Purpose
Nowadays, the interest of salvage treatments for locally relapsed prostate cancer after primary radiotherapy (RT) are boosting among radiation oncologists due to progress in image guidance and precision in dose delivery [1, 2]. Little has been reported on gastro-intestinal (GI) and genitourinary (GU) toxicity developing years after re-irradiation, while analyses correlating dosimetric and clinical parameters with long-term tolerance are scarce [3-5]. Indeed, in 2016, we have reported the long-term GI and GU toxicity profile for a group of 14 patients salvaged with external beam radiotherapy (EBRT) with or without a brachytherapy (BT) boost, for exclusive local failures following previous EBRT [6]. In a later report in 2018 [7], we have published an analysis on the correlation of rectal toxicity for the same patients with rectal dose-volume parameters quantifying late GI toxicity, with normal tissue complication models (NTCP).
The purpose of the present study was to retrospectively assess the possible association between treatment and dosimetry features and GU toxicity, thus aiming to identify potential predictors for severe GU toxicity after re-irradiation.
Material and methods
A cohort of 14 locally recurrent prostate cancer patients treated at the Radiation Oncology Department of the Geneva University Hospital between 1992 and 2008 was analyzed. Median (minimum-maximum) time interval between salvage and primary RT treatments was 6.1 years (range, 4.7-10.2 years). All but two patients with grade 2 GU toxicity were free of any GI or GU toxicities at salvage RT [6] as assessed using common terminology criteria for adverse events version 3. Patients were regularly seen for status verification during and after treatment completion [7]. Tables 1 and 2 present clinical characteristics, dose prescription, treatment techniques, and GU toxicity grading for the 14 patients as previously reported [6].
Table 1
Patients’ clinical and dosimetric characteristics
Table 2
Late genitourinary (GU) toxicity scores (CTCAE v.3.0 scale)
[i] HBOT – hyper-baric oxygen therapy, TURP – trans-urethral prostate resection, Re-printed with permission from Zilli T et al. [4]
Median dose delivered to the prostate in 2 Gy-equivalent dose (EQD2Gy) per fraction (EQD2Gy, α/β ratio = 1.5 Gy) was 74 Gy at primary irradiation. For salvage RT, four patients were treated with exclusive EBRT, and the remaining 10 patients were treated with a combination of EBRT and BT boost. Median EQD2Gy (α/β = 1.5) of the whole group at re-irradiation was 85 Gy (Table 1). High-dose-rate (HDR) BT boost was delivered in three or more consecutive days. When using intensity-modulated radiation therapy (IMRT) and salvage treatments, set-up verifications were performed using electronic portal imaging on bony anatomies, and an off-line protocol as previously reported [7].
In order to explore the data, and to assess dose parameters and clinical factors potentially associated with severe long-term GU toxicity, study population was stratified into two groups according to toxicity, including one low-grade group (grade ≤ 2, n = 6) and one high-grade group (grade ≥ 3, n = 8). Physical dose was translated to EQD2Gy, according to linear quadratic model, using a α/β = 1.5 Gy for prostate cancer and a α/β = 3 Gy for bladder and urethral late toxicity [7].
In order to analyze the data appropriately, contouring guidelines [8] were applied to re-contour the bladder both as a solid structure and as the bladder wall (defined as a wall thickness of 5 mm). An experienced radiation oncologist (TZ) contoured all patients and structures to eliminate inter-contouring variability effects from different radiation oncologist contouring in the analysis. Eclipse version 10 (Varian, Palo Alto, USA) treatment planning system was used for contouring. For primary treatments, it was possible to obtain BT dose contribution to the urethra and bladder in the form of dose-volume histogram (DVH) by using paper hard copies. This allowed us to investigate dose-volume relationships and if doses received at primary treatment could predict toxicity at re-irradiation.
In order to consider factors that might reduce the capability of each patient to recover from the irradiation, such as pre-existing vascular morbidity, which could increase the risk of damage to the bladder and urethra at re-irradiation, Framingham risk score was applied. For each patient, the predicted 10-year cardiovascular risk was calculated and was used to divide patients into high-risk and low-risk score groups using a cut-off value of 20% [9].
Mann-Whitney test was applied to evaluate the impact of continuous variables; for cross-comparison between toxicity (grade 0-2 vs. grade 3-4) and dichotomous variables, such as Framingham risk score (with/without BT boost), BT boost, and pelvic RT, a two-tailed Fisher exact test was used. SPSS software, version 22.0 (IBM USA) was employed for statistical analysis. This study was carried out within a retrospective research project on prostate cancer hypofractionation approved by local ethical committee (project No. 2018-00614). No signed informed consent was required.
Results
With a median follow-up after re-irradiation of 94 months (range, 48-172 months), the 5- and 8-year probability (± SD) for grade ≥ 3 late GU toxicity-free survival were 77.9 ±11.3% and 55.7 ±15.6%, respectively. Four cases of grade 3 and four cases of grade 4 GU toxicity were observed (Table 2). The estimated 5-year biochemical relapse-free survival was 36% [6]. The median time-interval between primary RT and salvage RT (6.6 vs. 5.7 months) as well as the median age at primary RT (58 vs. 62 years old) or at salvage RT (67 vs. 69 years old) were similar for the low- and high-grade toxicity groups (p = not significant [N.S.]). Whole pelvis RT at primary RT was not associated with the development of severe GU toxicity as well as the size of the bladder volume at salvage RT (p = N.S.).
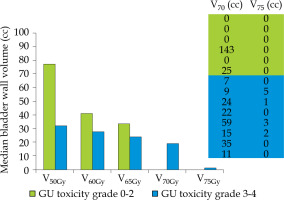
The median bladder wall V75Gy (volume receiving 75 Gy) at primary RT was predictive for grade ≥ 3 GU toxicity; 1 cc vs. 0 cc, p = 0.05 (Figure 1). In addition, both V70 > 0 cc and V75 > 0 cc were significantly associated with an increased toxicity: the rate was 8/10 vs. 0/4 for V70 > 0 and = 0 (p = 0.008, χ2 test). Similarly, the rate was 4/4 and 4/10 V75 > 0 and = 0 (p = 0.019, χ2 test).
Fig. 1
Median bladder wall volume (cc) receving a dose of 50, 60, 65, 70 and 75 Gy as a function of the GU toxicity grade: low vs. high
Late grade ≥ 3 GU toxicity was observed in 8 out of 10 patients treated with salvage EBRT and BT boost, while no patient out of the 4 treated with highly conformal EBRT developed severe GU toxicity (p = 0.008). If considering one patient treated with a pulse-dose-rate BT (0.5 Gy × 50) with EBRT only, the corresponding proportions became 8/9 patients vs. 0/5 patients (p = 0.002) (Table 1).
All patients salvaged with a BT boost and presenting with a baseline Framingham risk score of > 20% (n = 8) developed severe GU toxicity, while none of the six remaining patients developed grade 3 or more GU toxicity (p = 0.0003) (Table 1). Of note, 3 patients with a Framingham score of < 20% presented grade 1 GU toxicity only.
At primary RT, the median delivered EQD2Gy (α/β = 3 Gy) was 74 Gy (range, 72-76 Gy) and 73 Gy (range, 67-92 Gy) for the high- and the low-toxicity groups, respectively (p = N.S.). On the other hand, at salvage RT, the median EQD2Gy (α/β = 3 Gy) was significantly higher in the high-toxicity group (85 Gy, range, 71-85 Gy) compared with the low-toxicity group (77 Gy, range, 61-85 Gy) (p = 0.01). Differences were even more significant between the low- and the high-toxicity groups by using a α/β ratio of 1.5 Gy instead of 3 Gy (p = 0.007).
By cumulating the primary and salvage RT courses, the median delivered total EQD2Gy (α/β = 3 Gy) was 159 Gy (range, 145-161 Gy) and 156 Gy (range, 128-170 Gy) for the high- and low-toxicity groups, respectively (p = N.S.). A α/β = 1.5 Gy did not discriminate between the two groups.
Discussion
Although, the risk of severe GU toxicity after curative EBRT for prostate cancer is expected to rapidly increase with EQD2Gy above 80 Gy [10], our data suggests that a partial, though incomplete, repair of the bladder wall after first irradiation may allow a second irradiation to radical doses, especially if the interval between treatments is of at least 3-4 years, and a dose to the bladder lower than 75 Gy after first irradiation attempt. In the present study, by lowering the α/β ratio from 3 Gy to 1.5 Gy, we were able to better distinguish the effect of salvage dose prescription between patients with or without severe toxicity after re-irradiation. This result is consistent with data from previously published literature, suggesting an α/β ratio as low as 1 Gy for late GU toxicity [11, 12].
The encouraging results on early and late toxicities after stereotactic body RT (SBRT) re-irradiation [2, 3, 13-16] with doses ranging between 25 Gy and 36.25 Gy in 5 fractions, are consistent with our findings. Indeed, the corresponding EQD2Gy (α/β = 3) of SBRT salvage schedules ranged between 40 Gy and 74 Gy, well below the 80 Gy threshold emerging from our analysis. Moreover, in these series, the use of modern image-guided techniques for re-positioning and reduced treatment margins [1], together with SBRT treatments delivered to the local relapse only and not the whole prostate gland as in our series [17], can certainly attenuate the dose-effect relationship for severe GU toxicity observed in our study at re-irradiation, showing that safe salvage RT can be delivered with lower salvage doses. Nevertheless, the compromise may be a lower probability of disease control when primary and salvage EQD2Gy (α/β = 3 Gy) doses are lower than 130 Gy to the prostate [3]. There is strong need to investigate the dose limits and possible threshold effects on organs at risk.
Leeman et al. [18, 19] analyzed 23 studies (n = 2,232 patients) and have shown that even after SBRT as primary treatment, urinary toxicity can be the determining factor and strongly correlates with high-doses received by the urethra. Patients with 146 Gy maximum urethral dose metric had an estimated 5% risk of late grade 3 GU toxicity, with a predictive curve showing that above this value, toxicity may increase dramatically with a possible threshold dose of 150-160 Gy.
In the absence of complete DVH data on the two treatments, we considered the prescribed dose to the target to be a surrogate of the dose received by a portion of the bladder and by the prostatic urethra, as both were within planning target volume (PTV) and clinical target volume (CTV). Unlike exclusive EBRT, combined EBRT and BT treatments may have resulted in a marked inhomogeneous dose distribution within the target and nearby organs at risk, such as the bladder and urethra. This and the very high-dose per week delivered via brachytherapy at salvage RT may explain high-rate of severe GU complications due to the implant itself as well as the absence of correlation with total prescribed dose. Perhaps, this is what has limited our ability to define a combined ‘primary and salvage’ threshold dose for GU toxicity. On the other hand, it is worth mentioning the association between V70/V75 of the first treatment and the increased risk of toxicity.
As for the clinical factors observed for late severe GI toxicity [7], Framingham risk score at primary RT correlated with grade ≥ 3 late GU toxicity, and may help to select optimal candidates for salvage RT. These results are somewhat consistent with other studies that have reported age [20, 21], vascular problems [22], and smoking [23] to be associated with an increased risk of late GU symptoms after primary irradiation. Our study, besides the small sample size and its’ retrospective nature, presents a main flaw, which is the impossibility to evaluate the salvage RT dose contribution of BT to the bladder and the urethra in terms of DVH and/or dose distribution.
Conclusions
In summary, in the present study, we were able to identify possible dosimetric and clinical factors that could be involved with the development of long-term severe GU side effects after re-irradiation of a prostate cancer local failure. Our data support that salvage re-irradiation to EQD2Gy (α/β = 3) below 75-80 Gy to the bladder and urethra, may result in safe and acceptable long-term GU toxicity. Concomitant cardiovascular comorbidities, according to the Framingham risk score, may also predict the risk to develop severe GU toxicity.



