Purpose
Prostate cancer (CaP) is the second most common cancer and the fifth leading cause of death from cancer in men in the world [1]. Nowadays, many treatment options available are responsible for side effects, mainly urinary and sexual. During the announcement consultation when the patient is learning about his disease, sexual consequences are often undervalued by the patient who wants to eradicate his cancer. However, once the treatment is implemented, side effects are prominent and impair the quality of life. Life expectancy after curative treatment is more than ten years; therefore, the management of side effects is of paramount importance. Prostate brachytherapy (BT), with oncologically proven effectiveness [2,3], is one of the recommended treatment for patients suffering from low-risk CaP [4]. Erectile dysfunction (ED) after BT with or without external beam radiotherapy (EBRT) has been reported to vary from 19% to 80% [5]. Indeed, published studies on ED after BT are very heterogeneous: 1. Different inclusion criteria in populations: high-grade CaP, high-rate of preoperative hormonotherapy (HT), or preoperative ED; 2. Various methods with different BT techniques; and 3. Difference in the definition and assessment of ED, i.e., patient interview, physician-reported scores, many different questionnaires such as International Index of Erectile Function five item score (IIEF-5), IIEF-15, or Mount Sinai questionnaire [6,7,8,9,10]. In a recent review of Wortel et al. regarding the challenges in interpretation studies on ED after radiation therapy for CaP, only 4 studies reported on ED after exclusive low-dose-rate BT were identified, with 24-58% of patients completing the study at 3-5 years and only 3 patients using an IIEF questionnaire [5].
A recent interesting study has addressed the issue of the impact of radiation therapy (i.e., BT, EBRT, or both) on erectile function (EF) [11]. In their articles review, the authors included 103 studies assessing ED after: BT (n = 17,057 patients), EBRT (n = 8,166), or both (n = 1,046), and performed a meta-analysis and meta-regression. The meta-regression showed no difference for ED after BT or EBRT, reaching about 33% at 1 year and 50% at 5 years. However, 7 common instruments were used to assess EF with 23 different cutoffs to define ED. The meta-analysis was conducted only on 21 studies that used the IIEF-5, also known as SHIM-questionnaire (sexual health inventory for men), it did not stratify according to RT modality (i.e., EBRT vs. BT), and the pooled analysis used the SHIM/IIEF-5 ED cutoff varying from 10 to 17.
In this context, we aimed to report the prospective evaluation of EF over 4 years after BT in patients with low-risk CaP, using the validated self-administrated questionnaire IIEF-5 in a well-defined EF population at baseline (IIEF-5 > 16) and using the same BT technique.
Material and methods
This study was a mono-centric retrospective cohort with prospective assessment. From February 2007 to July 2012, 179 patients with localized CaP were treated with BT. Inclusion criteria were the BT eligibility criteria [4]: 1. Low-risk CaP or intermediate favorable CaP according to D’Amico classification, i.e., Gleason score < 7 with PSA < 10 ng/ml and < T2b, or only one of the following criteria: PSA 10-15 ng/ml or T2b or Gleason 7 (3 + 4); 2. Less than half of positive biopsies; 3. Initial prostate volume < 50 cc or 80 cc decreased to 50 cc after 3 months preoperative HT; 4. IIEF-5 > 16 preoperatively without drug assistance. The exclusion criteria were: 1. Preoperative IIEF-5 ≤ 16; 2. History of pelvic EBRT; 3. Intaking of phosphodiesterase inhibitor 5 (PDE5i) before treatment of CaP. Patients were not randomized; they have selected their treatment after clear, fair, and appropriate information.
Of the 179 patients treated by BT, 163 completed the initial IIEF-5 questionnaire and among these patients, 103 had IIEF-5 > 16; 1 patient was excluded because of a preoperative PDE5i treatment. Therefore, 102 patients with localized CaP and having an initial IIEF-5 score > 16 were included. All patients were sexually active at baseline. Population characteristics are summarized in Table 1. No patient received EBRT and 12.7% (n = 13) had 3 months preoperative HT because of prostate volume > 50 cm3. No cancer recurrence occurred during follow-up.
Table 1
Population characteristics
During the study, PDE5i was introduced after BT at the request of the patient or proposed by the medical team in case of IIEF-5 < 16 at a follow-up visit (sildenafil 100 mg, tadalafil on demand 20 mg or 5 mg daily). No patient was treated with intra-cavernous injection, penile implant, or vacuum. IIEF-5 for patients using PDE5i corresponded to the score under treatment. Preoperative HT and PDE5i after BT were not considered as exclusion criteria.
All BT were performed by the same experienced team included a urologist, a radiotherapist, and a physicist. The technique of Iodine-125 (125I) loose seeds, as described by Stone and Stock, was already published in our previous studies, and was carried out with a prescribed dose of 160 Gy [12,13]. The BT modalities are presented in Table 2.
Table 2
Modalities for prostate brachytherapy
[i] D90 – dose received by 90% of prostatic volume, V100 – prostatic volume receiving 100% of the prescribed dose of 160 Gy, D2 cc – maximal dose received by 2 cc of the rectal volume, D0.1 cc – maximal dose received by 0.1 cc of the rectal volume, R100 – dose received by 100% of the rectal volume, D10 and D30 – dose received by 10% and 30%, respectively, of the urethral volume
Erectile function was prospectively assessed by a validated self-administered questionnaire IIEF-5. This questionnaire is the simplified version of 5 items of the IIEF questionnaire, which was developed by Rosen in 1996 [14]. It is recommended as a criterion of effectiveness in clinical studies on ED. It enables to classify patients into 5 classes such as: normal EF: > 21; mild ED: 17-21; mild to moderate ED: 12-16; moderate ED: 8-11; severe ED: < 8. A score below 5 is unevaluable. This questionnaire was completed by all patients included in our research before BT (or before HT) and annually for 4 years. An informed consent was obtained from every patient participating in the study. The primary endpoint was the IIEF-5 score. We used the Mann-Whitney test and the Chi-square test to analyze statistical difference between the years of follow-up.
Results
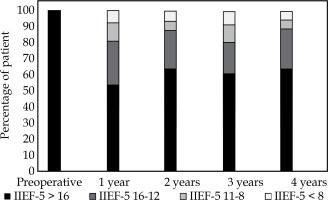
A population of 102 patients was analyzed. No CaP recurrence and death from CaP had occurred, and no patient received adjuvant oncological therapy during follow-up. One patient died at 18 months of follow-up from other cause. At 1 year after BT (Figure 1, Table 3), 98 subjects were analyzed. Among them, 8 were unevaluable (with an IIEF-5 score < 5) due to not having sex for other reasons than ED: 2 because of wife refusal, 1 because of painful ejaculation, 1 because of recent heart failure, and 4 because of no sex partner. Moreover, 18 were treated by PDE5i on demand. Of the 90 evaluable patients, 49 (54%) preserved an IIEF-5 > 16, enabling patient to have sexual relationship, and 73 (81%) had IIEF-5 above the score of 12. Moreover, only 7 (8%) presented a severe ED (score < 8). The average IIEF-5 score was 17 and lost 4 points during the first year.
Table 3
Evolution of erectile function after brachytherapy over 4 years
During the 2nd, 3rd, and 4th year, the proportion of patients maintaining an IIEF-5 score > 16 was not statistically different from that observed at the first year (p = 0.94; range, 60-64%). There was no statistical difference between the IIEF-5 means during follow-up (17-18). We did not observe any significant difference in the proportion of patients treated by PDE5i (18-21%; p = 0.85). At 2 years, 11 patients were lost to follow-up, one patient died (from other reason than CaP), and 10 were not evaluated because of 2 divorces and 8 without sex partner. At 3 years, 12 were lost and 14 were unevaluable because of 4 divorces, 2 wife refusal, and 7 no sexually active, who refused to talk about their sexual life or pretended to no longer be interested in the sexual aspect of life. At 4 years, 24 were lost and 12 were unevaluable with the same previous reasons.
At least a minimum of 54% and 80% of patients maintained a score > 16 and ≥ 12, respectively. Less than 10% suffered from severe ED during the 4 years post-BT, and the proportions of subjects belonging to different IIEF-5 groups remained stable over the 4 years following BT.
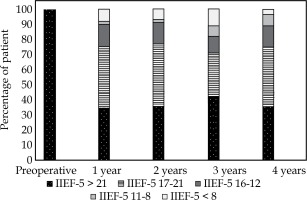
A subgroup analysis was performed on patients with preoperative IIEF-5 > 21 (Figure 2, Table 4): 6 patients were lost to follow-up at 4 years and 2 were unevaluable. At 1 year, 52 patients were analyzed (100%) and 4 had an unevaluable score (2 because of wife refusal and 2 no sex partner). Of the 48 evaluable patients with a preoperative IIEF-5 score > 21, 35% preserved a score above 21, and 75% preserved a score > 16. Nine patients were treated by PDE5i and the mean IIEF-5 score was 18. Those results were stable over 4 years, with a minimum rate of 35% keeping a normal EF and a minimum rate of 70% keeping a normal EF or mild ED (IIEF-5 > 16).
Table 4
Evolution of erectile function over 4 years after brachytherapy for patients with preoperative IIEF-5 > 21
Discussion
In this study with a longitudinal evaluation, we included a homogenous population (102 patients) treated with the same BT technique (one team, no external radiation, loose 125I seeds). We used a validated self-administrated questionnaire for ED. All patients had IIEF-5 > 16 at baseline and none had preoperative PDE5i treatment. We found that the proportions of subjects classified in the different IIEF-5 groups remained stable over 4 years following BT, and 63% maintained an IIEF-5 > 16 at 4 years. Less than 10% of patients were suffering from severe ED. Moreover, in the preoperative normal EF group (IIEF-5 > 21), about 70% of patients preserved a score > 16 and 35% had a normal EF.
There are few studies available on EF after exclusive BT, and most of them are retrospective. Additionally, they are very heterogeneous, which makes it difficult to compare. Indeed, the populations are not comparable; some studies included patients with high-grade CaP non eligible for BT treatment [7,10,15], and others included patients with preoperative ED [16,17] or high percentage of preoperative HT [7]. The methods of treatment were also heterogeneous, with different BT techniques [15,18], dissimilar questionnaires validated or not [17], and adjuvant treatment sometimes not mentioned. In 2011, Bannowsky et al. [19] published a study on EF after low-dose-rate BT for localized CaP and found a significant loss of EF in the third year highlighting a delayed effect on EF. However, this study included only 32 patients and no ED treatment after BT was done. Chen et al. described stable sexual scores over 2 years post-BT, but they only used the sexual domain of the prostate cancer symptom index score and analyzed only 78 BT patients at 2 years [20]. The Mont Sinai team showed stable results of EF over 5 and 10 years in a large cohort of patients (including different BT technique and various assessment of EF) [15].
We used the IIEF-5, which is a validated, simple, and reproducible questionnaire, recommended as the best tool to assess ED after radiation therapy for CaP [5]. This questionnaire has been already employed in other studies, but with several inclusion bias such as preoperative ED patients not excluded [16], low number of patients [19], or IIEF-5 not used for all patients [10]. For our study, we selected patients with an IIEF-5 score > 16 to avoid a bias due to preoperative ED. A cutoff score of 16 for inclusion of patients and their analysis allowed to obtain a representative sample of the general population and an analysis applicable to everyday practice. In 1998, Rhoden et al. [21] used this score to assess the prevalence of ED in 965 Brazilian patients aged 40-90 participating in a CaP screening program. In this study, in the 40-49 years old age group (most unaffected by ED), only 64% of men had a strictly normal EF (IIEF-5 > 21) and 91% had a score > 16. In the 60-69 years old age group, only 42% had a strictly normal EF, while 65% had a score >16. We chose a score above 16 for inclusion, because the mean age of our population was 65 years and, at this age, only 42% of men have normal EF and 65% have normal or mild ED [21]. Considering assessment of the results, we also voluntarily chose not to exclude patients under PDE5 inhibitors after BT mainly because it can be considered as a mini-invasive treatment and it’s a real-life practice. Moreover, our study fulfilled all the requirements recommended by experts for reporting on erectile function outcomes after radiation therapy for CaP, i.e., methods of patients selection, patients and treatment characteristics, the use and duration of HT, baseline sexual function, the use of validated questionnaire, follow-up ≥ 24-36 months, definition and proportion of erectile dysfunction, and the use of erectile aids [5].
A proportion of 54%, 64%, 60%, and 63% of patients preserved an IIEF-5 > 16 at 1, 2, 3, and 4 years, respectively, with no statistical difference between years (p = 0.94). Those results were stable over years as observed by Putora et al. (3 years stability of EF after BT) [16]. In the same way, Ong et al. published in 2014 an interesting study on long-term evolution of EF after prostate BT, using the IIEF-5 questionnaire [22]. They reported 59% of EF preservation at 5 years (median follow-up of 41 months), defined by an IIEF-5 score > 16. EF was stable from the first year post-BT. Our study found similar results, but only 18% of our patients needed PDE5i (40% in the study by Ong et al.), and we added the subgroup analysis of preoperative IIEF-5 > 21 patients. Moreover, patients treated with PDE5i before BT were not excluded in the Ong et al. study (8% had PDE5i before BT), and only 44% and 33% of the entire cohort was assessed at 4 and 5 years, respectively [20].
In our study, a minimum of 80% of the population presented IIEF-5 score ≥ 12. Delaunay et al. [23] published a study with 270 patients at 3 years after BT on EF and quality of orgasm after the treatment [23]. They observed that patients with an IIEF-5 > 12 had quite frequently intense orgasm than those with a score < 12 (26.7% vs. 2.7%; p < 0.001). Langley et al. found a proportion of 72% of patients who preserved potency (IIEF-5 > 11) at 5 years [24]. We also noticed that patients with a score of < 8 represented less than 10% of patients and among these patients, half of them refused PDE5i treatment and refused to talk about their sexual life or pretended to no longer be interested in the sexual aspect of life.
Some studies on EF after BT also sought to identify other ED risk factors in their population. Merrick et al. identified preoperative IIEF score as predictive factor of ED in a multivariate analysis, and Matsushima et al. identified age ≥ 70 years as ED risk factor [9,18]. We performed, at one year follow-up of our study, a multivariate analysis on ED risk factors, which was already the subject of another publication and was not the focus of this study [25].
We then conducted a multivariate analysis on predictive factors of ED with IIEF-5 ≤ 16 by logistic regression. The analyzed risk factors included: preoperative IIEF-5 score, age, cardiovascular disease, diabetes, preoperative HT, intake of PDE5i, and preoperative prostate volume. Preoperative IIEF-5 score ≤ 21 was the only independent predictive factor of ED identified in our cohort, with an odd ratio of 6.5 [IC 95%: 2.4-17.4 (p = 0.0002)]. None of the other analyzed parameter was identified as ED independent risk factor.
A limitation of our study may be, as observed in most studies on this topic, the proportion of patients lost to follow-up, i.e., 4% at 1 year, about 11% at 2-3 years, and 23.5% at 4 years. Additionally, in the study of Ong et al. using a large population with IIEF-5 > 16 at baseline, only 58%, 44%, and 33% of patients were assessed at 3, 4, and 5 years, respectively [22]. Furthermore, the lost to follow-up we observed in our study was lower in the subgroup of patients with normal baseline IIEF-5, i.e., 0-11.5%. The second limitation is the relatively small size of study group, however, homogenous at different points of view, as mentioned above. We also used a questionnaire that focuses only on EF and does not explore other aspects of human sexuality. A prospective multicenter study using a multimodal questionnaire on sexual life would be interesting to perform in order to have a wider assessment of sexuality in this population.
Conclusions
Studies on side effects of CaP treatments are crucial to guarantee a loyal and clear preoperative information and a choice of better treatment option according to patient’s request. In our cohort, EF in patients with low-risk cancer treated by BT remained stable during the first four post-operative years, and severe ED affected less than 10% of patients. This suggests that BT could be one of the primary options to be considered for patients with priority to preserve their EF. This hypothesis should be studied with a prospective multicentric design with assessing the impact of BT over other aspects of sexual life.