Purpose
High-dose-rate brachytherapy (HDR-BT) is an effective modality. However, its’ application involves complex and inter-disciplinary procedures. Safe and effective HDR-BT requires a collaboration of many specialists from different fields, with varied skill sets. There are complex interactions between members of this heterogeneous team using manual and automated systems, and there is a high degree of human-computer interface. Together, these attributes create an environment with a heightened risk of errors. Misadministration of HDR-BT through inappropriate clinical procedures or inadequate coordination of BT team activities can negatively affect patients.
Several organizations have developed practice guidelines as a resource for radiotherapy (RT) centers worldwide to reduce unwanted events. Primary aims of such procedures are to standardize and improve treatment outcomes of patients [1-10]. The American Society for Radiation Oncology commissioned the evaluation of a status of safety and practice guidance for HDR-BT [6]. The International Atomic Energy Agency reviewed the safety and quality of radiotherapy [11]. A risk assessment approach was introduced using TG-100 Methodology with failure modes and effect analysis (FMEA) for safety and appropriate use assessments [12-14]. Risk and benefit balance impact template [15] was also published to facilitate the implementation of new radiation oncology technologies and techniques.
Furthermore, the World Health Organization (WHO) encouraged global RT centers to review and use patient safety events (incidents, near-misses, and unsafe conditions) to improve and guarantee safe treatment. Methods for implementation of patient safety events were described in the WHO technical report, incident reporting, and learning system [16]. Moreover, the International Atomic Energy Agency promulgated safety in radiation oncology (SAFRON) program, an integrated voluntary reporting system for radiotherapy incidents and near-misses. It is an excellent resource for educating staff on the need of unified approach to improving safety culture [17, 18].
The 2019 annual survey of the Thai Association of Radiology and Radiotherapeutic Oncology revealed that HDR-BT was applied to 5,400 cancer patients in 31 RT centers throughout Thailand [19]. Almost 99% of these patients presented with gynecological cancer. Most Thai RT centers observed internationally recommended HDR-BT safety and quality practices. These protocols are described in documents produced by organizations, including the American Association of Physicists in Medicine, the International Atomic Energy Agency, and the European Society for Therapeutic Radiology and Oncology [21-25].
According to WHO technical report, most of the experience in patient safety incident reporting and learning systems has been gained from hospitals in high-income countries. Little has come from low- and middle-income countries. There are numerous obstacles in capturing and learning from incidents. Among them are: 1) insufficient leaders with the skill sets and passion needed to engage their workforce in safety; 2) the inability of healthcare professionals to freely report on occurrences of harm, errors, near-misses, and risks; 3) an inability to investigate a large volume of reports; and 4) a insufficient evidence base on how to reduce harm.
In 2012, the application of HDR-BT at our center has been transitioned from two-dimensional BT (2D-BT) to three-dimensional image-guided BT (3D-IGBT). Concurrently, a patient safety program was established to handle a more sophisticated technology and its’ workflows. Following 9 years of experience with HDR-BT, the present incident review was performed to elucidate the factors associated with RT safety. As a large academic center in Thailand, we anticipated that our incident learning would: 1) improve safety information at RT centers nationwide, and 2) upgrade quality management systems to help preventing reported RT incidents.
Material and methods
Patient safety program
Team members and safety perceptions
Complex 3D-IGBT procedures present many opportunities for errors. Their complexity requires comprehensive quality management and quality assurance for the treatment unit. Before the 3D-IGBT system was introduced into routine operations, chief BT medical physicist (MP) conducted safety training sessions for BT lead team to establish the best practices, and to develop an exceptionally reliable BT center in Thailand. With the perception of safety, each professional group clearly defined its’ roles and responsibilities for specific tasks related to introducing the upgraded technology. Clinical workflows, plans, and resources were reviewed, and FMEA analysis tool was applied to develop interventions to eliminate risks to patients arising from any treatment process.
To reduce risks, nursing team who aided radiation oncologist (RO) in the insertion theatre was trained in appropriate use and care of BT applicators. Nurses understood the impact of applicator failure on patients through poor construction, inadequate maintenance, and inappropriate applicator fixation or use. Radiation therapists (RTTs) acknowledged the impact of applicator slippage between the simulation process and treatment delivery, and the incorrect use of transfer tubes on dosimetric accuracy. Moreover, treatment planners and clinical MP understood all uncertainties from source commissioning using wrong image, wrong prescription dose, incorrect definition of the applicator length, incorrect definition of starting location for the source, incorrect catheter placement or shift from its’ intended position, and inappropriate optimization in 3D-IGBT.
We also established the treatment plan review procedure for consistency and validity of the data transferred from the treatment planning system to the treatment unit. Drawing upon the treatment plan, the responsible RTTs and MP checked accuracy of parameters for the source strength, dose per fraction, identified the number of applicators connected and their placement order, applicator length, step length, correct values of dwell location, and time for each channel. Before starting treatment, correspondence of the applicators with indexer channel number and treatment plan needed to be verified. This step ensured that the connections of transfer tubes to the applicators and the transfer tubes to the indexer were correct, and that the tubes were not bent.
Communication and verification
In addition to being a complex and busy task, HDR-BT is a multidisciplinary treatment modality requiring coordination of several professionals. Effective communication among BT team members is crucial for safety. At our center, an information sheet shown in Figure 1 was developed to transfer and receive all patients’ treatment details relating to individual fraction, from insertion to simulation, planning, treatment delivery, off-applicator, and safe discharge. Each workstation received this information sheet with a patient, and was required to review and understand all procedures carefully. Any concerns about patient’s name or identification number, fraction number, details of inserted or implanted applicators, simulation, bladder filling protocol, prescribed dose, or radiation monitoring safety were required to be fully resolved prior to any task being performed on the patient.
Knowledge management
In 2017, the department underwent significant development, with a noticeable increase in inexperienced staff and revised shift patterns (3-5 MPs per week and 3-4 RTTs every 2 weeks). As an academic center in Thailand, we already had the role of conducting RT education programs for residents, fellows, and national and international trainees. Therefore, to enhance the effectiveness, distribution security, and use of external and internal communications, we established an annual knowledge management program on HDR-BT for newly recruited staff, new residents, and trainees. In each program, a small group of 8 to 10 staff members with varied tasks was rotated through 3 knowledge stations. Training started at the insertion theatre. BT procedure was reviewed there, and key points relating to patient safety were emphasized. Given the advanced designs of applicators used in 3D-IGBT, we encouraged participants to familiarize themselves with the applicators by assembling them in hands-on sessions.
Senior BT RTTs were responsible for the second station (simulation and treatment delivery). Details of the routine imaging protocol and workflow for 3D imaging acquisition (using a GE computed tomography scanner and a Philips magnetic resonance imaging scanner) were shared and demonstrated. The same was done for correct and safe treatment delivery with Varian VariSource afterloader system.
At the third knowledge station, MP presented how quality treatment of an excellent standard was implemented through the physics and planning procedure. Descriptions were also given of potential errors and their causes (based on FMEA analysis), and methods for developing interventions to prevent their occurrence. Staff understanding of the workflow and lessons learned from reported errors were shared in a discussion session. This knowledge management strategy presented an excellent opportunity for staff education on proactively preventing potentially harmful errors.
Incident/near-miss reports
At our center, all unusual reported RT events were investigated, especially those presenting a potentially significant risk to patients. With BT service, the incidents involved non-dosimetric and dosimetric errors (actual incidents and near-misses), associated instruments, treatment procedure, and radiation safety at the time of discovery by frontline staff. In early years of operation, events were fully and accurately documented manually in an incident logbook; later, details were recorded electronically. Root cause analyses were performed for events related to patients’ safety by a team (consisting of RO, MP, RTT, and nurse), and through monthly department service and safety meetings. Events and review outcomes were reported to the faculty’s Risk Management and Patient Safety Division.
Results
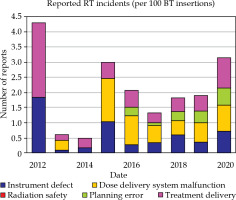
At the Radiation Oncology Division at Siriraj Hospital approximately 250 patients with 900 HDR-BT insertions were treated annually. Virtually, 99% of insertions were performed in endometrial or cervical cancer, with a tiny number in prostate, esophageal, or head and neck tumors. HDR-BT program for gynecological cancer patients with VariSource afterloader system evolved from simple 2D planning into 3D-IGBT in 2012. It subsequently moved almost entirely to hybrid computed tomography/magnetic resonance (CT/MR)-guided volumetric planning, with interstitial plus intra-cavitary technique in 2018 (Table 1). All issues arising from BT application (99% gynecological cancers with about 2% of HDR prostate implantation) during more than 9 years of implementation were collected and categorized (Table 2). Reported RT incidents over time by event type are shown in Figure 2. The incidents were classified as ‘near-miss’, ‘not harmful’, and ‘harmful’ according to the International Classification for Patient Safety [26]. They are summarized in Table 3.
Table 1
Statistical reports of HDR-BT patients, insertions, and techniques between 2012 and 2020
Table 2
Summary of overall reported occurrences and categorizations
Table 3
Summary of incident reports according to international classification for patient safety [26]
Incident analysis and learning
Principles and methodology of incidents’ analysis addressed system and human factors. All information on an incident was first gathered to determine what happened and its’ cause. A clear understanding of the nature of errors enabled investigative team to identify the key contributing factors and actions needed for improvement. The following cases describe the patient safety events, incident analyses, and learning outcomes of 4 dosimetric errors at our center. For dosimetric severity, a mild severity corresponds to a ≤ 5% tumor underdose or organ at risk (OAR) overdose of the intended doses throughout treatment, moderate severity refers to a > 5% and a ≤ 25% tumor underdose or OAR overdose, and high severity to a > 25% tumor underdose or OAR overdose [27]. Cases are summarized in Table 4.
Table 4
Patient safety events and incident analysis
Case 1: Double-treatment dose delivery (moderate severity, minor medical harm)
Source of error: mismatch between the planned prescribed dose and the target dose objective for inverse planning optimization.
A dosimetric error occurred during the first year after implementing the complex 3D-IGBT system. The incident stemmed from 2 planners working together to treat a patient with recurrent cervical cancer receiving many needle implants. The patient had a large necrotic mass located outside the vagina. It was planned to administer a treatment dose of 10 Gy in 3 fractions, and 2 planners helped each other run the complex protocol. The mistake arose when the first planner incorrectly prescribed an input dose of 5 Gy. After applicator reconstruction with more than 20 catheters was completed, the second planner decided to use inverse planning to create dose distribution for this complicated implantation. A correct target dose of 10 Gy was specified for inverse plan optimization, and 10 Gy isodose line coverage was generated for plan evaluation. Consequently, the second planner did not recognize any plan defect.
When the plan was completed, it was approved by a responsible RO and sent to the control workstation for delivery process. There, the incorrectly prescribed dose of 5 Gy was detected, and delivery dose was doubled. This decision was made against a background of an unfamiliar procedure, a fear that the patient would receive inadequate treatment dose, and inadequate understanding of treatment planning system. However, the core problem was a misunderstanding arising from a lack of communication between the two planners. An RT error was detected in fraction delivered the next day, and dose severity and harm to the patient were subsequently evaluated. Since the patient’s tumor mass was located outside the body, only the effect of overdose on gross tumor volume and urethra was determined, and reported to RO. Treatment was paused, and the dose to the urethra was found to have increased from 32 to 40 BEDGy3.
Case 2: Dose delivery to unintended location (mild severity, no medical harm)
Sources of error: incorrect treatment length input, different length transfer tube.
VariSource afterloader system uses two types of transfer tubes. One is black and the other is green, and they have different lengths. Black tube (ClickFit TransferGuide Tube Series GM 19001000) is used to connect with tandem, ring, and ovoid applicators. With a 32 cm long applicator attached, a black transfer tube is 120 cm long for treatment planning purposes. In contrast, the length of green transfer tube (Needle ClickFit TransferGuide Tube Series GM 19001000) is 100 cm. The channel length input depends on a specific length of the applicator being used.
Our center routinely use black transfer tubes to connect the first 3 channels of tandem, ovoid, and ring applicators. An incident occurred in a cervical cancer patient with a large high-risk clinical target volume. She received intra-cavitary/interstitial BT, with another tandem applicator added to improve dose coverage. The second tandem was labeled applicator number 9 in insertion theatre. The planner should have input a length of 132 cm in recognition that the applicators were connected to the green transfer tube, not the black transfer tube. However, the planner was new to BT planning and only familiar with a 120 cm treatment length for a tandem applicator. Consequently, the incorrect length of 120 cm was input rather than the correct length of 132 cm.
After the plan was completed and approved by RO, the plan review performed by MP and RTT failed to detect an error. As the incorrect delivery dose details were input to the aria information system, radioisotope source was loaded 12 cm lower than the intended location. However, this patient with implanted applicators was scheduled to undergo next fraction the following morning. The error in the channel length was discovered during plan review process. Dosimetric error and severity were evaluated and reported as a no harm incident to RO.
Case 3: Dose delivery to unintended location (mild severity, no medical harm)
Source of error: incorrect transcription of needle length.
One incident of planning error and a few near-misses were reported because of the insertion theatre staff’s incorrect transcription of the needle length. RT nursing staff acknowledged that a dosimetric error arose from incorrect information. Nursing team subsequently revised their training on specific types of applicators. Additionally, in a complex intra-cavitary/interstitial BT involving many implanted needles, it was determined that arranging the needles in order of length would help to reduce the potential for confusion when transcribing details, and assist treatment planner. An upgraded sign-out process for nursing team was also identified as crucial in reducing errors.
Case 4: Treatment delay due to delivery dose error (mild severity, no medical harm)
Source of error: source blockage, contaminated source.
Most often, occurrences that caused a treatment delay for patients were produced by source blockage and stuck sources from contamination with body fluids. In case of source blockages, the position of transfer tube connected to VariSource afterloader system was overly sensitive if it was not parallel and straight (in other words if it was bent or kinked). Of the 7 reported incidents, 4 patients received a lower treatment dose because the problem with applicators had to be terminated. The other 3 patients received the intended treatment dose after dose optimization was revised to maintain the required dose objective for remaining applicators.
Discussion
The provision of patient safety requires a framework of organized activities, which create cultures, behaviors, procedures, and environments that minimize or prevent adverse events, and reduce their impact on patients. Identifying communication failures and understanding the nature of errors would help to avoid mistakes and technical failings related to using HDR-BT. Achieving safe and effective HDR-BT requires systematic processes to identify critical incidents and manage them appropriately. The need to report incidents and belief that learning from incidents and near-misses would improve patient safety have been advocated and are widely accepted [28-30]. A well-designed safety management system should empower the workforce to identify, report, manage, and learn from incidents.
The most frequent rate of reported RT errors in radiation oncology results from failures in workflow and processes [31]. Another core factor in the effectiveness of treatment process and delivery is the functionality of equipment, which requires careful monitoring by MP and RTT [32]. In a risk analysis of BT accidents, many events arose from failure to embed human factor considerations in equipment design [32]. Regarding learning from treatment accidents, more than 500 HDR-BT accidents were provided in Report 97 of the International Commission on Radiological Protection [32]. The rate of medical events in HDR-BT was also estimated to be 0.02% of procedures [34]. Felder et al. reported a safety-related event rate of 2.8% for BT patients and 1.7% for BT fractions [35]. Adverse events associated with afterloading HDR-BT reported to the United States Food and Drug Administration were also described by Provenzano et al. [36].
In the present study, there were 164 RT events from 8,100 fractions, and data on 2,216 patients were collected and identified by type and severity. Most incidents were non-related dosimetric events resulted from system malfunctions and shortcomings in treatment delivery procedures. The review discovered that 45 of the 164 incidents were related to tube sensor failures and source blockages. These delivery errors equated to 0.56 incidents per 100 insertions and 2.03 incidents per 100 patients, inconveniencing patients with treatment delays.
In the early years of our experience with VariSource afterloader system, we encountered several source loading problems. They were especially prevalent among cases using multichannel Miami-style applicators. We eventually solved this problem by loading iridium-192 radioisotope source in 6 implanted needles, thereby replacing the 6 loading tunnels in Miami applicator. Regarding source blockage from the tube sensor, the failure came from curving, a snaking effect, and an error in the parameter encoder for wire drift. The source drive mechanism of VariSource system uses a timing belt and a high-speed drive motor, with setup parameters for pulse encoder, step motor, and force acting. The experienced problems in our system were most often related to force acting, which was negatively affected by dirt in high-speed motor and working condition of the belt. These causes indicated that more frequent system maintenance was needed.
Our review identified that 4 patients received a minor dosimetric impact due to source blockage. However, most blockages resulted in treatment delay, patient inconvenience, and increased nursing care risks for some patients. Also, Provenzano et al. [36] reported that nearly half of their adverse events were caused by a device malfunction. It is worth noting that an optimal design of BT applicators and operating systems is crucial to sustaining safe and reliable treatments for BT patients.
As an indirect measure, low-rate of dosimetric errors demonstrated the efficacy of our safety program (0.14 per 100 BT insertions and 0.5 per 100 BT patients). Equipment performance, uncommon or complex procedures, and human factors (knowledge and skill sets, workloads, shift patterns, and workplace communications) were contributing factors. We conclude that our verification process effectively detected near-misses that could be immediately corrected. In addition, our knowledge management program proved to be a powerful learning tool to enhance patient safety, improve teamwork, streamline communications, and promote a culture of safety.
The chief limitation of this study is the voluntary nature of reporting, which could include inaccurate or incomplete information. Moreover, the events described in the present study occurred in only gynecological cancer patients receiving HDR-BT, with no incidents reported in a tiny group of prostate cancer patients.
Conclusions
The hospital’s incident reporting system was successful because it enabled data to be turned into learning points and facilitated the appropriate management of patients’ care process. Our report represents an opportunity to support the actions of implementing centers and related healthcare organizations to improve the quality of HDR-BT services and patient safety.