraphPad Prism, version 5 (GraphPad Software Inc., San Diego, CA, USA) was used for statistical analysis.
Results
Tumor characteristics
Nineteen patients were included into the study and Table 1 presents their characteristics. They had a total of 50 small (< 2.5 cm) lung lesions, with 2.6 ±2.2 (range, 1-10) metastases per patient. The lesion size was 1.6 ±0.4 cm. The mean time between diagnosis by CT scan of the primary tumor and the first discovery of pulmonary metastasis was 12.7 ±4.8 (range, 8-21) months. Also, three patients had liver metastasis and two had bone metastasis.
Table 1
Characteristics of the patients with small lung metastasis treated with CT-ISI
CT-ISI
CT-ISI was performed 37 times in those 19 patients, with 2.3 ±0.7 punctures (range, 1-5) per lesion. A total of 840 seeds were used, with 44.2 ±33.6 (range, 10-160) seeds per patient. The mean D90 for CT-ISI was 134.5 ±7.5 Gy (Table 2).
Use of the 5-ml syringe to guide metastasis access
Treatment intervention for eleven patients was performed using a 5-ml syringe as a guide during CT-ISI (median, 2; range, 1-5 times) in 28 lung metastases. Eight patients received treatment without the 5-ml syringe during CT-ISI (median, 2; range, 1-4 times) in 22 lung metastases. There were no differences between the two groups regarding the characteristics of patients (Table 1), except that the syringe group had a higher frequency of males. There were no differences in total dose and number of implanted seeds between the two groups, but the number of punctures per lesion was lower in the syringe group than in the no-syringe group (1.9 ±0.5 vs. 2.9 ±0.6, p < 0.001), suggesting a higher puncture accuracy.
Complications
All patients tolerated the procedures well. No CT-ISI- related deaths occurred, and no patient needed premature procedure termination. Only one patient treated without a 5-ml syringe experienced grade 2 chest tightness, chest pain, intraoperative needle tract bleeding, and post-operative blood in sputum. All complications were mild to moderate and required only symptomatic treatment (Table 3).
Table 3
Complications with or without the 5-ml syringe as a guide for CT-ISI treatment of small lung metastasis (based on the Common Terminology Criteria for Adverse Events, version 4.0.)
Follow-up
The median follow-up period was 13 months, ranging from 8 to 27 months. During follow-up, four patients died, one patient was lost to follow-up, and 14 survived. One patient in the no-syringe group experienced subcutaneous tumor implants, considered to result from multiple punctures. All the CT-ISI in the SMTIL were well-controlled at 6 months follow-up (RR 100%, see Table 4). In the first month of follow-up, there were 8 SMTIL that only reached PR according to m-RECIST criteria. In the 3rd and 6th months of follow-up, there were still 5 metastases meeting criteria for PR, likely related to relatively large lesion size (≥ 2 cm), insufficient 125I seed arrangement, or local dose correlation. The RR at 1, 3, and 6 months after the CT-ISI procedure was 100% in the 50 targeted tumors (Table 4).
Discussion
Small lung metastases move with respiration, making localization difficult, and often increasing the number of punctures for access. Accurate puncture can reduce trauma to lung tissue and accelerate patient’s recovery [15,16,17,18]. The objective of the present study was to present our experience with the technique of using a local anesthesia 5-ml syringe as a guide for CT-ISI. The results suggest that puncture with a 5-ml syringe as a guide during CT-ISI is probably a more accurate option for patients with small (< 2.5 cm) lung metastasis.
Stage IV cancer is incurable, but highly selected cases may undergo surgery of isolated lung metastasis [2,3,4,5,21,22]. Unfortunately, lung metastases are usually small and multiple, and generally do not have obvious clinical symptoms [1,2]. Small asymptomatic lung metastases may be simply followed, and more aggressive options can be maintained for symptom control once the lesions become symptomatic [6,7,8,9,23]. Systemic therapy and radiotherapy can be used for lesion control [6,7,8,9]. During the treatment of metastases with SBRT, the risk of radiation-induced lung injury is higher than ISI, the mean lung doses in ISI were significantly lower than those in SBRT (1.952 ±0.713 vs. 5.618 ±2.009 Gy, p < 0.0001) [10]. Percutaneous ablation, with using either radiofrequency or ICI, may reduce patient’s treatment load and provide good outcomes [12,13,24,25,26].
Based on our previous studies, pulmonary metastases of 1-2 cm in size can be diagnosed and located in the lungs, and patients are able to tolerate the treatment [12,13]. In particular, when 125I seed implantation is utilized, the general arrangement of 10-20 seeds can achieve local CR. The treatment does not require central location of the lesion to be directly targeted and based on the 1 cm effective range of 125I seeds, local accumulation of seeds covering the lung metastasis can achieve adequate radiation doses. Nevertheless, access to small lesions (< 2.5 cm) is prone to failure, but the methods for an accurate positioning of the needle access are limited. Penetrating directly through the rib into the lesion for seed implantation is contrary to the intention of minimally invasive therapy, and we do not recommend this approach; in our study, puncture-related complications rated as only grades 1 or 2 (Table 3) [27]. Computed tomography fluoroscopy-guided percutaneous 125I seed implantation has been studied, but it is advisable to apply this approach cautiously for small lesions < 2.5 cm [28]. Novel 3D-printing template technologies can be used to guide the puncture and solve the problems of continuous respiration-associated movements of the lungs [29,30,31], but it requires printing systems, a CAD software, and the ability/experience to use them.
Iodine-125 seed implantation is often carried out under local anesthesia, and it is difficult to locate small lesions in the lungs during respiration. Han et al. [32] were the firsts to use a puncture needle to localize the tumors during CT-ISI for the treatment of primary lung cancer. We further improved this technique by using a conventional surface marker combined with CT scan line positioning. When a 5-ml syringe was used for local anesthesia, the syringe was inserted in the direction of metastasis and placed on the skin, followed by a CT scan to determine the location of metastasis. While using a local anesthesia syringe as a guide, the ISI puncture needle can more easily access the target metastasis regardless of lung movement. Moreover, the syringe can assist in anchoring the small metastases, reducing the degree of small metastases movement.
In the present study, the use of the syringe led to a smaller number of punctures per lesions to achieve a successful ISI. Because the average number of punctures was lower, the procedure-related complications for the syringe was smaller than in the no-syringe group. Most of the complications were grade 1. The only patient with grade 2 complications was in the no-syringe group, but this observation is unreliable and cannot be used in the conclusion. One patient in the no-syringe group experienced subcutaneous tumor implants, which was considered to result from multiple punctures. Therefore, reducing the number of punctures and improving the accuracy of punctures can reduce the risk of tumor metastasis.
The present study has some limitations. This was a retrospective, single-center study with a limited sample size and a short follow-up period. Furthermore, there was a selection bias, since only the patients with small metastases (< 2.5 cm) were included. We focused on the accuracy and complications of the use of a 5-ml syringe as a guide during CT-ISI, and survival analysis was not performed in this study. A large sample multi-center cohort study is warranted to evaluate the long-term outcomes associated with this quite simple method to improve the accuracy of CT-ISI.
Conclusions
Puncture with a 5-ml syringe as a guide during CT-ISI can improve the accuracy of puncture and it is not associated with any additional complications in patients with small (< 2.5 cm) lung metastases.
Data availability statement
The datasets used and/or analyzed during the current study are available from the corresponding author on request.



