Introduction
Natural killer cells (NK cells) are a subset of non-B and non-T peripheral blood lymphocytes, which appear to play a crucial role in the human innate immune responses. NK cells represent an “evolutionary bridge” between innate and adaptive immunity [1, 2]. They mediate a cell contact-dependent cytolysis of target cells, including those expressing foreign major histocompatibility complex (MHC) molecules, immunoglobulin-bound antigens, virally infected cells, and tumor cells. The function of NK cells is important for clearance of tumor cells, removal of immunoglobulin-bound antigens, and for controlling viral infections [3, 4].
The role of NK cells in reproductive processes, such as implantation, trophoblast invasion, and spiral artery remodeling is critical, but still poorly understood [5]. The accumulating evidence links elevation of NK cells with unfavorable pregnancy outcomes in women [6-12]. NK function has been reported to be decreased in healthy pregnant women, persons with primary immunodeficiencies, and those with late-stage human immunodeficiency virus infections [13-15]. Resent progress in development of better methods for quantitative and functional assessment of NK cells might help to define their role in reproductive development and other clinically important mechanisms. For example, NK cell activity has already been established as an important indicator for predicting patient outcomes in certain cases [13-16].
Peripheral blood NK lymphocytes are a highly heterogeneous population of cells that differ by expression of surface ligands, a repertoire of KIR receptors, ability to secrete cytokines, and cytotoxic responses. All these NK cells share the ability to express CD56 in the absence of CD3 [17, 18]. Therefore, the heterogeneity of NK subsets is the major contributor to their multi-functionality. This population is named after the cytotoxic reaction measured by the NK assay against HLA-negative K562 cell line, yet only a part of NK cells manifests cytotoxicity. In our previous study, we demonstrated that only a subset of peripheral blood NK lymphocytes is able to respond to the K562, and that only a fraction of this subset determines levels of NK cytotoxicity in the entire population [19]. The amount of active NK cells varies from individual to individual, and their function can be easily modulated. However, this very group of cells tightly correlates with the actual NK cytotoxicity [20]. We also showed that balanced “optimal” counts of this fraction strongly correlate with successful embryo implantation in IVF stimulation cycle. Although, misbalance of NK activity is (decreased or increased active NK counts) negatively impacts the embryo implantation in IVF patients [16]. Interestingly, total NK cell counts did not appear to correlate with such clinical outcomes. Instead, NK quantity seemed to convert to NKc indirectly via a functional subset of active NK cells “the NK activity coefficient”.
In the present study, we analyzed the effect of various levels of correlation between NK cytotoxicity and NK frequency, depending on NK cytotoxicity and NK lymphocytes levels-status.
Material and methods
Study design
In this retrospective study, we analyzed our laboratory data of 2,804 women, who underwent routine immunology investigation after IVF pregnancy failure. Data was acquired from March 2008 till October 2015 (1-3 months after last IVF cycle or 3-6 months after last pregnancy failure). The patients were under 39 years (average, 31.2) and had at least 1 episode of idiopathic IVF failure (average, 2.32) or at least 1 episode of idiopathic first trimester pregnancy failure (average, 2.04). The patients had no supplemental treatment with immunoglobulin or corticosteroids in the last 3 months. None of the patients had an active autoimmune disease or a history of autoimmune disease, and no patients had an active infection within at least 3 weeks. All of the study subjects signed an informed consent form before being enrolled into the study (approved by the Biomedical Ethics Committee of Institute of Pediatrics, Obstetrics, and Gynecology, National Academy of Medical Sciences of Ukraine, No. 6 dated December 17, 2012, and Clinical Immunology and Allergology Committee of Ministry of Health, No. 11 dated February 14, 2013, according to Declaration of Helsinki). Peripheral venous blood samples were drawn by venipuncture, and immunological studies (NK frequency and NK cytotoxicity) were performed in the Laboratory of Immunology, Institute of Pediatrics, Obstetrics, and Gynecology of the National Academy of Medical Sciences, Kyiv, Ukraine.
For NK cytotoxicity distribution, we analyzed our laboratory data of 9,535 individuals from similar clinical group. In isolated study for comparison of NKc data, we analyzed 823 individuals’ data, where NKc was measured by TDA relies assay. All investigations were made in 5 h period after obtaining blood samples.
Flow cytometry of lymphocyte subsets
Lymphocyte subsets were identified by three-color flow cytometry, using erythrocyte-lysing whole blood method of lymphocyte staining by FITC-, PE-, or PE-Cy5-conjugated monoclonal antibodies (BD Bioscience, San Jose, USA). Stained samples were lysed, washed, and analyzed by FACScan flow cytometer, using CellQuest software (BD Bioscience, San Jose, USA). NK lymphocyte was calculated as the percentage of CD3-CD56+ lymphocyte (include CD56++ subsets).
NK cytotoxicity assays
Briefly, a total of 2 × 106 PBL purified from peripheral blood of patients by Histopaque-1077 (Sigma-Aldrich, MO, USA) gradient were washed in PBS. Flow cytometry NK cytotoxicity assay (FCA) was described previously [20]. We used CMFDA (Molecular Probes, Eugene, OR, USA) for labeling K562. After 4 h of incubation, dead cells were labeled by PI. Less than 5% spontaneous lysis of target cells was observed in these experiments. We used 50% permeabelized K562 culture for targets-gate correction. 100 µl target cell was killed by 50 µl ethanol (96%). After 10 sec wortexing, 100 µl of PBS and 100 µl of unaffected targets were added. The suspension obtained consisted of 50% of alive and 50% of permeable cells.
TDA release NK cytotoxicity assay
TDA release NK cytotoxicity assay (TDARA) was described previously [20]. Targets K562 (a human erythroleukemia cell line) in RPMI 1640, supplemented with 10% bovine serum were labeled at concentration of 1 × 106 cells/ml, with 5 µl BATDA reagent (bis acetooxymethyl 2,2’ : 6’,2’’-terpyridine-6,6’’-dicarboxylate, PerkinElmer Wallac, Turku, Finland) for 20 min at 37°C. Labeled cells were washed three times in RPMI/10% bovine serum and incubated with PBMC at various effector/target ratio (E : T 20 : 1, 10 : 1, 5 : 1). After co-incubation of the effector and target cells for 4 h at 37°C in a humidified 5% CO2 incubator, the cells were mixed and centrifuged, and 20 µl of the supernatant was mixed with 180 µl europium solution (PerkinElmer Wallac, Turku, Finland) and incubated for 15 min at room temperature, while shaking in a flat-bottom 96-well plate (Nunc, Denmark). Time-resolved fluorometry was carried out with ARCUS (Wallac Oy, Turku, Finland). The percentage of specific lysis was calculated as ((experimental release-spontaneous release) / (maximal release-spontaneous release)) × 100%. The maximal release was determined by addition of Triton X100 in a final concentration of 0.25%. The assay was performed in quadruplicate for each E : T ratios.
For both assay individual, cytotoxic activity was calculated as % of cytotoxicity (E : T ratio 15 : 1 as average from levels at 20 : 1 and 10 : 1).
Intra-laboratory normal levels were obtained from 90% CI calculated for 74 healthy fertile women with subsequent normal pregnancy development [11].
Results
Distribution of values
We analyzed the distribution of NK lymphocyte levels in 2,804 patients and determined the average counts to be 12%, typical for healthy Caucasian women [21, 22]. Only 1.9% (55/2,804) of patients had NK levels > 26.1%, and 4.6% (131/2,804) patients had NK levels < 4.8% (Fig. 1A). According to our clinical normal reference [1, 23] for patients with favorable implantation and pregnancy prognosis (5.5-21%), the decreased NK% was observed in 5.1% (144/2,804) patients and increased NK% in 6.2% (174/2,804) of the investigated patients’ population.
Fig. 1
(A) Distribution of levels natural killer (NK) lymphocytes and (B) NK cytotoxicity in patients’ groups. A) NK (CD56+CD3-) cells % from all lymphocytes (n = 2,804), B) NK cytotoxicity (E : T, 15 : 1) was measured by FC NK assay against K562 (n = 9,535). * normal value for pregnancy prognosis (3-12%) [6], ** normal value for pregnancy prognosis (5-18%) [7], *** our laboratory favorable value for implantation and pregnancy prognosis (5.5-21%) [11], **** reference value for young Caucasian women (4.8-26.1%) [38], @ our laboratory favorable value for implantation and pregnancy prognosis (12.5-41%) [11]
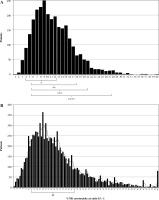
Distribution of NKc and NKc average levels was comparable to published results from other groups [13, 24], with a notable trend towards increased and decreased values. According to our clinical references for patients with favorable implantation and pregnancy prognosis [11, 23], we showed that 13.1% (369/2,804) of patients presented decreased NKc, whereas 19.4% of patients (545/2,804) had increased NKc levels (Fig. 1B).
NKc was elevated in 20.7% (51/246) of patients with 1 episode of reproductive failure (pregnancy or IVF failure) in contrast to 34.5% and 31.3% of patients with 2 and more IVF failure (297/843), or 2 and more pregnancy failure (316/1,008) episodes.
Correlations
Consistently with our previous reports [20], the frequency of NK lymphocytes in the whole clinical population strongly correlated with NK cytotoxicity (r = 0.4268, p < 0.0001) (Fig. 2 and Table 1). Also, we did not find a significant correlation between NK counts and NK cytotoxicity in the group of patients with “normal NK levels”. In contrast, this correlation was strong in patients’ groups with decreased or increased NK frequency. To investigate this phenomenon closer, we divided the population into groups (n = 400) according to the individual NK lymphocyte counts. We found strong NK%-NKc correlation (r = 0.2021, p < 0.0001) in a group of patients with high NK% (> 17.5%) (Table 1). Lower but also significant correlation was observed in patients with NK% between 15% and 17.5% (r = 0.1213, p = 0.0155). The absence of NK%-NKc correlation was observed in the patient groups with following NK% (12-15%), (12-10%), (10-8%), and (8-7%). However, strong correlation (r = 0.2689, p < 0.0001) between NK%-NKc re-appeared in the patients’ group with NK levels < 7 % (1.7-7%) (Table 1).
Fig. 2A
Correlation of natural killer (NK) frequency with NK cytotoxicity in whole population (n = 2,804) and (B) in separate zones of NK% distributions
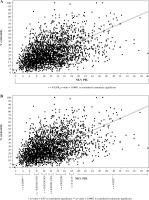
Table 1
Correlation between natural killer (NK) frequency and NK cytotoxicity in groups divided according to NK numbers
These findings agree with data obtained using an alternative NKc assay (Eu-Badta). In contrast to FC NK assay, where dead cell targets became permeable for PI, this modification measured cytotoxicity as Badta reagent release from lysing targets. Similarly, to the study using FC NK assay, we grouped patients according to NK lymphocyte levels (each group of approximately 200 patients). Our data analysis revealed significant NK%-NKc correlation in patients’ groups with NK% < 7.5% (r = 0.212, p = 0.0026) and > 15% (r = 0.2596, p < 0.0001). In patients with NK% (7.5-10.5%) and (10.5-15%), NK%-NKc correlation was not significant (r = 0.123, p = 0.08) and (r = 0.14, p = 0.07), respectively. Therefore, we showed that NK%-NKc correlation depends on NK frequency status.
Next, we wanted to determine whether NKc-NK% correlation may depend on NK cytotoxicity status. Similar to our previous analysis, we investigated NK%-NKc correlation in patients grouped according to their NKc score. Significant correlations were observed in groups with accentuated/elevated (> 45.5%) or downregulated (< 13%) NKc at E : T ratio of 15 : 1 (r = 0.2725, p < 0.0001) and (r = 0.1707, p = 0.0006), respectively. In groups with moderate NKc levels (> 13 and < 45.5), no significant correlation was found (Table 2).
Table 2
Correlation between natural killer (NK) cytotoxicity and NK frequency in group divided according to NK cytotoxicity levels
Here, we demonstrated that variation of quality status of NK population plays an important role in correlative association with their cytotoxic activity. Therefore, the quantitative changes within accentuated zones affect the quality of associative character in NK%-NKc and its impact on clinical outcomes.
Discussion
NK frequency and NK cytotoxicity are significant parameters that determine clinical outcomes. Recently, several independent groups showed that individuals with elevated NK lymphocyte levels and NK cytotoxicity present lower risk and better clinical outcomes in viral infections and oncology diseases, compared to patients with lower NK% and NKc levels [25-29]. In contrast, increased NK levels after ischemic stroke have been associated with increased risk of sepsis [30]. Also, elevated NK activation correlates with liver injury in chronic hepatitis B patients [31]. In transplantation patients, NK role was shown to be ambiguous, according to recent reports [32, 33]. In these studies, NK cells exacerbated T cell responses during transplant rejection, but also promoted tolerance induction under immunosuppression. On animal model, NK cells play a critical role in cardiac allograft vasculopathy and long-term kidney allograft injury [34, 35]. Growing amount of evidence [6-12] suggests a link between increased NK% and NKc and reproductive failure.
Despite these facts, the question still remains: what are the normal (optimum) levels of NK? NK% varies depending on gender, ethnical, and racial backgrounds [36-39]. Moreover, NK% and NKc levels are higher in males compared to females. This difference is significant in reproductive age. In females 40 and older, these differences become less pronounced [40, 41], while NK% and NKc levels are markedly reduced in pregnant women. Not surprisingly, NK system attracts special interest in the field of immunology of reproduction. It is accepted that NK cells play a critical, yet poorly defined role in reproductive processes, such as implantation, trophoblast invasion, and spiral artery remodeling [5]. On the one hand, elevated NK cell frequency [7, 8] cytotoxicity [9] and imbalance between inhibitory and activating receptor expression on NK cells [10, 11] are associated with different reproductive failures [12]. On the other hand, NK cell activity is also necessary for reproduction; it was shown that fetal growth restriction is associated with reduced proportions of NK in the decidua basalis [42], and that NK cell depletion in mice leads to placental abnormalities [43], thickening of the spiral artery [44], and fetal loss [45]. At different times, several groups proposed to define “reproductive normal values” of NK frequency [7, 46, 47]. Following values were suggested as reference points < 12%, < 15%, or < 18%, respectively, as favorable values for implantation and normal pregnancy outcome. In contrast, the elevated NK counts were suggested as indicators of increased risk of reproductive failure in women.
Undoubtedly, the predictability of prognosis using NK frequency as well as the role of elevated NK% in reproductive outcome are still up for debate [48, 49]. Many investigators agree that measurement of the functional activity of NK cells is more important for clinical prognosis [1]. However, limited affordability, complexity, and lack of standardization were the main reasons that this marker is still not a part of the routine clinical diagnostics. In contrast, the characterization of NK phenotype markers is much easier and possesses comparable clinical utility. It was shown, for example, that misbalance between KIR receptor activation and ligand subsets is associated with reproduction failures [48, 50].
In this study, we demonstrated that different qualitative states of NK population have different correlative associations with cytotoxicity. The absence of correlation between NK% and NK cytotoxicity was found in the moderate-to-normal zone of NK frequency. This could mean that in this zone, the counts of NK cells do not directly determine cytotoxicity. Instead, the NK lymphocytes counts in the moderate-to-normal zone are sufficient and are compatible with the generation of any NKc levels. Associations between NK% and NKc are more quantitative in nature. Within the balanced “normal” zone, NK cytotoxicity levels rather than NK frequency is determined by proportions of active subsets. This regulatory mechanism adjusts NKc to the actual (necessary) situation, thus, reversing the role of NK frequency in these zones. Here, in the “normal zone”, NK numbers are not directly equated to NKc. In contrast, accentuation of NK through inflammation-affectation results in decreased/increased NK numbers and deregulation of the clonal population balance. Affectation of regulatory mechanisms results in the direct impact of NK frequency on cytotoxicity. Therefore, the quality directly determines functional quantity. Hence, it is possible that increase or decrease in NK frequency is regulated largely through cytotoxic NK subsets proportions.
Increased NK% and NKc correlation is a first unfavorable accentuation marker displayed during reproductive development [7]. Previously, we showed that some accentuations have negative predictive value for implantation and pregnancy outcome. The majority of those outcomes were associated with NK surface markers expression and their activity. Among them, NK accentuations possess bilateral features. Decreased as well as increased CD8, CD158 expression, and CD69 activation proved to be predictive of the negative clinical outcome [16, 23, 51]. In the case of accentuated T lymphocytes, negative predictive markers were increased (HLA-DR CD56, CD158) or decreased (CD4 levels) [52].
Analyzing the predictive significance of CD8 expression on NK, we demonstrated that elevated levels are associated with implantation failures, while decreased CD8 levels were associated with subsequent pregnancy failures. We also have shown that both accentuated phenotypes’ individuals have a higher frequency of other accentuations compared to patients with balanced NK-CD8 expression [51].
In this investigation, we demonstrated that in addition to clinical manifestation, NK% accentuations lead to a deregulation of NKc. Thus, NK frequency indirectly affects NK cytotoxicity through the regulation mechanisms and subsets balance. However, in the accentuated zones, this mechanism is not enough to correct and modulate physiological (necessary) conditions. A demonstrated phenomenon supports our idea about immune accentuation and sheds light on its physiological role.
In this study, we analyzed the levels of NK cells in patients with difficulties in reproduction. According to the levels of NK cells, the patients almost conformed to the known reference levels for young healthy women of the same ethnic group [21, 22]. Many studies reported higher levels of NK cells in healthy fertile women compared to IVF (in vitro fertilization) and RSA (recurrent spontaneous abortion) patients [6-11, 54]. The probable explanation might lie in dissimilarity of reproductive state within the “somatic healthy” women group. In today’s world and in our local population, most of women delay childbirth to a later point in their life. In recent years, the prevalence of sub-fertile and infertile states markedly increased. Most of these states are not associated with general health indicators and are hard to predict. Therefore, the likelihood of occurrence of such adverse states in some of the “somatic healthy” women at a future time point is relatively high. Additionally, IVF failure and RSA diagnoses are being made usually after three consecutive first-trimester pregnancy failures with the normal fetus (chromosomally and without pathology), or three consecutive IVF failures with a high quality of embryos. In our group, most patients chose not to wait for the confirmation of their clinical state and requested immunodiagnostics early in pregnancy. Thus, significant part of the study population had no definite diagnosis of idiotypic IVF failure and RSA. Similarly, the population labeled as the “somatically healthy” women were not formally confirmed as reproductively healthy. In fact, reproductive disturbances of the immune genesis are not often accredited to the general clinical status, but rather a situational combination of adverse immunological, physiological, and genetic factors. Therefore, it is not always true that the genetic mother-fetus combination and functional state of a female patient is a lasting independent predictive marker of unfavorable outcome. Moreover, the fertility can only be confirmed by the onset of pregnancy and carrying of pregnancy in the given cycle instead of the normal range of different parts of the immune and endocrine systems. Interestingly, the fact that the NK percentage is gaining a new role when passing a certain threshold, might help to determine the physiologically-balanced standard range for specific clinical groups. Our study demonstrates that by knowing this range, we might be able to predict and/or create more favorable corridor for the course of pregnancy.
NK cells testing is still a controversial issue in RM and RIF patients. This controversies start to be a separate topic of debates [53, 54].
In multivariate analysis [55], it was shown that blood NK cells expansion was an independent risk factor for RF (both recurrent miscarriages and implantation failures). Significance of this risk factor is growing with patient’s age. In contrast, in same year and in similar patients’ numbers was shown [56] that elevated pNK cell activity is not independent risk factor for subsequent miscarriage. In same investigation, untypical age-associations with subsequent miscarriage were observed. Older women (32-35 years old) have better pregnancy outcome than younger (30-31 years old). Also, it was reported that Japanese local population was practically devoid of HLA C2 carriers and predominantly presented KIR AA+ [57]. Frequency of risk pregnancy combination (fetal HLA C2+ with mother KIR AA) in this population was minimal, which could possibly reduce the significance of NK activity for clinical outcome.
Conclusions
It is possible that accentuated NK function form unfavorable background for reproduction, but this risk is realized indirectly through associations with other immune-endocrine factors, genetic deviations, or mother/fetus combinations. It was observed [58] that NK cytotoxicity is the effector mechanism of placental dysfunctions in alloimmune thrombocytopenia model. Anti-β3 integrin ab and endothelium cells activation was the origin of pathology that causes damage in placenta through NK cells cytotoxicity. Inhibitions of NK cytotoxic receptors completely revoke placenta dysfunction.
It is too early for a reliable NK cells testing in routine diagnostic, but at same time, it is too late to ignore these parameters in the formation of reproductive unfavorable conditions.


