Introduction
Rheumatoid arthritis (RA) is characterized by chronic proliferation of synovium, cartilage destruction, and periarticular erosion/bone loss [1]. This strong interface between subchondral bone, cartilage, and synovial membrane of the inflamed joint induce a release of particular protein fragments into the serum and urine, which may be used as specific biomarkers [2]. The articular ligament predominantly comprises of two essential lattice proteins sort II collagen and proteoglycans including aggrecan. Sort II collagen is framed by the relationship of three indistinguishable α1 chains, in a triple helix aside from the two finishes, in the C- and N-telopeptides. They are integrated by synovial cells and chondrocytes because of the impact of development of components and cytokines [3-5] with the arrangement of neoepitopes, which are discharged into the dissemination and discharged in urine. In scientific investigations, a relationship between urinary sort II collagen C-telopeptide (u) CTX-II and radiographic hip joint space narrowing and hip torment was discovered [6, 7].
Cartilage oligomeric matrix protein (COMP) is a 435-kDahomopentameric non-collagenous extracellular matrix protein of the thrombospondin family with calcium binding proteins [8]. COPM is essentially situated in the hyaline ligament on a very basic level to hold on the integrity of collagen arrange; it is additionally present in ligaments, skin, and synovial fibroblasts [9, 10]. COMP has been broadly considered in RA, with a few reviews presuming that COMP can possibly be an analytic and prognostic pointer, a marker of ailment seriousness, and a marker of the impact of treatment [11-17]. Though, different analysts indicated contradicting sees for the utilization of COMP as a natural marker for RA [18-20]. This shows the need for a more noteworthy comprehension of the biochemical ramifications of COMP in RA finding, forecast, and treatment observing.
While most conditions of arthritis include joint remodeling, a unique feature of RA is the existence of bone erosion only without signs of repair (osteophytes). It has been suggested that suppression of signaling in the Wingless (Wnt) signaling pathway is the key master in the imbalance between bone resorption and replacement signals in RA joints [21]. Activation of the “canonical Wnt signaling pathway” (involving Wnt and β-catenin) upregulates osteoprotegerin (OPG), which blocks RANKL-induced osteoclastogenesis and results in the inhibition of bone resorption [22-24].
Dickkopf-1 (DKK1), an endogenous inhibitory factor in the canonical Wnt signaling, could augment the expression of the osteoclast differentiation factors and RANKL together with the decrease in the expression of OPG. In addition, a results of a study have shown a vital part of DKK1 in the advancement of synovial angiogenesis [23]. Moreover, other researches have demonstrated that upregulation of DKK1 may actuate tumor corruption figure receptor-1 tumor necrosis factor (TNF receptor-1) in developing particular mesenchymal cells [25, 26], and TNF-α inhibitor could downregulate DKK1 in ankylosing spondylitis [27].
The objective of this study was to evaluate differences in serum levels of CTX-II, COMP, and DKK1 in patients with RA compared to healthy controls, and to investigate the correlation between these markers and disease activity.
Material and methods
Sixty-three patients fulfilling the 2010 ACR-EULAR classification criteria for RA [28] were recruited from the in-patients and out-patients’ clinic of the Rheumatology, Rehabilitation, and Physical Medicine Department of Assiut University hospitals. Full history recording with thorough general and musculoskeletal examination (age, sex, disease duration, age of onset, duration of morning stiffness, presence of extra-articular manifestations, and their current treatment) were completed.
The disease activity in RA patients was assessed by the 28 joint count disease activity score (DAS28), using the number of swollen and tender joints, erythrocyte sedimentation rate (ESR), and patient’s global status and pain evaluated by the visual analogue scale (VAS), ranging from 0 to 100 mm [29].
Fifty persons age and sex matched apparently healthy individuals from the hospital personnel; undergraduates, medical, and nursing staffs were also included as a control group. The local ethics committee approved the study and all participants gave a written informed consent before being enrolled into the study.
Serum samples and laboratory measures
Five milliliters of peripheral blood were collected after an overnight fasting from each patient and control participant in two EDTA tubes. One set of the tubes were used for complete blood count (CBC), first-hour erythrocyte sedimentation rate (ESR) by Westergren’s method in mm/hour, and rheumatoid factor (RF) tests in the same day of blood collection. The other samples were allowed to clot and centrifuged at 3,000 rpm for 20 min, after which, a serum were collected, aliquoted, and kept frozen at -20°C until analysis in the biochemistry lab. Assays were carried out to determine serum levels of CTX-II, COMP, DKK1,TNF-α, and interleukin 17 (IL-17) by a sandwich enzyme-linked immunosorbent assay (ELISA).
Assays were carried out to determine serum levels of COMP, CTX-II, DKK1,TNF-α, and interleukin 17 (IL-17) utilizing monoclonal antibodies directed against separate antigens of human serum COMP, CTX-II, and DKK1. The kits for COMP and CTX-II assay were provided by Elabscience Biotechnology Co., while DKK1 was provided by Thermo-Fisher Scientific. These sandwich ELISAs were carried out according to the supplier’s protocols, and optical densities were determined using an automated reader (BioRad Co., USA).
Statistical analysis of data
The clinical and laboratory data were collected, categorized, and processed by Statistical Package for Social Sciences (SPSS), version 20. Continuous data were expressed as mean ±standard deviation (SD), while the categorical data were expressed as numbers and percentages. For statistical evaluation, unpaired Student’s t-test was used for comparison of continuous variables between the two groups. Mann-Whitney U test was used to compare non-parametric variables. Categorical data were analyzed by the χ2 test. Spearman rank correlation coefficient was used to evaluate the correlation between different parameters in each group and also with studied risk factors. Multivariate logistic regression analysis of variables was additionally used. A probability (p-value) of < 0.05 was considered statistically significant.
The threshold value for optimal sensitivity and specificity of our markers were determined by the receiver operating characteristics (ROC) curve, which was plotted by calculating sensitivity and specificity at several cut-off points. The ROC curve can be used to select the best cut-off for identifying the sensitivity and specificity of the diagnostic test. The area under the curve (AUC) of the ROC plots ranged from 1.0 (perfect separation of test values into two groups) to 0.5 (no distributional differences). An AUC > 0.7 indicated a discriminating strength of statistical significance and AUC > 0.8 indicated excellent discriminating power of the test [30].
Results
Sixty-three patients were enrolled into this study, with 57 females and 6 males. Healthy control (n = 50) consisted of 38 females and 12 males (p = 0.170), with a mean age of 45.89 ±11.39 years and 44.92 ±9.65 years, respectively (p = 0.632). All the patients were diagnosed with rheumatoid arthritis, and the mean duration of disease was 8.3 ±6.05 in years, morning stiffness was 0.36 ±0.55 in hours, and the mean of DAS28 score was 4.41 ±1.17. Fifty-seven patients were rheumatoid factor (RF) positive (90.5%). All this data are presented in Table 1.
Table 1
Demographic data for both rheumatoid arthritis (RA) patients and controls, and clinical data for RA patients
Table 2 shows the comparison between RA patients and control regarding markers. The mean levels of serum COMP in ng/ml, CTX-II in ng/ml, DKK1in pg/ml, TNF-αpg/ml, and IL-17 pg/ml were 5.71 ±7.04, 0.45 ±0.27, 5619.1 ±1731.4: 2.70 ±1.31, 0.23 ±0.16, 3276.96 ±1306.77, 6.4 ±2.25: 1.55 ±0.61, and 14.3 ±3.88: 5.86 ±1.09, respectively, with highly significant difference (p = 0.001, for five biomarkers).
Table 2
Comparison between rheumatoid arthritis (RA) patients and controls regarding markers
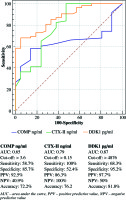
COMP showed sensitivity of 58.7 % and specificity of 85.7%, with cut-off value > 3.6 ng/ml, CTX-II demonstrated sensitivity of 100% and specificity of 52.4%, with cut-off value > 0.15 ng/ml, and also DKK1 showed sensitivity of 68.3% and specificity of 95.2%, with cut-off value 4876 pg/ml (Fig. 1).
Table 3 shows a correlation of serum levels of COMP, CTX-II, and DKK1 with the age, disease duration, morning stiffness, number of swollen joint, number of tender joint, patient global assessment, DAS28, WBCs, ESR, HB, platelet count, TNF-α, and IL-17. COMP were significantly correlated with number of swollen joint, No of tender joint, patient global assessment, DAS28, ESR, HB, TNF-α, and IL-17 (r = –0.28, p< 0.026; r = –0.33, p< 0.007; r = –0.4, p< 0.001; r = –0.52, p< 0.001; r = –0.39, p< 0.002; r = 0.25, p< 0.04; r = –485, p = 0.000; and r = –0.425, p = 0.001), respectively. CTX-II was significantly correlated only with patient age (r = 0.28, p< 0.026). DKK1 serum levels were significantly correlated with number of swollen joint, number of tender joint, patient global assessment, DAS28, ESR, HB, and platelet count (r = 0.5, p< 0.001; r = 0.67, p< 0.001; r = 0.42, p< 0.001; r = 0.8, p< 0.001; r = 0.43, p< 0.001; r = –0.49, p< 0.001; r = 0.3, p< 0.001), respectively.
Table 3
Correlation between COMP, CTX-II, and DKK1 with clinical and laboratory data of the rheumatoid arthritis patients
To investigate the correlation between the CTX-II concentration and the histopathological aging of articular cartilage, degeneration was detected as a significant correlation between CTX-II and age, with p< 0.05. Furthermore, there was a highly significant correlation between COMP and DKK1 levels and grades of disease activity (DAS28) activity, with p< 0.001 (Table 4). Linear regression for the three markers are presented in Table 5.
Table 4
Correlation between COMP, CTX-II, and DKK1 with grades of DAS28
Table 5
Linear regression for COMP, CTX-II, and DKK1
Discussion
In spite of the fact that the determination of RA transfers on an accumulation of signs and manifestations bolstered by serology radiograph, there is a difficulty in building an early finding method for RA [31]. Besides, already established clinical example, RA has a tendency to rise after some time, and deficient example is frequently introduced in the early months or even years of the disease.
The prognostic significance of CTX-II debasement markers for ligament harm has been built up in various models of degenerative joint pain, for example, osteoarthritis (OA) [32-34]. Additionally, Mazieres et al. [35] recognized that patients with difficult hip OA in whom urinary CTX-II qualities were in the upper quartile of the dispersion had a more serious danger of movement than patients in the lower two quartiles of the circulation. Moreover, patients with excruciating hip OA in whom urinary CTX-II qualities were in the upper quartile of the dispersal had a predominant danger of development than patients in the lower two quartiles of the dispersal. In our study, we found that CTX-II serum levels were altogether superior in RA patients contrasted with the solid gathering, which concurs that these ligament digestion variables might be tangled in the pathogenesis of RA. Furthermore, our study revealed that there was a strong correlation between serum levels of CTX-II and age, and thus it was related to the severity of articular cartilage degeneration and histopathological changes in the articular cartilage. This was in agreement with other studies who reported that CTX-II and RA were proposed as feasible biomarkers for early detection and monitoring of arthritic diseases, and their concentrations imitated the processes that was unswervingly implicated in the synthesis or degradation of articular cartilage and bone mineralization, which permitted a valuation over smaller periods of time compared to conservative radiography [23]. Good correlation of the levels of CTX-II to the sternness of knee cartilage blemishes on MRI in subjects with principally normal outcomes on radiographs has also been reported [13]. In humans, high concentrations of CTX-II levels in children to young adults (20 to 25 years) were defined, and steady levels were perceived in adults between 30 and 50 years [36]. However, high CTX-II levels and cartilage turnover were observed in young rats from 5 to 7 months of age [36].
The results of Mazieres et al. indicated an agreement with our review, there were a critical raises of C-receptive protein (CRP) before the start of clinical sickness [35]. In this manner, demonstrating the symptomatic estimation of serum CTX-II in the early motioning of joint aggravation is comparable to the benefit of measuring a systemic marker of irritation (CRP). Gradually, these discoveries will assign that CTX-II is an early pointer of joint aggravation as well as an impression of dynamic ligament harm. In our study, we found that serum CTX-II demonstrated affectability of 100% and specificity of 52.4%, with cut-off > 0.15 ng/ml, However, Jung et al. perceived that an uCTX-II exhibited a specificity of 87.5% and affectability of 75.5% for anticipating X-beam characterized knee OA [37].
A couple of years back, serum COMP test was propelled as the new marker for determination of RA. Our review uncovered that patients with RA had factually expanded serum COMP levels contrasted with the controls, and that the levels were contrarily associated with ESR, inflammatory cytokines (TNF-α, IL-17), the quantity of swollen joints, delicate joints, quiet worldwide score, and the DAS28 score. Furthermore, serum COMP level of 3.6 ng/ml was the ideal cut-off, an incentive for perceiving between RA patients and sound subjects, where the rate of effectively grouped patients was 58.7% (i.e. affectability) and the rate accurately delegated not having RA was 85.7% (i.e. specificity). These discoveries were in concurrence with a study of El Defrawy et al., who revealed a fundamentally higher serum COMP in patients with RA contrasted to healthy subjects; nonetheless, serum levels were emphatically related to joint degradation [38].
Moreover, the negative relationship between serum COMP levels and both ESR and RF, presented predictable theory that aggravation and tissue demolition were not specifically connected in RA [39]. Additionally, Wisłowska’s and Jabłońska’s reviewed obviously maintained part for serum COMP as a marker, reflecting that the procedures were not specifically connected to the irritation in RA [40]. Thus, it was reported that expanded COMP levels in RA could be a potential marker of articular ligament harm [11] or it may reflect ligament turnover [41]. In view of this, we can state that serum COMP levels can possibly be utilized as an organic biomarker of inflammation and ligament digestion before radiographic discoveries in RA, which is by all accounts not connected with the fiery occasions. Nevertheless, it is still not clear whether the pattern level or the transient change in serum COMP levels is a superior indicator of joint devastation. The serum COMP levels in RA patients might be influenced by changing of medications during the course of disease, which may reflect ligaments harm. Along these lines, an estimations of this marker should be perceived that require additional clinical and longitudinal investigations of different varieties in COMP serum and its relationship with joint pain manifestations.
We additionally discovered expanded serum DKK1 levels in RA patients comparing to sound controls. Prominently, these abnormal states were decidedly connected with ESR, the quantity of swollen joints, delicate joints, quiet worldwide score, and the DAS28 score.
Our assessment agrees with the past review by Diarra et al. who evaluated DKK1 level both in serum and in the synovium of RA patients [42]. They concluded that DKK1 level was hoisted inside the synovium of RA patients than osteoarthritis patients, while serum DKK1 level was higher in RA patients than in healthy subjects. Furthermore, DKK1 level emphatically corresponded with infection movement [42] and the blockage of DKK1 terminated bone disintegration in an incendiary mouse model. In addition, Daoussis et al. revealed that serum levels of DKK1 were altogether expanded in patients with RA, which related with increased danger of bone disintegrations and osteoporosis [43]. Besides, a noteworthy reduction in the serum levels of DKK1 was seen with TNF-adversary treatment in those patients [42]. Moreover, in our study, we found that DKK1 level is a critical marker of disease progress, as it is altogether directly raised in high action RA patients as compared with patients with low action or those excluded. Thus, DKK1 may fill in as another clinical biomarker for RA patients.
Conclusions
Measurement of some serological biomarkers such as COMP, CTX-II, and DKK1 that reflect bone and ligament demolition in RA patients could be utilized to demonstrate early joint affection and disease progression, however, with low affectability and specificity. The COMP serum is a potential biomarker in RA, which indicates disease activity and damage to the articular cartilage as well as to monitor treatment efficacy.