Purpose
Cancer of external auditory canal (EAC) is a rare malignancy representing less than 0.2% of all head and neck cancers, with only four percent of cancers of the outer ear confined to the EAC. The annual incidence of carcinoma of the EAC was estimated to be between one to six per million population [1]. Due to the rarity of this condition, evidence on its’ diagnosis and management is scarce, and limited to retrospective studies. Several staging and prognostic systems could apply. If considered as a cutaneous squamous cell carcinoma (SCC) of EAC, the seventh edition of the American Joint Committee on Cancer (AJCC) stages the primary tumor according to the size, extent, and presence of high-risk features, such as thickness, depth of invasion, perineural invasion, degree of differentiation, and anatomic location. Size, number of lymph node metastasis, and presence of distant metastasis are also considered. On the other hand, the University of Pittsburgh staging system evaluates the primary tumor according to the level of bony erosion, size, and involvement of the middle ear. The presence of involved lymph nodes denotes advanced stage of the disease with a poor prognosis [2]. Squamous cell carcinoma is the most common histology of cancers of EAC and temporal bone, accounting for 90% of the cases. Other primary histologic types of neoplasms include adenocarcinoma, adenoid cystic carcinoma, mucoepidermoid carcinoma, basal cell carcinoma, ceruminous carcinoma, and rhabdomyosarcoma. Chronic otorrhea, inflammation, or cholesteatoma within the EAC are associated risk factors in developing EAC SCC [3].
Management of EAC cancer is challenging due to its anatomical location and invasive nature. Although several treatment modalities have been described in the literature, there is a lack of consensus on the optimal treatment, mainly due to the absence of prospective randomized studies. The most frequently reported treatment involves surgical resection with or without adjuvant radiotherapy. Outcomes for chemotherapy and brachytherapy have been reported, but their exact role remains to be determined [4]. Oncologic resection and plastic reconstruction are technically challenging and associated with significant morbidity; therefore, there is a growing interest for non-invasive modalities, such as brachytherapy. Brachytherapy may be utilized for patients with early-stage EAC lesions with deep infiltration and those who are medically inoperable [5].
Case presentation
A 21-year-old male presented with persistent foul-smelling, purulent discharge from the right ear. A 2 cm mass was seen in the right EAC during otoscopy, and was biopsy-confirmed as SCC. CT scan showed a 1.1 cm × 0.8 cm ipsilateral pre-auricular lymph node. He was clinically diagnosed as stage III (cT1N1M0). After multidisciplinary team conferences and presentation of the case in the institutional head and neck tumor board, a consensus on definitive non-surgical treatment was reached. The patient underwent definitive high-dose-rate (HDR) intracavitary brachytherapy, 340 cGy per fraction for 14 twice-daily fractions, followed by external beam radiotherapy (EBRT) to cover the grossly enlarged pre-auricular node, ipsilateral intra-parotid nodes, and cervical lymph node levels II and III.
Material and methods
HDR intracavitary brachytherapy
Applicator design
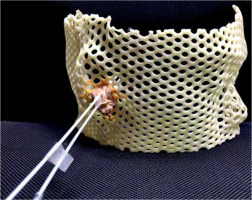
Prior to the procedure, a customized thermoplastic mask (Figure 1) was fitted to the patient in the left lateral decubitus position. Two ProGuide catheter needles were fixed in the mask into the EAC, ensuring that the surface of the tumor was well-covered.
Treatment planning
With the mask in place, the patient underwent a computed tomography (CT) simulation scan with 1 mm slice thickness to visualize and evaluate the catheter positioning, depth, and length. Gross tumor was contoured in the CT scan as high-risk clinical tumor volume (CTV-HR), and tumor bed plus a 3 mm margin was delineated as the intermediate-risk CTV (CTV-IR). Flexitron iridium-192 (192Ir) HDR brachytherapy plan was generated using Oncentra brachytherapy planning system (Oncentra Brachy, Elekta AB). The plan was generated using two channels, single unidirectional stepwise moving source, and remote afterloading with flexible catheters.
Treatment delivery
Clinical and radiographic assessment of the catheter positions before each fraction was done to ensure accuracy of treatment delivery. CT markers were placed in the catheters, then orthogonal radiographs were acquired. After verification, CT markers were removed and replaced with transfer tubes. Radiation was delivered to CTV-HR at a dose of 340 cGy, and to CTV-IR at a dose of 250 cGy for 14 twice-daily fractions, with an inter-fraction interval of at least six hours. Assuming no tumor repair in the six-hour interval, the dose for CTV-HR was D90 ≥ 6.8 Gy/day, and more than 5 Gy/day for CTV-IR D90. Assuming an α/β of 10 Gy for SCC of the head and neck [6], CTV-HR total dose was 47.6 Gy with EQD2 of 66.6 Gy. For CTV-IR, the total dose was 35 Gy with EQD2 of 43.8 Gy.
External beam radiotherapy
The patient was simulated in the supine position with a thermoplastic head and shoulder mask. EBRT was delivered using six coplanar intensity-modulated fields. The enlarged right pre-auricular node received 66 Gy in 2 Gy fractions, while the right intra-parotid nodes and cervical lymph nodes levels II and III simultaneously received 59.4 Gy in 1.8 Gy fractions.
Results
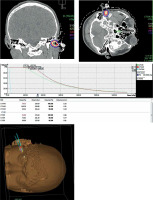
Isodose plots and dose volume histogram (DVH) of the brachytherapy and EBRT plans are shown in Figures 2 and 3, respectively. Both plans were delivered as prescribed. For HDR brachytherapy, the approved plan had an average CTV-HR D90 of 341 cGy/fraction, resulting in 47.7 Gy (biologically effective dose [BED] of 80.3 Gy, and EQD2 of 66.9 Gy). The average CTV-IR D90 was 251 cGy/fraction, with a cumulative dose of 35.1 Gy (BED of 52.8 Gy, and EQD2 of 44 Gy). Maximum dose (Dmax) to the ipsilateral cochlea was limited to only 32 cGy and median of 25 cGy.
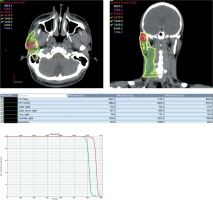
The accepted IMRT plan delivered at least 62.7 Gy to 95% of the enlarged pre-auricular lymph node, and 56.4 Gy to 95% of the high-risk nodal regions. Organs at risk were kept below their respective dose constraints.
At the end of both procedures, no grade ≥ 2 treatment-related adverse events were documented. The patient experienced grade 1 dermatitis in the right pre-auricular and cervical areas during the course of EBRT. Fifteen months after the completion of treatment, the patient has no evidence of disease; however, he was noted with stenosis of the right EAC with ipsilateral moderate conductive hearing loss. Thyroid function was normal at 15 months after EBRT.
Discussion
Cancers of the external ear are traditionally managed surgically followed by post-operative radiotherapy to improve loco-regional control in high-risk cases. Surgical techniques for EAC malignancies include sleeve resection, lateral temporal bone resection (LTBR), sub-total temporal bone resection (STBR), or total temporal bone resection (TTBR), which is chosen according to the site and extent of the lesion. LTBR is the primary surgery of choice for T1 and T2 tumors, while sleeve resection is indicated only for low-grade tumor of cartilaginous EAC skin without bone involvement. STBR is usually indicated as treatment of choice for T3 and T4 tumors, and TTBR is typically advised for advanced T4 tumors. Neck dissection is performed for clinically node-positive disease and advanced tumors. The most commonly affected lymph nodes are the cervical lymph nodes level I-IV. The evidence for prophylactic neck dissection for early-stage clinically N0 neck is unclear. The parotid is also susceptible to extension of tumor in the EAC. It has been reported that the incidence of parotid involvement in SCC of the EAC is between 10 and 62% of patients [7].
There are no prospective or randomized trials about EAC cancer treatment, and there is no clear consensus about treatment strategies, especially in advanced cases. A retrospective study done in Japan showed similar 5-year overall survival in those treated with definitive radiotherapy compared with surgically treated patients [8]. Outcomes of definitive radiotherapy for EAC SCC was reported by Pemberton et al., wherein they delivered 55 Gy in 20 fractions of RT. Six months post-RT, complete response was documented in 50% of patients and 27% showed partial response. At 5 and 10 years, respectively, freedom from local recurrence was observed in 56% and 56%, disease-free survival was 45% and 43%, cancer-specific survival was 53% and 51%, and overall survival was 40% and 21%. Cancer-specific survival for patients with early-stage disease was 85% and 78% at 5 and 10 years, respectively, as opposed to 44% and 44% for individuals with late-stage disease. Recurrence is mainly loco-regional, and usually salvaged surgically. Bone necrosis was documented in six patients and soft tissue necrosis in two cases [9]. A retrospective review by Hashi et al. showed the role of primary EBRT or surgery with EBRT in early-stage EAC cancers. RT dose was 65 Gy in 26 fractions for patients treated with RT alone, while 30-75 Gy in 12-30 fractions was applied for patients treated with perioperative RT. The 5-year survival rates for eight patients with T1 disease and eight with T2 disease were 100% and 38%, respectively. In the eight patients with T1 disease, disease control was 100%. No late complications related to radiotherapy (e.g., hearing problems) were observed. The authors concluded that radiotherapy is a viable treatment option for patients with T1-stage disease, while surgery with radiotherapy is recommended as standard care for tumors with bony invasion [10].
Chemotherapy is not routinely used but usually recommended as an adjuvant treatment for advanced disease or due to the presence of residual disease. However, an individual patient meta-analysis showed the benefit of chemoradiation (CRT) as a pre-operative or adjuvant treatment, or definitive treatment in T3-T4 tumors. The regimens used in pre-operative and post-operative settings were fluorouracil-based and intravenously administered platinum-based regimens, respectively. The 5-year overall survival rates of patients who received surgery ±RT, pre-operative CRT, definitive CRT, and post-operative CRT were 53.5%, 85.7%, 43.6%, and 0.0%, respectively. Their data suggested that pre-operative CRT may improve the survival of surgically treated patients with EAC SCC, and that definitive CRT may be equivalent to surgical resection [11].
On the other hand, brachytherapy using remote after-loading HDR radioactive sources (e.g., 192Ir) has the advantage of delivering very high doses of radiation to the target volume with high conformity and precision. It also has the advantage of better sparing of surrounding critical structures. According to the Indian Brachytherapy Society guidelines and recommendations for brachytherapy in head and neck malignancies, selection of candidates for brachytherapy depends on the accessibility of location of the primary tumor, proximity to cartilage or bone, size of less than 2 cm, presence or absence of trismus/submucous fibrosis (SMF), and previous treatment. In early-stage node negative disease, 45-54 Gy, 4-4.5 Gy per fraction in 10-12 fractions may be given (BED, 65-84 Gy) as radical treatment. Recommended dose for HDR brachytherapy boost after external irradiation is 16-22 Gy, 4-4.5 Gy per fraction in 4-5 fractions (BED, 26-32 Gy) in T1 disease, while in T2 disease, the recommended dose is 22-26 Gy, 4-4.5 Gy per fraction in 5-6 fractions (BED, 32-40 Gy) [12].
Published case reports showed the value of brachy-therapy as a boost after definitive EBRT. This approach led to improved local tumor control up to four years, with less side effects and better cosmesis [13]. Moreover, brachytherapy can be utilized as salvage treatment in cases of local recurrence after surgery and full-course EBRT [14]. In the current report, we presented a case of SCC of the EAC with low tumor burden, treated radically with HDR brachytherapy and EBRT to address the lymph node regions at high-risk for metastasis. Despite the more robust data on EBRT for the treatment of head and neck cancers, brachytherapy may be of value in sub-sites that are confronted by dosimetric challenges due to the proximity to air cavities or sinuses. This holds true for the EAC, which is a cavity in itself and is located in close proximity to other air cavities (e.g., mastoid air cells or maxillary sinus). The result of a study of Seif et al. revealed that the presence of air cavities leads to the generation of overdose and underdose regions in the intersection of air-tissue, and it also is a contributing factor in inhomogeneity and fluctuation in dose distribution. Specifically, there is a dose reduction before air cavity due to the lack of scattered and returning beams. Meanwhile, since air cavities have a lower density compared with tissues, beam attenuation is lower, and more beams pass through and get deposited farther. Therefore, there is an increase in absorbed dose after cavities. The dose increase or decrease in the air-tissue interface are dependent on geometry, size, and depth of the cavity [15]. The capability to address dose inhomogeneity, shorter overall treatment time, and better cosmetic outcomes of brachytherapy make it ideal for EAC cancers. However, for the presented case, brachytherapy was limited in addressing lymph node stations at risk for tumor spread; hence, EBRT was necessary.