Purpose
Immunotherapy rapidly became an integral part of multimodal treatment concepts in modern cancer therapy and its implementation is constantly evolving [1]. Immunotherapy can be defined as treatment approach that influences and enhances anti-tumor immune response. Immune checkpoint inhibitors that intervene in inhibitory signaling pathways on immune cells [cytotoxic T-lymphocyte-associated protein 4 (CTLA-4) and programmed cell death 1/ligand (PD-1/PD-L1)], have been approved and are frequently used to date [2]. New concepts for combinatorial therapies using immune checkpoint inhibitors are emerging, especially approaches combining checkpoint inhibitors and radiotherapy. In this context, PACIFIC trial, in which the immune checkpoint inhibitor durvalumab was used as an effective and well-tolerated consolidation therapy following chemoradiotherapy in patients with primarily unresectable non-small-cell lung carcinoma (NSCLC), showed promising oncological outcomes [3]. Currently, immune checkpoint inhibitors are routinely used in patients with melanoma, urothelial carcinoma, and head and neck tumors [4-7], while a large number of studies are examining application of checkpoint inhibitors in other entities. Besides immune checkpoint inhibitors and CAR-T cells, which have already been successfully established, there are several other immuno-therapeutic approaches, including OX-40, CDX-301, or Toll-like receptor (TLR) agonists as well as therapeutic vaccines, which differ considerably in their mechanism of action and biological behavior; they have not yet reached regular clinical use and are only available in experimental studies [8].
Despite promising pre-clinical results, a durable tumor response under immunotherapy is not observed in all patients and can rarely be predicted [9]. Nevertheless, a number of biomarkers exist to predict the therapeutic response to immune checkpoint inhibitors. Besides the exact stratification of suitable patients, a combination with radiotherapy can be a potential approach to improve the efficacy of immunotherapies, and lead to synergistic effects of both modalities. This synergism is based on immunomodulatory effects of radiotherapy [10]. The majority of pre-clinical data investigating the synergistic effect of immunotherapy in combination with radiotherapy have used external beam radiotherapy (EBRT). As radiation-induced immune response seems to be influenced by timing, dose, target volume, and fractionation of radiotherapy, brachytherapy may be advantageous over EBRT in this context [11-13]. Brachytherapy enables highly conformal dose distribution by providing high, but also heterogeneous intra-tumoral radiation dose. This heterogeneity depends on the distance of the source from the target volume. Then, different dose levels lead to various effects; while lower doses lead to a temporary depletion of tumor-infiltrating lymphocytes (TILS), cytokine release, and immune cell infiltration, high doses result in direct or indirect cell death and finally to the release of tumor antigens [14]. Therefore, the combination of brachy- and immunotherapy seems to offer an additional interesting possibility to improve treatment of patients in both definitive and palliative settings. In this article, we focused on these possibilities and reviewed the current status and future perspectives of combining brachy- and immunotherapy.
Current status and approaches in targeting immune response
Immuno-therapeutic agents
Immune checkpoint inhibitors are blocking crucial inhibitory signaling pathways, which are controlling the activation of T cells, and thereby promote anti-tumor effects. Since the first approval of pembrolizumab in 2014, five additional monoclonal antibodies targeting PD-1 or its ligand PD-L1 have been permitted by the Food and Drug Administration/European Medicines Agency (FDA/EMA). Meanwhile, the number of active clinical trials has increased rapidly. The majority of this expanding landscape of clinical trials includes combinations of anti-PD-1/PD-L1 agents with other cancer therapies. In 2019, there was a total of 2,975 active trials, of which 2,251 investigated a combination of anti-PD-1/PD-L1 with other modalities. Most of these studies examined a combination of anti-PD-1/PD-L1 with anti-CTLA4 agents, followed by a combination with chemotherapy and anti-angiogenic agents targeting the vascular endothelial growth factor receptor (VEGFR) axis [15].
In addition to immune checkpoint inhibitors, there are numerous other approaches to influence and enhance anti-tumor immune response. For example, T cell stimulating immuno-therapeutic agents, such as OX-40 agonists, genetically modified T cells (CAR-T cells), and bi-specific antibodies as well as vaccination approaches against tumor-specific or tumor-associated antigens. Furthermore, the tumor micro-environment can be influenced by immuno-stimulating cytokines, transforming growth factor β (TGF-β) blockade, or TLR agonists [16].
Immuno-genetic effects of radiotherapy/brachytherapy
For local tumor irradiation, immuno-stimulatory and immuno-suppressive effects have been reported [10].
Pro-immunogenic properties of radiotherapy are described by aspects, such as the abscopal effect (tumor regression in non-irradiated lesions) or the in situ vaccination (enhancing immunogenicity of an immuno-suppressive tumor micro-environment), including both local and systemic immune responses [17, 18]. According to systemic immune response, cell death of tumor cells triggered by irradiation has immuno-genic properties [19]. Tumor-specific antigens and danger signals, with inflammatory cytokines and damage-associated molecular patterns are released, causing activation of innate and subsequently, the adaptive immune system. MHC-1 molecules in the tumor micro-environment are up-regulated, improving the “visibility” of tumor cells for the immune system [20]. In addition, radiotherapy can induce a pro-angiogenic shift [21]. Changes in the endothelium of the tumor blood vessels caused by irradiation, lead to an adhesion of circulating immune cells and facilitate the immigration of effector cells into tumor tissue [22]. In particular, induction of SDF1 leads to infiltration of myeloid cells and TIE2-expressing monocytes, resulting in increased angiogenesis. These immuno-suppressive and pro-angiogenic tumor-associated macrophages could promote tumor recurrence and diminish therapeutic benefit [23-25]. Especially hypofractionated radiotherapy can alter the extracellular matrix and vascular permeability by promoting reconstitution of the tumor vasculature. As already mentioned, this may improve immune cell infiltration and reduce tumor hypoxia. Apart from that, high-dose radiotherapy, such as stereotactic body radiation therapy (SBRT), is considered to induce severe vascular damage, leading to indirect tumor cell death [26-28]. Effected by these phenotypic changes, T cell response is further enhanced by the maturation of dendritic cells and antigen-cross-presentation [29]. Even though in vitro experiments and pre- and post-therapeutic patients’ samples have shown that the infiltration of anti-tumor immune effector cells significantly increased in irradiated tumors as compared to non-irradiated tumors, irradiation leads to a temporary depletion of radiation-sensitive immune lineages, including CD4- and CD8-positive lymphocytes, which can result in long-term lymphopenia [30]. Moreover, irradiation leads to an upregulation of PD-L1 in the tumor tissue and can stimulate the secretion of various pro-inflammatory cytokines and chemokines, such as TGF-β, chemokine CC motif ligand 2 (CCL2), colony-stimulating factor 1 (CSF-1), CXC motif chemokine ligand 12 (CXCL12), or insulin-like growth factor 1 (IRF1) [31-33].
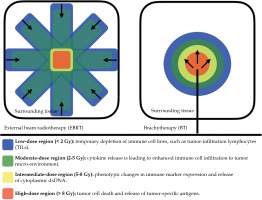
In general, higher radiation doses seem to have more immune-stimulating effects, while lower radiation doses (< 2 Gy) may present with more immuno-suppressive and anti-inflammatory effects [34]. Therefore, brachytherapy may have potential advantages over EBRT in achieving tumor immuno-modulation. While EBRT passes through considerably volumes of healthy tissue to reach targeted tumor volumes, brachytherapy enables a highly conformal dose distribution by providing a high, but also heterogeneous intra-tumoral radiation dose (Figure 1). This heterogeneity depends on the distance of the source from the target volume [14]. Patel et al. assumed that highly conformal dose distribution enables sparing of normal tissue, especially circulating immune components and lymphatic tissue, which minimizes off-target effects. Instead, in the target itself, a dose gradient is created according to linear quadratic formulation depending on the distance to the source. This causes multiple immuno-genic mechanisms, ranging from immuno-genic cell death and the release of tumor-specific antigens in regions of the highest dose, to an increased release of cytokines in the medium-dose area, and finally, to a short-term depletion of suppressive tumor-infiltrating lymphocytes in surrounding tumor tissues [34].
Combination of immunotherapy agents and radiotherapy/brachytherapy
There is ample evidence for important interplay between radiotherapy and the immune system [35]. Reports on abscopal effects (tumor regression in non-irradiated lesions) in patients with metastatic tumors receiving immune checkpoint inhibitors and undergoing local radiotherapy, are supporting the thesis of immune-modulatory effect of irradiation [36-38].
For example, cytokines are a diverse family of signaling glycoproteins of the immune system, which enable the immune cells to communicate with each other and influence the surrounding micro-environment in autocrine, endocrine, and paracrine manners. Such a communication plays an important role in the regulation and control of immune reactions. In that way, cytokines can influence the activity of immune cells and regulate their maturation and reproduction. In a pre-clinical murine tumor model, intra-tumoral delivery of cytokines seems to enhance the in situ vaccination effect of EBRT. In combination with anti-CTLA4 agents, further synergistic effects could be achieved [39]. Moreover, subcutaneous injections of granulocyte-macrophage colony-stimulating factor (GM-CFS) in combination with EBRT (35 Gy in 10 fractions) in 26.8% of 41 patients with metastatic solid tumors, have generated abscopal tumor response [38]. However, the anti-tumor effect of cytokines in combination with radiotherapy could not fulfill the initially high expectations, especially the increased toxicity has been a problem after systemic administration of these agents [40]. At present, only a few cytokines are used for a limited number of oncologic indications and in a small number of early phase clinical trials, in which interleukin 2 (IL-2), interferon α (IFN-α), GM-CSF, and tumor necrosis factor α (TNF-α) are analyzed as a combinative treatment with radiotherapy. Others investigate intra-tumoral administration of cytokines, decreased toxicity, and relevant efficacy.
Irradiation induces an up-regulation of PD-L1 in tumor tissue and the secretion of various pross-inflammatory cytokines and chemokines, such as TGF-β. These facts are important arguments for combining radiation with immuno-therapeutic agents, including PD-1/PD-L1 blockade and TGF-β blockers, as in this way the pro-immunogenic properties of radiation can be exploited while blocking further immunosuppression [41]. A variety of ongoing clinical trials are currently investigating the efficacy of SBRT combined with PD-1/PD-L1 blockade in patients with NSCLC, and this combination has shown promising results and good response rates in pre-clinical and clinical studies, especially in metastatic disease stages [42]. In advanced stage non-small cell lung cancer, for example, a recent, single-center unexpected post-hoc analysis of KEYNOTE-001 trial [35] demonstrated a benefit in terms of progression-free survival (PFS) and overall survival (OS) in patients with metastatic NSCLC who received pembrolizumab and had previously underwent radiotherapy, compared to those who had not underwent radiotherapy, thereby overcoming the effect of PD-L1 expression in multivariate analysis [43]. Similarly, a single-center cohort study of patients with metastatic lung cancer receiving PD-1 or PD-L1 inhibitors found that there was a trend for longer overall survival in patients with prior (or concurrent) thoracic radiotherapy than patients without radiotherapy [44]. The PEMBRO-RT, a randomized, multi-center phase 2 clinical trial, reported a two-fold increase in the proportion of patients with metastatic NSCLC who achieved an objective response after PD-1 blockade and SBRT of previously non-irradiated NSCLC lesions. There was no increased toxicity compared with PD-1 blockade alone. Despite the augmenting effect of SBRT on PD-1 blockade response, no meaningful clinical benefit was observed. However, in a sub-group of PD-L1-negative patients, a significant benefit was observed [45]. Nevertheless, these data indicate that in oligometastatic settings, combinatorial therapies improve therapeutic response rates and may only cause a low-rate of acute and late toxicities [46]. Toxicities from immunotherapies are enormously diverse, but a recent meta-analysis was able to show that the combination of immune checkpoint inhibitors and radiotherapy has a comparable toxicity to immune checkpoint inhibitors alone. Accordingly, the combination of these two modalities did not show to significantly increase toxicity [47]. However, a sequential administration of multiple therapeutic modalities could be favorable when a tri-modal therapy approach is intended. In particular, a combination of chemoradiotherapy and immunotherapy could have a considerably rate of cumulative toxicities, so that a sequential administration could be advantageous over a simultaneous administration [48].
While most of these studies included EBRT, there is only a small number of trials including brachytherapy, reflecting the predominant role of EBRT [34] (Table 1). Nevertheless, as described above, brachytherapy may have the potential to influence the immune system in a positive manner.
Table 1
Overview of active clinical trials investigating the combination of brachytherapy and immunotherapies. The initial search was performed on clinicaltrials.gov. In total, 17 trials were identified, while only nine trials were active in October 2020
Pre-clinical approaches seem to support this consideration. In a carcinoembryonic antigen-positive (CEA+) mouse model, a CEA-directed vaccine was used in a combination with brachytherapy by implanting iodine-125 (125I) seed into the primary tumor site. The combination of both modalities led to a significant reduction of metastatic burden in the lungs, while neither vaccination nor brachytherapy alone could cause a similar effect [49]. In another pre-clinical report, Rodriguez-Ruiz et al. combined brachytherapy with anti-PD-1 and CD137 antibodies, inducing a cross-priming in a mouse model. Only the triplet of brachytherapy and anti-PD-1 and CD137 antibodies generated a response in a contralateral and therefore, non-irradiated tumor lesion, suggesting an abscopal regression [50]. In addition, pre-clinical data shows an elevated expression of PD-L1 in a time interval after high-dose brachytherapy, demonstrating immuno-modulatory effect of this treatment modality and suggesting the use of PD-1/PD-L1 checkpoint blockade [51]. In a clinical study, with the objective to assess effectiveness of diffusing alpha emitters radiation therapy (DaRT), a spontaneous regression of untreated distant lesions was observed in a patient with multiple SCC lesions after treatment of a single-lesion with DaRT and with no immuno-therapeutic agent. This observation provides new evidence for an abscopal effect after local treatment with an alpha emitter, and suggests an activation of immune system mediated by radiation [52]. Finally, Suzuki et al. have reported the first abscopal effect after high-dose-rate brachytherapy in 2019. After a combination of interstitial high-dose-rate brachytherapy and nivolumab, a patient with a renal cell carcinoma metastasis showed a response and thus, reduction of tumor burden at non-irradiated sites. Nivolumab was given before and after the application of 35 Gy in five fractions [53]. However, the treatment response of non-irradiated lesions could also be considered a response to immunotherapy alone.
Optimizing the efficacy of radioimmunotherapy
There is a major interest in finding and developing a rational combination of radiotherapy and immunotherapy, concerning simultaneous or sequential use, dose, and fractionation as well as other variables, in order to improve synergistic effects, enhance response rates, and overcome resistances.
The appropriate timing and sequencing of radiotherapy and immunotherapy is one of the fundamental issues, i.e., understanding the dynamics and changes of the innate and adaptive immune systems in response to radiotherapy, which seems to be crucial in finding the right timing and optimal sequencing for the administration of immunotherapies. While concurrent administration of radio- and immunotherapy may have yielded better results, the concerns about cumulative toxic effects have led to a sequential administration, with the assumption of blocking checkpoints before initiating radiotherapy and turning immunologically ‘cold’ tumors into ‘hot’ tumors [54]. Substantial, conceptual, and technical improvements, including intensity-modulated radiotherapy (IMRT), image-guided radiotherapy (IGRT), stereotactic radiotherapy, and image-guided brachytherapy, have allowed precise delivery of high doses of radiotherapy, and reduce collateral damage at organs at risk and lymph nodes [55, 56]. In this context, brachytherapy could provide a significant advantage. Due to highly conformal dose distribution, resulting in sparing of the draining lymph nodes, there is no influence on the immune-specific T cells at the site of antigen presentation [57]. Regulatory T cells (Tregs), which create an immuno-suppressive environment and therefore, impair an effective anti-tumor immune response, are rather less sensitive to radiation than other T cell populations [58]. Comparing different fractionation schemes, there is an evidence that intermediate doses of about 7.50 Gy per fraction lead to a significant depletion of Tregs and thus superior tumor control compared to lower doses or very-high single radiation doses [59].
Fractionated radiotherapy can induce a robust up-regulation of checkpoint molecules. It is most likely that radiation dose and fractionation influence this effect [41]. Hypofractionated radiotherapy has distinct effects on the tumor micro-environment and tumor cell phenotype by promoting immune cell recruitment through released antigens, pro-inflammatory molecules, and danger signals as well as MHC class I expression and other immunogenic molecules, fostering the anti-tumor immune responses [60, 61]. Immunogenic cell death and the release of danger signals is the basis of in-situ vaccination hypothesis, which is a part of the immune-stimulatory potential of radiotherapy [62]. As a result of cell death induced by radiotherapy, tumor antigens and debris are released into the tumor micro-environment. Next, antigen-presenting cells, like dendritic cells, are recruited and transported to lymph nodes, where the antigens are presented. However, antigen presentation and T cell interaction are subjected to circadian fluxes, and alterations, accompanied by an increased T cell infiltration and PD-L1 expression, create an immuno-suppressive matrix. A concurrent administration of anti-PD-1 or anti-PD-L1 agents could highjack these mechanisms and may help to achieve optimal therapeutic efficacy [63]. In this regard, an increased infiltration of innate and adaptive immune cells into solid colorectal tumors was observed after hypofractionated radiotherapy. In particular, hypofractionated radiotherapy resulted in infiltration and activation of dendritic cells (MHC-II+), but also in increased infiltration of macrophages (CD11bhigh/F4-80+) into the tumor micro-environment. Immune cell infiltration is temporal and occurs only between day 5 and 10 after the first irradiation [64]. Increased expression of MHC class I and II is associated with improved disease control in this context [65].
Similar results have been reported for infiltration of tumor-infiltrating lymphocytes (TILs) between day 5 and 8 after hypofractionated radiotherapy. Because lymphocytes are a radiation-sensitive cell population, shortened hypofractionated regimens with biologically equivalent doses could be beneficial. These data support performing hypofractionated radiotherapy before PD-L1 blockade [66].
In a TS/A (metastasizing mammary mouse adenocarcinoma cell line) breast cancer and MCA38 colon cancer mouse model, tumor control was demonstrated after both ablative single-dose radiotherapy and fractionated dose regimens. However, in a combination with a CTLA-A-4 antibody, immune-mediated abscopal effects were observed after hypofractionated radiotherapy. However, intermediate doses (3 × 8 Gy) showed a better effect than lower doses (5 × 6 Gy), indicating a specific therapeutic window for the combination of radiotherapy and CTLA-4 blockade, which may increase the efficacy of both modalities [67].
This immune-stimulatory potential, particularly when combined with immunotherapy, could improve the response rate of these two modalities. To date, optimal dose concepts and fractionation regimens remain ambiguous and seem to vary depending on the specific tumor histology. Nevertheless, a dose and fractionation regimen adjusted for an optimal local response may be expected to differ from that optimized for a distant abscopal response [68]. Therefore, intermediate doses appear to provide superior control of tumor growth as well as induction of an anti-tumor immune response. These findings provide important clues for sequential treatment planning and are relevant from the perspective of brachytherapy. Brachytherapy may play a decisive role in this approach. In particular, the doses favored in the intermediate dose range can be applied by brachytherapy and are often generated in a few fractions. During weekly intervals, immune cells have sufficient time for reconstitution. A highly conformal dose distribution allows sparing of lymphatic drainage pathways, lymph nodes, and surrounding organs at risk, which facilitates the management of side effects (Figure 2).
Brachytherapy and immunotherapy – perspectives and challenges
Uterine cervical cancer
Chemoradiotherapy with subsequential brachytherapy is the state-of-the-art treatment for locally advanced uterine cervical cancer. Several studies are examining the use of immuno-therapeutic drugs in the definitive treatment of uterine cervical cancer. Ongoing CALLA trial (NCT02635360), a randomized, multi-center, double-blind, placebo-controlled, global, phase III study investigates the efficacy and safety of durvalumab additional to chemoradiation or chemoradiation alone. Furthermore, KEYNOTE-A18 (NCT04221945) evaluates the efficacy and safety of pembrolizumab plus concurrent chemoradiotherapy compared to placebo and chemoradiotherapy, followed by brachytherapy in patients with locally advanced cervical cancer. These studies are amongst the first phase III trials combining a curative intended treatment, including brachytherapy, combined with immunotherapy. Brachytherapy is an integral part of the treatment regimen to achieve optimal local tumor control in adherence to dose constraints. Apart from uterine cervical cancer, other virally-driven tumors, such as head and neck squamous cell carcinoma (HNSCC), anal squamous cell carcinoma (ASCC), or Merkel cell carcinoma, are known for their intrinsic radiation sensitivity and are considered as more immunogenic [69].
Head and neck squamous cell carcinoma
Head and neck cancers are considered more immunogenic, and immunotherapy has become an integral part of treating recurrent or metastatic disease. Chemo-/radiotherapy is the primary treatment modality for HNSCC, and in combination with immunotherapy, its potent anti-tumor response has been reported. Unfortunately, HNSCCs tempt to relapse, and after a failure of the first-line or second-line therapies, treatment options are limited due to toxicities resulting from previous treatment modalities [70]. The indication of brachytherapy for head and neck cancers differs from curative to palliative settings and from definitive to adjuvant settings, with or without surgery or EBRT, and especially as re-irradiation. Published results should encourage to further investigate brachytherapy as a part of the treatment of HNSCC to achieve local dose intensifications [71]. Therefore, concurrent application of chemotherapy can improve survival and response rates [72]. Accordingly, the combination with immunotherapy could offer further advantages. In particular, as palliative intent or salvage brachytherapy, the effectiveness of irradiation alone could increase, while toxicity remains low.
Skin cancer
Brachytherapy is a proven and valuable tool to achieve high rates of local tumor control for non-melanoma skin cancers. Especially for delicate regions, including face and scull as well as in non-resectable settings, recurrent stages after surgery, and EBRT, brachytherapy provides a treatment alternative, preserving function and good cosmetic results [73]. While immunotherapy is well-established in treating melanoma, other immuno-therapeutic approaches in treating different entities of skin cancer are evolving. In 2018, anti-PD-1 antibody cemiplimab was approved by the FDA as the first systemic treatment for advanced squamous cell skin cancer [74]. The combination of brachytherapy and PD-1-blockade may provide an evident impact on local tumor regression and long-term response, and could be the first step towards highly personalized oncology in treating non-melanoma skin cancer.
Triple-negative breast cancer
Atezolizumab has been approved by the FDA and EMA to treat non-resectable locally advanced or metastatic triple-negative, PD-L1-positive breast cancer in combination with nab-paclitaxel. Previously, atezolizumab has significantly prolonged progression-free survival compared to patients treated with nab-paclitaxel alone [75]. Despite narrow indications, these data suggest a potential benefit of using immunotherapies in treating breast cancer. Accelerated partial breast irradiation (APBI) has been proven as an effective alternative to standard EBRT after breast conserving surgery. Following breast-conserving surgery and adjuvant radiotherapy, in addition to a lower rate of local recurrence, improved distant metastasis-free survival was also shown, which could indicate a systemic effect of local radiotherapy on micro-metastases [76]. Furthermore, brachytherapy is suitable as a re-irradiation for patients with local recurrence after breast-conserving surgery or mastectomy [77]. In this setting, systemic treatment could be beneficial or even necessary. Regarding their side-effect profile, immunotherapies may offer a tolerable synergy.
Challenges
Brachytherapy is an efficient and cost-effective treatment option for several tumor entities. However, its use decreased over the past decades among other treatment modalities due to the implementation of modern external beam irradiation techniques. Therefore, it becomes difficult to investigate the interaction between immunotherapy and brachytherapy in large prospective trials, as many centers do not offer brachytherapy. Also, the planning of such studies is challenging, especially that the optimal dose of brachytherapy or appropriate timing of immunotherapy administration relative to brachytherapy is still unknown. In spite of these challenges, it is of utmost importance to initiate prospective trials, focusing on the interaction of immunotherapy and brachytherapy, since there are clinical evidence that such treatment approaches may lead to better outcomes in several tumor entities.
Conclusions
Radiotherapy could be an optimal partner for immunotherapies. To date, immunotherapies are radically changing the management of an enormous array of different malignancies, and combinative therapies may enhance distance or systemic disease control via radiation-induced immune response and inducing abscopal effects. Nevertheless, the efficacy of immunotherapies differs widely, and there is an urgent need to define biomarkers, determine resistance mechanisms, and identify strategies to increase response rates. These could help to stratify patients, who have the best benefit in relation to adverse effects, and acute and late toxicities.
Brachytherapy, in combination with immunotherapy, appears to be a considerable partner to enhance local tumor control and may systemize responses of non-irradiated lesions. Even though we focused on the combination of checkpoint inhibitors in this article, there is a variety of approaches in immunotherapy. These therapies include adaptive T cell therapy, immune modulators, e.g., cytokines, therapeutic cancer vaccines, and therapies using oncolytic viruses. These therapies are going to provide a vast number of opportunities and challenges in future cancer treatment that were superficially summarized in this review. At this point, given the scarcity of relevant literature, we can only speculate about the potential of immuno-therapeutic combinations and approaches from the perspective of brachytherapy.