Introduction
Primary hypertension (PH) is a dominant form of arterial hypertension in teenagers, and its incidence in children and adolescents is rising as a consequence of increasing prevalence of obesity and excessive salt intake [1, 2]. PH in considered to be not only a disease of the cardiovascular system but also a systemic disorder involving inappropriate body composition and fatty tissue distribution, sympathetic overdrive, immune system activation, and premature (early) vascular aging with endothelial injury [3]. Endothelium forms an internal lining of all blood vessels and is an extremely important regulator of blood pressure and tissue perfusion. Arterial hypertension causes shear stress to endothelial cells leading to their activation and dysfunction [4].
Adhesion molecules include two families: the selectin family (E-selectin, P-selectin, and L-selectin) and the immunoglobulin family (intercellular and vascular adhesion molecules – ICAM-1 and VCAM-1) [5]. They are localised on the membranes of activated cells: leukocytes, platelets, and endothelium. Soluble forms of these adhesion molecules originate either from enzymatic cleavage or alternative splicing of the messenger RNA. E-selectin and ICAM-1 are adhesion molecules, produced almost exclusively by activated endothelial cells. E-selectin mediates leukocyte rolling on the endothelium and platelet–leukocyte interaction, whereas ICAM-1 is a molecule responsible for leukocyte migration into subendothelial layers of the vascular wall [4, 5]. Soluble ICAM-1 concentration was found to be linked to future incidence of coronary heart disease [6], and E-selectin concentration was strongly related to traditional cardiovascular risk factors [7].
Subclinical inflammation and endothelial activation contribute to the development and progression of atherosclerosis [3]. Over the last decades, a vast number of studies have implicated endothelial dysfunction in the progression of atherosclerosis and target organ damage in primary hypertension [3, 6, 7]. Several biomarkers have been established to reflect the status of subclinical inflammation in cardiovascular diseases, including high-sensitivity C-reactive protein (CRP), interleukin (IL)-6, and complete blood count-derived indices [8].
There are only a few studies on markers of endothelial injury and subclinical inflammation in paediatric patients with PH [9, 10]. In our previous study we showed a significant relation between blood pressure, urinary albumin loss, and subclinical inflammation in adolescents with PH [8].
Little is known about factors predisposing to endothelial damage in paediatric patients with PH. Also, there is no data on dependence between grade of subclinical inflammation and endothelial damage in these subjects. Unmasking these factors and dependences could result in interventional studies aimed at improvement of endothelial function and in implementation of therapeutic measures focusing at endothelial health. Thus, the aim of this study was to establish the relation between endothelial damage measured as E-selectin and ICAM-1 concentrations, subclinical inflammation evaluated from complete blood count, and clinical and biochemical parameters in children and adolescents with PH, including ambulatory blood pressure monitoring (ABPM).
Material and methods
In this cross-sectional study we analysed the clinical data of 77 paediatric patients aged from 6.58 to 17.92 years, with PH treated at one tertiary paediatric nephrology centre. The enrolment into the study took place between April 2016 and April 2018. The inclusion criterion was: arterial hypertension diagnosed according to Polish up-to-date guidelines [1, 2]. The exclusion criteria were: secondary forms of hypertension, heart defects, vascular pathology, and acute infections (temporary exclusion).
Venous peripheral blood was collected after overnight fasting, centrifuged to obtain serum, and stored at –80°C until analysis. Concentration of markers of endothelium injury was measured in serum samples using enzyme-linked immunosorbent assays (ELISA) (sICAM-1/CD54 ELISA Kit catalogue number: 950.540.096, sE-selectin/CD62E/ELAM-1 ELISA Kit catalogue number: 850.530.096, both Diaclone SAS, Besancon Cedex, France; ng/ml). Complete blood count was performed using a Coulter LH 780 haematological analyser (Beckman Coulter Inc., Brea, CA, USA), and the following markers of subclinical inflammation were analysed: numbers of neutrophils (NEU; 1000/µl), lymphocytes (LYM; 1000/µl), platelets (PLT; 1000/µl), mean platelet volume (MPV; fl), and neutrophil-to-lymphocyte and platelet-to-lymphocyte ratios (NLR and PLR, respectively).
In addition, the following clinical parameters were assessed: age (years), sex, duration of PH (months), height (cm), weight (kg), and body mass index (BMI; kg/m<sup>2</sup>). Anthropometric variables were expressed as Z-scores [11]. Overweight and obesity were defined in accordance with World Health Organisation definitions as BMI Z-score values > 1 and > 2, respectively.
Peripheral office blood pressure was measured using an oscillometric device (Welch Allyn VSM Patient Monitor 300, Welch Allyn Inc., Skaneateles Falls, NY, USA) and expressed as mm Hg and Z-score [12]. 24-hour ABPM was performed using a SUNTECH OSCAR 2 device (SunTech Medical, Inc., Morrisville, NC, USA) and interpreted according to American Heart Association (AHA) guidelines [13]. The methodology and data interpretation were described in detail previously [8]. In the final analysis we included the following ABPM-derived parameters: systolic, diastolic, and mean arterial pressure (SBP, DBP, MAP) during 24 hours (mm Hg), MAP 24 h Z-score [13], pulse pressure (mm Hg), heart rate (beats per minute), SBP and DBPL load during 24 hours (%), nocturnal blood pressure dip (%), blood pressure variability (standard deviation of all measurements) (SBPV, DBPV), and ambulatory arterial stiffness index (AASI) [14]. Disturbed circadian blood pressure rhythm was defined as nocturnal SBP or DBP dip less than 10% [13].
The following biochemical parameters were evaluated: serum creatinine [mg/dl], uric acid [mg/dl], total low-density lipoprotein (LDL), and high-density lipoprotein (HDL) cholesterol (mg/dl), triglycerides (mg/dl), calcium (mg/dl), phosphorus (mg/dl), 25 hydroxyvitamin D (25[OH)]D; ng/ml), alkaline phosphatase activity (ALP; IU/l), and daily urinary albumin loss (albumin excretion rate – AER; mg/24 h). Biochemical parameters were assessed by dry chemistry (Ortho Clinical Diagnostics, Raritan, NJ, USA), and vitamin D by chemiluminescence (ARCHITECT i1000SR, Abbott Laboratories, Abbott Park, IL, USA). Glomerular filtration rate (GFR) was calculated according to the Schwartz formula [15]. Hyperuricemia was defined as uric acid > 6.0 mg/dl, hypercholesterolemia as cholesterol ≥ 200 mg/dl, and hypertriglyceridemia as triglyceride ≥ 100 mg/dl (children aged 0-9 years) or ≥ 130 mg/dl (children aged 10-19 years) [16]. Abnormal AER was defined as albuminuria > 30 mg/24 h [1, 2].
The research project was approved by the local Ethics Committee (approval No. KB/58/2016). All procedures were performed in accordance with the Declaration of Helsinki on the treatment of human subjects. Informed consent was obtained from all participants (≥ 16 years) and their representatives.
Statistical elaboration was performed using Dell Statistica 13.0 PL software (Dell Inc., AlisoViejo, CA, USA). Variables were presented as the mean ±standard deviation (SD) and interquartile range (IQR). Normality of data distribution was tested using Shapiro-Wilk test. The following tests were used (depending on data distribution): Student’s t-test, U Mann-Whitney test, ANOVA, Kruskal-Wallis test, Pearson linear correlation, Spearman rank correlation, and Fisher’s exact test. Multivariate analysis was performed using a general regression model. Parameters that correlated with markers of endothelial injury with p < 0.300 in univariate analysis were included in the final model. A p-value < 0.05 was considered statistically significant.
Results
Clinical parameters in the studied children are presented in Table 1. Overweight was revealed in 31 (40.3%) and obesity in 11 (14.3%) patients. In the study group there were 27 (35.1%) patients with newly diagnosed and untreated PH (patients referred for the first time to the Department) and 50 patients already treated for hypertension. Among the 50 patients treated for PH 33 received antihypertensive medications and 17 patients were treated only with non-pharmacological measures. In the subgroup of subjects on antihypertensive drugs, 12 were treated with more than one antihypertensive drug (10 patients with two medications, and two patients with three medications). The results of ambulatory blood pressure monitoring are presented in Table 2. Fifteen among 27 (55.6%) patients with newly diagnosed hypertension had isolated systolic hypertension. Ten among 33 (30.3%) patients receiving pharmacological treatment and nine among 17 (52.9%) on non-pharmacological treatment had poor control of hypertension in ABPM. Disturbed circadian blood pressure profile was revealed in 30 (39.0%) patients. Markers of endothelial damage, markers of subclinical inflammation, and remaining biochemical results are depicted in Table 3. Hyperuricemia was found in 29 (37.7%), hypercholesterolemia in nine (11.7%), and hypertriglyceridemia in 16 (20.8%) patients. Increased albumin urinary excretion was revealed in 10 (13.0%) children with PH.
Table 1
Clinical data of children with primary hypertension
Table 2
Results of ambulatory blood pressure monitoring in children with primary hypertension
Table 3
Markers of endothelial injury, markers of subclinical inflammation, and biochemical data in studied children
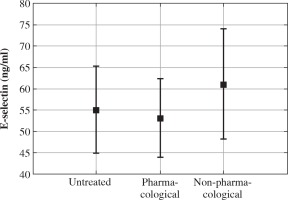
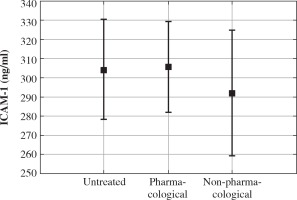
Concentrations of markers of endothelial injury in different groups of patients are presented in Figures 1 and 2. The subgroups of untreated patients, patients treated with antihypertensive medications, and patients treated only with non-pharmacological measures did not differ significantly in terms of E-selectin and ICAM-1 (55.13 ±23.29 vs. 53.17 ±25.44 vs. 61.18 ±33.33 [ng/ml], p = 0.600, and 304.29 ±53.13 vs. 305.67 ±77.92 vs. 292.02 ±67.39 [ng/ml], p = 0.782, respectively). Also, no relation between treatment with the two most commonly used antihypertensive drugs (calcium channel blockers and angiotensin converting enzyme inhibitors) and the concentration of markers of endothelial damage was found. E-selectin was significantly higher in 50 boys than in 27 girls (60.02 ±26.56 vs. 47.49 ±24.80 [ng/ml], p = 0.047), but there was no significant difference in ICAM-1 between boys and girls (312.63 ±72.44 vs. 282.80 ±51.86 [ng/ml], p = 0.062).
The relation between markers of endothelial injury and selected clinical, biochemical, and inflammatory parameters are depicted in Table 4. In the whole group of 77 patients E-selectin and ICAM-1 concentrations correlated with BMI Z-score. ICAM-1 correlated also with uric acid, HDL-cholesterol, ALP, PLR, and with SBPV and MAPV; additionally, in boys ICAM-1 correlated with MPV (r = 0.29, p = 0.038). The results of multivariate analysis are shown in Table 5. In multivariate analysis using a general regression model the only significant predictors of markers of endothelial injury were mean arterial pressure during 24 hours in ABPM expressed as Z-score for E-selectin (β = 0.329, 95% CI: 0.012-0.646, p = 0.042) and uric acid for ICAM-1 (β = 0.430, 95% CI: 0.040-0.819, p = 0.031).
Table 4
The correlation between markers of endothelial injury and selected clinical, biochemical, and inflammatory indicators in children with primary hypertension
[i] ICAM – intercellular adhesion molecule, BMI – body mass index, SBP – systolic blood pressure, DBP – diastolic blood pressure, GFR – glomerular filtration rate, HDL – high-density lipoprotein, LDL – low-density lipoprotein, NEU – neutrophils, LYM – lymphocytes, PLT – platelets, MPV – mean platelet volume, NLR – neutrophil-to-lymphocyte ratio, PLR – platelet-to-lymphocyte ratio, AASI – ambulatory arterial stiffness index
Table 5
Multivariate analysis of factors influencing concentrations of E-selectin and ICAM-1 in children with primary hypertension
[i] MAP – mean arterial pressure, BMI – body mass index, GFR – glomerular filtration rate, AASI – ambulatory arterial stiffness index, LDL – low-density lipoprotein, PLR – platelet-to-lymphocyte ratio, DBP – diastolic blood pressure, SBP – systolic blood pressure, NLR – neutrophil-to-lymphocyte ratio, HDL – high-density lipoprotein
In the subgroup of 27 patients with newly diagnosed hypertension there were no differences in concentrations of E-selectin and ICAM-1 between 15 children with isolated systolic hypertension and 12 children with systolic and diastolic hypertension. In this subgroup of 27 patients E-selectin correlated negatively with DBP dipping (r = –0.54, p = 0.004) and positively with AASI (r = 0.51, p = 0.012).
Discussion
Our cross-sectional analysis revealed that the strongest predictors of endothelial damage in pediatric patients with primary hypertension were MAP in ABPM (for E-selectin) and serum uric acid (for ICAM-1). Additionally, overweight and obesity, low HDL-cholesterol, increased BP variability, and higher degree of subclinical inflammation may also be factors predisposing to endothelial dysfunction in this group of patients. Interestingly, in the subgroup of untreated, newly diagnosed patients we found also positive correlations of E-selectin with night-time DBP dipping, and most importantly with stiffness evaluated as AASI.
The Stanislas study evaluated the concentration of adhesion molecules in healthy children and adults [17]. Both ICAM-1 and E-selectin were inversely related to age in paediatric patients, but this relationship disappeared in multivariate analysis for E-selectin [17]. There was no relation between sex and level of adhesion molecules. In our cohort we found no relation of these biomarkers with age, and the relationship with sex was not confirmed in multivariate analysis. The discrepancy between our results and results in healthy individuals must be analysed with caution. It is probable that other factors determine endothelial function in hypertensive and normotensive individuals.
There is a pathophysiological link between inflammation and concentration of circulating adhesion molecules. Leukocytes secrete cytokines that stimulate expression of adhesion molecules on the membrane of endothelial cells, thus facilitating adhesion and transfer of leukocytes into the arterial wall. Such a link was already proven in adults for TNF-α and E-selectin [17] and for CRP and ICAM-1 [18]. Also, in obese paediatric patients there was a positive relation between SBP, CRP, and VCAM-1 and between diastolic blood pressure, CRP, and ICAM-1 [10]. NLR and PLR, as well as MPV, are easily accessible indicators of subclinical inflammation. In our cohort, ICAM-1 correlated positively with PLR and, in boys, with MPV. In our previous study we found a positive relation between these indices and ambulatory blood pressure, albuminuria, and plasma renin activity in untreated adolescents with primary hypertension [8].
E-selectin and ICAM-1 correlated positively with BMI Z-score, ICAM-1 positively with uric acid, and negatively with HDL-cholesterol. The positive association between markers of endothelial injury and BMI is consistent with earlier observations in adults (E-selectin) [7, 17] and in children (E-selectin and ICAM-1) [9]. Increased levels of E-selectin in overweight, obesity, and dyslipidaemia could be explained by the adipocyte producing pro-inflammatory cytokines that induced E-selectin gene expression [19]. However, the relation between endothelial dysfunction and dyslipidaemia may be more complex because HDL-cholesterol was found to inhibit TNF-α and IL-1, cytokines that induce expression of adhesion molecules [20]. Accordingly, in our cohort there was a negative correlation of ICAM-1 and HDL-cholesterol. Similarly, in a study from Malaysia patients with low HDL-cholesterol were characterised by higher E-selectin and ICAM-1 concentrations [20]. Whereas, in the ELSA-Brasil study the highest E-selectin concentrations were associated with the highest total and LDL-cholesterol and triglycerides without differences in HDL-cholesterol [7]. In a paediatric study by Glowinska et al. total cholesterol and triglycerides correlated positively with E-selectin and ICAM-1 [9].
The increase in serum uric acid is considered to be not only a hallmark but also one of the triggering factors of PH [21]. Elevated serum uric acid may directly damage endothelial cells [22]. In our cohort there was a positive correlation between serum uric acid and ICAM-1, also confirmed by multivariate analysis. The same relation was found in obese prepubertal children [23]. Similarly, a negative relation between flow-mediated dilation and serum uric acid was found in obese boys [24]. There remains an unanswered question, whether lowering uric acid can reverse the negative phenomena observed in PH, including endothelial injury. Feig showed that treatment with allopurinol can normalise blood pressure in hypertensive adolescents [25], but a recently published trial failed to prove a beneficial role of allopurinol in normalisation of endothelial dysfunction in adults with PH [26].
Endothelial dysfunction promotes vascular smooth muscle proliferation and fibrosis of arterial wall, thus leading to impaired arterial contractility. The positive correlation found in our untreated hypertensive subjects between E-selectin and AASI might confirm this phenomenon. AASI is an ABPM-derived marker of arterial elasticity [14]. Although its clinical value in children remains unclear, it was found to be elevated in children with hypertension [27] and IgA nephropathy [28]. Nevertheless, the small number of patients (only 27 untreated children), the relationship found only in univariate analysis, and the uncertain significance of AASI preclude drawing final conclusions on the role of E-selectin as a serum marker of arterial stiffness. Eikendal et al. found a similar relation between E-selectin and aortal pulse wave velocity (PWV) in young adults [29]. In contrast, in a study by de Faria P-selectin and VCAM-1 but not E-selectin and ICAM-1 were predictors of increased arterial stiffness assessed by applanation tonometry [30].
In our patients, multivariate analysis unmasked a relationship between E-selectin and mean arterial pressure in ABPM. Positive correlations between ICAM-1, E-selectin, and SBP and DBP in ABPM were revealed in paediatric patients with type 1 diabetes mellitus [9, 31]. Also, E-selectin correlated positively with office blood pressure in adult individuals [7]. In addition, an association was found between genetic variants of E-selectin and risk of PH [32]. This positive correlation between blood pressure and markers of endothelial activation and injury highlights phenomenon of negative impact of blood pressure (“shear stress”) on endothelium. Of note, in our group a relationship between endothelial dysfunction was found only for MAP during 24 hours. Oscillometric devices directly measure the mean arterial pressure, and MAP during 24 hours seems to reflect most precisely the cardiovascular burden as shown in the ESCAPE study [33].
Our patients showed a positive relation between ICAM-1 and blood pressure variability. Increased blood pressure variability is an independent cardiovascular risk factor [34]. In adults a positive correlation between blood pressure variability and E-selectin [35] and ICAM-1 [36] has already been found. To the best of our knowledge, this is the first study to confirm this relation in paediatric patients with PH. The hypothesis that short-term blood pressure oscillations lead to vascular damage may be a causative link between these two phenomena [35]. In 27 individuals with newly diagnosed hypertension we found a negative correlation between E-selectin and night-time diastolic blood pressure dip. Paediatric patients with PH and disturbed circadian blood pressure profile are at risk of left ventricular hypertrophy [37]. Moreover, many studies showed that nocturnal hypertension is a risk factor for kidney function decline [38]. There is still debate whether endothelial dysfunction leads to blunted night-time dip by increasing arterial stiffness. Conversely, according to opposite theory, increased night-time shear stress damages endothelium. The cross-sectional character of our study precludes drawing final conclusions on this topic.
Increased urinary albumin loss is not only a marker of glomerular hyperpermeability but also reflects endothelial dysfunction with strong correlations between albuminuria and cardiovascular risk [39]. In diabetic adults ICAM-1 showed a positive correlation with AER. Moreover, individuals with the highest values of ICAM-1 had the highest increase in AER after three years [40]. No relation was revealed between markers of endothelial injury and albuminuria in our cohort. Of note, there are no data linking albuminuria in children with hard cardiovascular end-points in prospective observation, and the American Academy of Pediatrics resigned from routine assessment of albuminuria in hypertensive individuals [41].
In our cohort ALP activity was positively associated with E-selectin and ICAM-1 concentrations. The same correlation was observed also in healthy individuals [17] and for ICAM-1 in patients with hepatic disorders [42]. It must be emphasised that ALP activity remained within normal limits in all our subjects, and none of them suffered from bone or hepatic pathology. We did not find in the literature any explanation for the positive relation between endothelial adhesion molecules and ALP in a physiological state.
The limitations of our research are a lack of analysis of adhesion molecules in healthy individuals, indirect analysis of arterial stiffness only by ABPM, and assessment of target-organ damage limited to albumin excretion. In addition, this was a cross-sectional study, therefore a causal relationship cannot be inferred. Also, the study group was not large and not uniform, especially in terms of the treatment used; thus, detailed analysis in subgroups was not possible and final firm conclusions cannot be drawn uncritically. Nevertheless, owing to the fact that we analysed a relatively large group of individuals and blood pressure is a continuous variable with arbitrary definition of hypertension, our result can be cautiously extrapolated to the whole paediatric population.