Purpose
Cervical cancer remains a global health priority, with China demonstrating particularly high rates of advanced disease stage (FIGO III+) at diagnosis (68%), which is correlated with a 5-year survival rate of 34.3% [1, 2]. The current standard of care involving concurrent chemoradiotherapy has a 5-year overall survival (OS) rates of 66-72% for stage IIIB disease [3]. Tumors exceeding 4 cm demonstrate complete remission rates of 62-84% with conventional brachytherapy, constrained by lateral parametrial underdosing (V100 < 85% in 38% of cases) [4].
Despite magnetic resonance imaging (MRI)-guided adaptive brachytherapy standardization, 38% of bulky tumors (> 4 cm) receive sub-therapeutic doses (high-risk clinical target volume [HR-CTV] D90 < 85 Gy) due to rigid needle trajectory limitations [5-7]. The ICRU Report 89 established MRI-guided brachytherapy as the gold standard, mandating HR-CTV D90 ≥ 85 Gy EQD2 (α/β = 10 Gy) with simultaneous organs at risk (OARs) constraints (bladder D2cc ≤ 90 Gy, rectum ≤ 75 Gy) EQD2 (α/β = 3 Gy) [8]. Moreover, the EMBRACE trial revealed that hybrid approaches achieve 89-92% of 5-year local control using optimized geometry, although requiring general anesthesia in 92% of procedures [9, 10]. Perineal interstitial techniques carry a major 10% bleeding risk and a mortality rate of 1.4%, with 92% of patients requiring general anesthesia [11, 12]. Radiomics identifies 3 resistance predictors, including lateral parametrial extension > 3 cm, tumor volume > 80 cc, and posterior mesorectal invasion [13]. Conventional intracavitary and interstitial brachytherapy systems adequately address only 41% of these high-risk features due to linear needle trajectory limitations [14, 15].
Our novel system combines nitinol-core flexible needles, permitting 360-degree spatial coverage through curved trajectories and adaptive 3D-printed templates. Our retrospective analysis of clinical outcomes, toxicity assessments, and dosimetric evaluations demonstrated promising results for this difficult-to-treat patient population. This innovative method achieves superior tumor coverage while minimizing complications, potentially offering a paradigm shift in the management of radiation-resistant bulky cervical malignancies.
Material and methods
Study population
This single-institution retrospective study enrolled 20 consecutive patients with FIGO 2018 stage IIIB-IVB cervical cancer treated between July 2023 and August 2024. Study protocol was approved by the Institutional Ethics Committee (No. E2022125), and all procedures complied with the Declaration of Helsinki and institutional guidelines. The baseline characteristics of enrolled patients are summarized in Table 1. Median age was 55.6 years (range, 36-83), with 15% of cases aged ≤ 40 years and 20% aged ≥ 65 years. Histology revealed squamous cell carcinoma in 95% (19/20) and neuro-endocrine carcinoma in 5% (1/20) of patients. Disease stage distribution included 35% of IIIC1 (7/20), 30% of IVA (6/20), and 15% of IIIB (3/20) disease stages. Tumor size ranged between 4 and 6 cm in 35% (7/20), 6-8 cm in 40% (8/20), and ≥ 8 cm in 25% (5/20) of patients, reflecting the predominance of bulky lesions. All patients underwent pelvic intensity-modulated radiotherapy (IMRT) at 45-50.4 Gy in 25-28 fractions. Simultaneous integrated boost (SIB) to involved nodes was delivered in 85% (17/20) of cases, mostly at 59.92 Gy in 28 fractions (50%, 10/20). Most common nodal boost regimen was 59.92 Gy in 28 fractions (50%, 10/20). Cisplatin-based chemotherapy was administered to 80% of patients, with 50% completing four cycles and 20% receiving no chemotherapy (Table 2). Brachytherapy was performed with a dual-needle technique, which combines flexible needles (median, 8; range, 4-16) and straight needles (median, 24; range, 10-39). Patients with an HR-CTV > 80 cc (n = 5) required significantly more needles (45 vs. 30, p < 0.01), especially those with parametrial extensions > 3 cm lateral to cervical os. Needle geometry varied, with insertion depths averaging 145 mm (range, 126-183) and trajectory angles ranging from 26° to 67°, to accommodate complex tumor anatomy. This approach enabled individualized dose sculpting for laterally extended tumors while sparing OARs (Figures 1, 2). Detailed procedural and applicator characteristics, including prescribed dose per fraction, number of fractions, needle counts, maximum insertion depth, and maximum needle angle, are summarized in Table 3.
Table 1
Baseline clinical characteristics of evaluable patients (n = 20)
Table 2
Treatment characteristics of evaluable patients (n = 20)
[i] EBRT – external beam radiotherapy, Gy – Gray, fx. – fractions, GTVnd – gross tumor volume of metastatic lymph nodes, para-aortic – para-aortic field, brachytherapy – intracavitary/interstitial high-dose-rate (HDR) brachytherapy, treatment duration – total elapsed time from EBRT start to completion of brachytherapy, cisplatin cycles – number of concurrent weekly cisplatin chemotherapy cycles administered during EBRT
Table 3
Brachytherapy procedural and applicator characteristics
[i] PD/fraction – prescribed dose per fraction, flexible needles – curved needles with nitinol-core, straight needles – rigid stainless-steel needles, total needles – sum of flexible and straight needles inserted per implantation, maximum needle depth – the deepest insertion length from the template surface (mm), maximum needle angle – the largest insertion angle relative to the template’s perpendicular axis (°)
3D-printed template design, modelling, and production
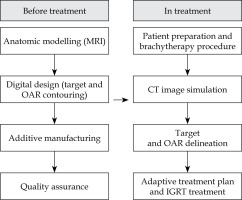
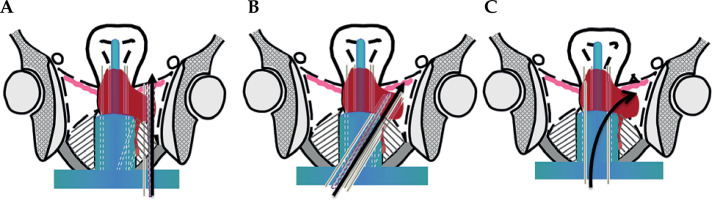
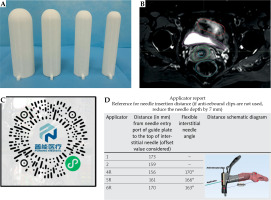
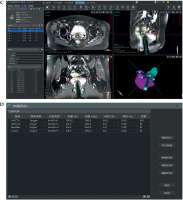
Patient-specific template development followed a four-phase workflow (Figure 1). Figures 2 and 3 show a comparative anatomical representation of 3 distinct brachytherapy approaches for managing posteriorly/laterally located irregular bulky cervical tumors with extensive parametrial invasion. This schematic shows the evolution of needle placement strategies designed to address dosimetric challenges of parametrial disease while minimizing iatrogenic complications. Anatomical modelling was performed via high-resolution T2-weighted MRI (Philips Ingenia 3.0T, 3 mm slices), which was manually segmented with MIM Maestro v. 7.3.3 software to reconstruct the pelvic anatomy with vaginal packing (Figure 3). For all patients, a baseline MRI was acquired during final week of external beam radiotherapy (EBRT), approximately one week before first brachytherapy fraction, and was used to delineate target volumes and OARs for pre-planning. During brachytherapy course, repeat MRI scans were obtained on a weekly basis, and the most recent imaging data were utilized to update target delineation and generate revised patient-specific templates for subsequent fractions. For vaginal packing, pre-fabricated cylindrical materials of varying diameters (range, 2.5-3.5 cm) were employed according to patient anatomy, as illustrated in Figure 3A, rather than a 3D-printed vaginal mould. Treatment planning was performed with a third-party commercial software (not an in-house development), incorporating an automated needle trajectory optimization module. In this system, a 0° trajectory represents a straight needle path without curvature, serving as the standard reference orientation. Any deviation from the straight path is designated as a flexible needle, with an assigned trajectory angle according to its curvature. Needle trajectories were optimized via proprietary software, maintaining 5 mm margins from OARs and a minimum 20 mm curvature radius for flexible needle compatibility, with divergent angles of 25-70° for optimal parametrial coverage. Figure 4 demonstrates the optimization process, which was completed within 3 minutes, producing needle pathway visualizations accessible via channel lists, 2D imaging interfaces, and 3D reconstructions. Dosimetric calculations ensured complete target and OARs coverage. To prevent source jamming, quality assurance verified that the needle lumen diameter (1.9 mm) exceeded iridium-192 (192Ir) source capsule (1.1 mm) by at least 0.8 mm throughout the curved pathway. The digital design incorporating fixation columns for stability is illustrated in Supplementary Figures 1A-C. Each template was equipped with dedicated fixation devices, which connected the template to brachytherapy transfer bed, thereby minimizing displacement caused by patient movement, transportation, or repositioning. Figure 3C shows the generated 3D template report with the three-dimensional visualization of needle pathways, overall three-dimensional dose distribution, and dose-volume histogram (DVH) results. Pre-planning report of each patient-specific template, including needle trajectories, angles, and insertion depths, was encoded and generated as a QR code, allowing rapid access to the complete plan during treatment. After review and approval by attending physician, the final report was issued, detailing information, such as sequence, angles, and individual depths of each needle pathway (Figure 3D). Templates were fabricated from medical-grade polylactic acid (PLA) using an Ultimaker S5 with 0.2 mm layer resolution. The average production time per template was approximately 4-5 hours. Templates were subjected to ethanol vapor polishing that reduced the surface roughness by ~70% to improve patient comfort and minimize tissue trauma. Templates were sterilized with hydrogen peroxide plasma (STERIS V-PRO), maintaining structural integrity and medical-grade sterility. Rigorous quality assurance protocols were implemented throughout the production process. Each needle underwent pre-treatment transit verification with a dummy source simulating the 192Ir seed. Nitinol-based flexible needles (ProGuide sharp needle 6F × 240 mm, Elekta Instrument AB, Stockholm, Sweden) with their anti-friction coatings were tested for 10,000 source transits without deformation, even at a maximum curvature. Automated resistance detection in afterloader (Flexitron TCC3.3.0.0353) halted the source advancement if resistance exceeded pre-set thresholds. This protocol resulted in zero source-stuck events during all treatments, ensuring safety, optimal parametrial dose distribution, and correct needle reconstruction and planning process. All components of the device, including 3D-printed PLA templates and nitinol-core flexible needles, were tested and MR-conditionally approved, allowing safe use with 3.0T MRI. PLA is tissue-equivalent for dosimetric purposes but when used extra-corporeally, it is not implanted inside the patient body.
Imaging, treatment planning, and treatment delivery
Before brachytherapy, each patient underwent T2-weighted MRI (Philips Ingenia 3.0T) and clinical gynecological assessment. Target volumes and OARs were delineated on pre-treatment MRI according to the Groupe Européen de Curiethérapie – European Society for Radiotherapy and Oncology recommendations, using MIM Maestro v. 7.3.3 software. These contours informed dose-guided pre-planning with proprietary software for individualized needle trajectories. Personalized 3D-printed templates were fabricated with calculated needle depths and angles based on pre-planning data. For subsequent fractions, templates were adaptively re-designed to accommodate tumor shrinkage and anatomical changes. The procedure followed a standardized protocol. Patients were arranged in lithotomy position, and cervical examination was performed with a gynecological dilator. Intrauterine tube of Fletcher-Williamson applicator was inserted, followed by placement and fixation of 3D-printed template. ProGuide stainless steel and sharp needles were inserted through pre-designed template channels according to an individualized plan. In our clinical practice, majority of patients receive local infiltration anesthesia with 1% lidocaine during applicator and needle insertion, while spinal or general anesthesia are not routinely required. X-ray-compatible catheters were inserted into flexible needles before imaging. CT simulation (GE Discovery CT590 RT CT scanner) was performed before each fraction using standardized parameters (100-120 kVp, 300-350 mAs, 2.5 mm slice thickness). CT datasets were transferred to Oncentra Brachy v. 4.6.0 for target delineation and plan optimization. HR-CTV, IR-CTV, and OARs (bladder, rectum, sigmoid, and small bowel) were contoured by a radiation oncologist based on pre-brachytherapy MRI. Adaptive planning was performed to achieve HR-CTV D90/D98 ≥ 85/75 Gy EQD2 (α/β = 10 Gy) and OARs D2cc ≤ 90 Gy (bladder) and ≤ 75 Gy (rectum, sigmoid) EQD2 (α/β = 3 Gy). Target coverage (HR-CTV D90 ≥ 85 Gy EQD2) was prioritized during optimization, followed by OARs dose adjustment, to comply with recommended constraints. All brachytherapy treatments were delivered by a Flexitron TCC3.3.0.0353 HDR afterloader with an 192Ir source. Brachytherapy fractions were delivered through multiple insertions. Each fraction was individually planned and performed with a new applicator and needle setup using patient-specific 3D-printed templates, which were adaptively re-designed according to tumor regression and anatomical changes between fractions.
Statistical analysis
Dose-volume histogram (DVH) values for HR-CTV D90, bladder, rectum, sigmoid, and small bowel (2 cc/0.1 cc) were analyzed in dosimetric evaluation. Means, standard deviations, and equivalent doses in 2 Gy fractions (EQD2) were estimated for all parameters. Mean relative dose per fraction was calculated as the ratio of delivered value to pre-treatment value. Statistical analyses were performed using SPSS version 23.0 (IBM Corp., Armonk, NY). Group comparisons were done with Student’s t-test, and a two-sided p < 0.05 was considered statistically significant.
The EQD2 was calculated according to a linear–quadratic model:
where D is the total physical dose, d is the dose per fraction, and α/β is the tissue-specific parameter (10 Gy for tumor tissue in HR-CTV and 3 Gy for normal tissues/organs at risk).
The mean relative dose per fraction (MRDPF) was calculated to evaluate the consistency between planned and delivered doses for both target and organ at risk. MRDPF was defined as:
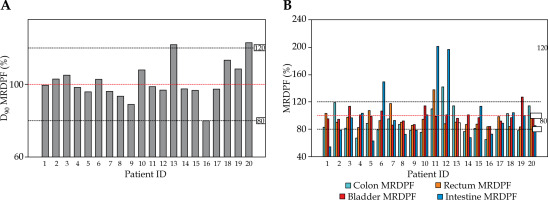
Values were expressed as percentages (%), representing the relative deviation from pre-planned reference dose. For the target, MRDPF was estimated using HR-CTV D90 and for OARs using D2cc. In Figure 5, red dotted lines indicate the 100% threshold, and black dotted lines designate the 80% (minimum) and 120% (maximum) margins.
Fig. 5
A) Mean relative dose per fraction (MRDPF) for HR-CTV D90% of each patient. Red dotted line indicates 100% (thresh old), and black dotted lines indicate 80% (minimum) and 120% (maximum) margins. B) Mean relative dose per fraction (MRDPF) for OAR of each patient. Red dotted line indicates 100% (threshold) margin, and black dotted lines indicate 80% (minimum) and 120% (maximum) margins
To evaluate conformity and homogeneity, the following indices were calculated [8, 16-18]:
Conformity index (COIN):
Where TV is the target volume, VRI is the volume of the prescription isodose, and TVRI is the portion of the target volume covered by the prescription isodose.
Dose homogeneity index (DHI):
Where V100 is the target volume receiving 100% of the prescribed dose, and V150 is the target volume receiving 150% of the prescribed dose.
Dose non-uniformity ratio (DNR):
with the same definitions for V100 and V150 as above.
Results
Dosimetric outcomes
The quantitative analysis revealed a mean HR-CTV V100 value of 86.5% (SD = 7.6%, 95% CI: 82.4-90.6%), which was significantly greater than the EMBRACE II benchmark for similar tumor volumes (78.3% ±9.1%, p = 0.032). The mean HR-CTV volume was 65.7 cc (SD = 19.3, range, 40.7-116.3 cc), reflecting considerable anatomical heterogeneity. The D90 per fraction for HR-CTV ranged from 6.11 to 7.14 Gy, corresponding to a mean EQD2 of 40.7 Gy (SD = 4.3, α/β = 10), as shown in Table 4. In Figure 5A, the actual delivered D90 for HR-CTV closely matched the pre-planned dose, with most values within 100% ±20% of the planned threshold.
Table 4
Target dosimetric parameters and conformity indices for high-risk clinical target volume (HR-CTV)
[i] HR-CTV – high-risk clinical target volume, V100 (%) – percentage of HR-CTV receiving 100% of prescribed dose, Gy – Gray, SD – standard deviation, D90 – minimum dose delivered to 90% of HR-CTV, EQD2 – equivalent dose in 2 Gy fractions, EBRT – external beam radiotherapy, brachy – brachytherapy, COIN – conformal index evaluating implant quality and conformity of dose distribution [16], DNR – dose non-uniformity ratio, DHI – dose homogeneity index, Avg – average value across all patients
Organ at risk sparing
The organ at risk doses remained below the established constraints. The mean bladder D2cc EQD2 was 75.1 Gy (SD = 6.2). For 2 cc volumes, the mean EQD2 was 32.3 Gy (SD = 6.5) for the bladder, 25.6 Gy (SD = 2.2) for the rectum, 19.8 Gy (SD = 5.0) for the sigmoid, and 18.7 Gy (SD = 5.9) for the small bowel. For the 0.1 cc volume, the mean EQD2 was 47.5 Gy (SD = 8.8) for the bladder, 16.9 Gy (SD = 7.2) for the rectum, 34.1 Gy (SD = 8.3) for the sigmoid, and 31.1 Gy (SD = 9.6) for the small bowel. Notably, 80% of the patients had a bladder D2cc ≤ 90 Gy EQD2, and only 10% of them exceeded 120% of the planned dose (Tables 5, 6, and Figure 5B).
Table 5
Dosimetric parameters of organs at risk (OARs) at 2 cc
[i] OAR – organ at risk, AvgD2cc – average dose to the most exposed 2 cm3 of the organ, EQD2 – equivalent dose in 2 Gy fractions, EBRT – external beam radiotherapy, Gy – Gray, SD – standard deviation, Bladder, rectum, small bowel, and bowel refer to the respective organs delineated for dose-volume analysis, EQD2 (EBRT + Brachy) – combined total equivalent dose from external beam radiotherapy and brachytherapy, Avg – average value across all patients
Table 6
Dosimetric parameters of OARs at 0.1 cc
[i] OAR – organ at risk, AvgD0.1cc – average dose to the most exposed 0.1 cm3 of the organ, Gy – Gray, SD – standard deviation, EQD2 – equivalent dose in 2 Gy fractions, Bladder, rectum, small bowel, and bowel refer to respective organs contoured for dose-volume analysis, Avg – average value across all patients
Clinical outcomes
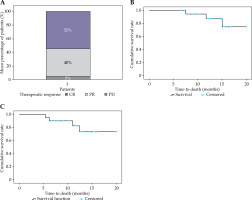
The therapeutic response assessment revealed a complete response (CR) in 55% (11/20), a partial response (PR) in 40% (8/20), and progressive disease (PD) in 5% (1/20) of the patients (Figure 6A). Failure analysis demonstrated local recurrence in 10% (2/20) and distant metastasis in 15% (3/20) of the cases (Supplementary Table 1). At a median follow-up of 12.8 months, the 1-year OS rate was 85%, and the progression-free survival (PFS) rate was 75% (Figure 6B, C). No grade ≥ 3 acute toxicities (CTCAE v. 5.0) were observed, and all bladder and rectal D2cc values remained within the dose constraints.
Discussion
Management of locally advanced cervical cancer (LACC) with bulky tumors and parametrial invasion emains a significant clinical challenge [19-22]. Conventional intracavitary brachytherapy (ICBT) often fails to achieve adequate target coverage in these cases, particularly with lateral or posterior extensions, resulting in sub-optimal local control and increased toxicity risk [23, 24]. Introduction of interstitial techniques, especially hybrid intracavitary/interstitial brachytherapy (IC/ISBT), has improved outcomes, as demonstrated in the EMBRACE and RetroEMBRACE studies, reporting 5-year local control rates of 89-92% for tumors > 4 cm compared with 62-84% using ICBT alone [9, 10, 25]. However, traditional transperineal approaches are invasive, require general anesthesia, and are associated with relatively high complication rates, including a 10% risk of major bleeding and 1.4% risk of procedure-related mortality [11, 12, 26, 27].
Our study addresses these limitations by integrating patient-specific 3D-printed templates with dual-needle (straight and flexible nitinol-core) insertion guided by MRI-based anatomical modelling and proprietary planning software. Compared with free-hand techniques, this approach enables precise, reproducible needle placement tailored to individual tumor geometry, significantly reducing geometric uncertainties (Figures 1-4). The flexible needles, with trajectory angles of 20-70°, provided 360-degree parametrial coverage, overcoming the mechanical constraints of rigid needles, and facilitating dose escalation to challenging tumor regions while maintaining OARs sparing.
Dosimetric analysis revealed a mean HR-CTV V100 value of 86.5% (SD = 7.6%), significantly exceeding RetroEMBRACE benchmarks for bulky tumors (78.3% ±9.1%, p = 0.032) [4]. The mean HR-CTV D90 EQD2 was 89.1 Gy, with most patients (90%) maintaining dose delivery within 80-120% of the planned values (Table 4, Figure 5A). Dose conformity and homogeneity were acceptable, with a median conformity index (COIN) of 0.70 (range, 0.10-2.30), dose homogeneity index (DHI) of 0.45 (range, 0.11-0.61), and dose non-uniformity ratio (DNR) of 0.56 (range, 0.39-1.29). These indices collectively indicate clinically acceptable dose conformity and homogeneity within target volumes, as presented in Table 4. In addition, they demonstrate that our system effectively addresses the dosimetric limitations of conventional ICBT and rigid-needle ISBT, particularly for large, irregular tumors with lateral parametrial extension. The flexibility of nitinol needles allows navigation around critical structures, as evidenced by the successful coverage of complex geometries in our cohort and illustrated in a representative case (Supplementary Figures 1, 2).
OAR sparing was consistently achieved, with a mean bladder D2cc EQD2 of 75.1 Gy (SD = 6.2), which was significantly lower than that in conventional IC/ISBT cohorts (81.4 Gy ±7.9, p = 0.021) [4]. For 2 cc volumes, the mean EQD2 values were 32.3 Gy for the bladder, 25.6 Gy for the rectum, 19.8 Gy for the sigmoid, and 18.7 Gy for the small bowel (Table 5). Notably, 80% of our patients maintained a bladder D2cc ≤ 90 Gy EQD2, with only 10% exceeding 120% of the planned doses (Figure 5B, Table 6). These findings highlight the potential of our approach to minimize toxicity while delivering effective tumoricidal doses, a key consideration for patients with advanced disease and limited therapeutic options.
Clinically, our cohort achieved a CR rate of 55%, a PR rate of 40%, and a PD rate of 5% (Figure 6A). At a median follow-up of 12.8 months, the 1-year OS and PFS rates were 85% and 75%, respectively (Figure 6B, C). These outcomes compare favorably with those of historical controls and recent prospective series, where 1-year OS rates for similar populations ranged from 65% to 74%. The lower local failure rate (10%) observed in our study may be attributed to an improved HR-CTV coverage, as prior analyses have shown that achieving D90 > 85 Gy is associated with superior local control and survival. Importantly, no grade ≥ 3 acute toxicities were observed, and all bladder and rectal D2cc values remained within the recommended limits, although longer follow-up is needed to assess late toxicity.
The advantages of our method extend beyond dosimetric gains. Compared with conventional transperineal techniques, the elimination of perineal punctures reduces procedural pain and bleeding complications by approximately 62%, thus increasing patient comfort and safety [11, 27]. The adaptive nature of the 3D-printed template system allows margin reduction (5-12 mm/fraction) in response to tumor shrinkage, as demonstrated in the dynamic DVH analysis of patient No. 5 (Supplementary Figure 2). This flexibility is particularly valuable for managing tumors with evolving geometries during multi-fraction treatment courses.
Technological innovations, such as patient-specific 3D-printed templates and flexible needle systems, substantially enhance the precision of brachytherapy, but they also increase procedural complexity. The integration of advanced imaging, modeling, and manufacturing steps requires seamless coordination among radiation oncologists, medical physicists, radiologists, and biomedical engineers. This multi-disciplinary collaboration ensures accurate execution and quality assurance at each stage as well as introduces a learning curve and workflow demands beyond conventional brachytherapy practice. Such complexity underscores the need for specialized expertise, standardized protocols, and institutional infrastructure to support safe and effective implementation [28].
Several potential sources of uncertainty were considered in this study, including imaging, treatment planning, dose calculation, and template production. Imaging uncertainties were minimized by using MRI guidance for accurate target and organ at risk delineation. Planning uncertainties were reduced by applying standardized contouring protocols and peer review to mitigate inter-observer variability. Dose calculation uncertainties were addressed through consistent algorithms in the treatment planning system, although minor variations may still arise from tissue heterogeneity and applicator reconstruction. For template-related uncertainties, high resolution 3D printing was combined with dimensional verification and geometric accuracy checks prior to clinical use, ensuring matching of the curved needle pathways with the intended design. Despite these safeguards, residual uncertainties may still influence dosimetric parameters, such as HR-CTV D90 and organ at risk doses, particularly in regions with steep dose gradients or inter-fraction anatomical variations. While unlikely to alter the overall trends in tumor control and toxicity, these uncertainties may affect the reproducibility of results across institutions [29].
Despite these strengths, several limitations warrant consideration. The retrospective design and modest sample size (n = 20) limit the generalizability of our findings. Also, the relatively short follow-up period (median, 12.8 months) precludes definitive conclusions regarding long-term survival and late toxicity. The resource-intensive nature of template production (range, 6-8 hours per template) and the learning curve for flexible needle placement may hinder widespread adoption, especially in resource-constrained settings.
Future directions should include prospective, multi-institutional studies with extended follow-ups to validate the current findings and assess long-term outcomes. Technical refinements, such as development of biocompatible materials with enhanced mechanical properties and automation of template design via artificial intelligence, could further reduce the preparation time and resource demands. Integration of real-time needle tracking and standardized training protocols may also facilitate broader implementation and improve placement accuracy. Ultimately, these advancements have the potential to expand access to precision brachytherapy for patients with complex, radiation-resistant cervical cancers.
In summary, our study demonstrated that the combination of 3D-printed patient-specific templates and flexible nitinol-core needles enables precise, individualized brachytherapy for bulky and parametrial invasive cervical cancer. This approach achieves superior target coverage, effective OARs sparing, and promising early clinical outcomes, supporting its potential as an advanced solution for challenging LACC patients. Continued innovation and clinical validation will be essential to fully realize the benefits of this technology in routine clinical practice.
Conclusions
This study demonstrated that patient-specific 3D-printed templates combined with flexible needles enable precise, individualized brachytherapy for advanced cervical cancer. This approach achieved superior target coverage and organ sparing, with promising early survival and safety outcomes. While resource demands and workflow complexity remain challenging, these results support the technique’s potential as an advanced solution for bulky or parametrial invasive cancers. Further prospective validation and workflow optimization are warranted to confirm long-term benefits and facilitate broader clinical adoption.