Purpose
Radiotherapy is a potentially curative treatment modality for patients with prostate cancer. However, depending on the individual risk factors and comorbidities, 10-40% of patients experience a biochemical recurrence within 10 years [1,2,3]. Patients with localized recurrence following definitive radiotherapy may be cured with a salvage treatment, such that the adverse effects of androgen deprivation therapy may be avoided or postponed. Several previous studies have described their experiences with this treatment modality and have indicated promising results for salvage treatments [4]. Salvage brachytherapy (BT) and radical prostatectomy are included as possible treatment options for patients with local recurrence according to the National Comprehensive Cancer Network (NCCN) guidelines [5]. However, data on long-term outcomes after salvage BT are still relatively sparse [6,7,8,9,10]. In order to provide an adequate guidance to patients who may qualify for salvage therapy, more data on treatment results are required.
In Japan, the use of the iodine-125 (125I) seed source was approved in July 2003, and the public health insurance program began to cover the cost of robot-assisted radical prostatectomy (RARP) in April 2012. Brachytherapy and RARP became standard options for initial treatment in 2003 and 2012, respectively, and are gradually beginning to be used in the salvage setting. To the best of our knowledge, our study is the largest case series of salvage BT and salvage RARP reported from Japan to date. Therefore, the purpose of this study was to describe our experience with whole-gland salvage BT and salvage prostatectomy among patients with pathology-proven, locally recurrent prostate cancer following primary definitive radiotherapy.
Material and methods
We retrospectively reviewed the medical records of patients who were diagnosed with local recurrence only and received whole-gland salvage treatment at our institution between February 2010 and July 2017. We identified 18 patients who met these criteria. Patient characteristics are shown in Table 1. Primary definitive external beam X-ray radiotherapy (EBRT) consisted of a median dose of 76 Gy (range, 72-76 Gy), and carbon-ion therapy consisted of a dose of 57.6 GyE. All patients underwent transperineal template-guided mapping biopsy (TTMB) using the standard 125I BT template with a 5 × 5 mm grid and were diagnosed with pathology-proven locally recurrent prostate cancer. The patients underwent various forms of examination, including computed tomography (CT), magnetic resonance imaging (MRI), bone scintigraphy, and 18F-fluorodeoxyglucose (FDG) positron emission tomography (PET)/CT, and had no evidence of regional or distant recurrence. MRIs consisted of T2-weighted imaging and diffusion-weighted imaging with or without contrast-enhanced imaging. 11C-choline, 11C-acetate, and prostate-specific membrane antigen (PSMA) have not been approved as PET-CT tracers in Japan. Treatment strategies were determined based on individual patient preference and physician judgment. Salvage BT was not recommended for patients who experienced grade ≥ 3 genitourinary (GU) or gastrointestinal (GI) toxicity or had residual grade ≥ 2 GU or GI toxicity after initial radiotherapy. Twelve patients received 125I BT and six patients underwent non-nerve-sparing salvage RARP. One patient in the BT group received 9 months of hormone therapy at the judgement of the treating physician. An intra-operative planning protocol was implemented using VariSeed Software (Varian Medical System Inc.). The prescribed radiation dose was 110 Gy, and the dose to 90% of the prostate (D90) was aimed at 120% of the prescribed dose. 125I seeds, model 6711 (GE Healthcare) or STM 1251 (Bard Medical Division), were used for BT. For patients who received a second BT, an alternative seed type was selected to distinguish the newly administered seeds from the original seeds. Post-implant CT-based dosimetry was performed at day 30 after implantation. The dosimetric data for patients undergoing salvage BT are presented in Table 2. We performed obturator lymph node dissection in five out of six patients and found no lymph node metastases.
Table 1
Patient characteristics
| BT(n = 12) | RARP(n = 6) | |
|---|---|---|
| Initial diagnosis | ||
| Age* (years) | 66 (56-78) | 60 (55-69) |
| PSA* (ng/ml) | 7.7 (4.5-24) | 7.7 (6.5-11.5) |
| T stage† | ||
| 1c | 2 | 4 |
| 2 | 10 | 1 |
| Unknown | – | 1 |
| GS | ||
| ≤ 6 | 4 | 3 |
| 3 + 4 | 6 | 2 |
| 8 | 2 | 1 |
| Primary radiotherapy† | ||
| Seed | 7 | 3 |
| Seed + EBRT | 3 | – |
| EBRT | 2 | 1 |
| Carbon-ion therapy | – | 2 |
| Salvage treatment | ||
| Pre-salvage PSA* (ng/ml) | 3.3 (0.0-8.0) | 2.7 (1.7-5.5) |
| GS† | ||
| ≤ 6 | 2 | – |
| 3 + 4 | 3 | 1 |
| 4 + 3 | 4 | 3 |
| 8 | 1 | – |
| Unable to be graded | 2 | 2 |
| Interval to relapse* (mo) | 62 (40-98) | 57 (33-127) |
| PSA doubling time* (mo) | 11 (4.7-18) | 12 (3-34) |
| Total biopsy core† | 31 (21-48) | 36 (24-44) |
| Positive core† | 1 (1-11) | 4 (1-10) |
| MRI† (yes/no) | 5/7 | 6/0 |
| ADT† (yes/no) | 1/11 | 0/6 |
| ADT duration* (mo) | 9 (9-9) | – |
Table 2
Dosimetric parameters for salvage brachytherapy
| Prostate | |
| D90 (Gy) | 131 (110-149) |
| V100 (%) | 98 (90-100) |
| V150 (%) | 54 (29-74) |
| Rectum | |
| V100 (cc) | 0.18 (0-0.6) |
| Urethra | |
| D30 (Gy) | 142 (121-174) |
Clinical follow-up evaluations included symptoms assessment and prostate-specific antigen (PSA) tests at 3- to 4-month intervals. Post-salvage treatment GU and GI toxicities were graded according to CTCAE version 4.0. Late toxicities were defined as any symptom that occurred more than 12 months after the completion of salvage treatment. Duration of follow-up was measured from the time of the salvage treatment to the date of the last clinical evaluation.
Descriptive statistics were analyzed for patient characteristics and disease outcomes. PSA doubling time was calculated using the Memorial Sloan Kettering Cancer Center calculation tool [11]. PSA failure was determined using the Phoenix definition (nadir + 2 ng/ml) following BT and a PSA level of > 0.2 ng/ml following RARP. The Kaplan-Meier method was used for calculating biochemical relapse-free survival (BRFS) rates. All analyses were performed using the R 3.4.3 statistical package (R Foundation for Statistical Computing, Vienna, Austria; http://www.r-project.org/).
This study was approved by our institutional review board (IRB number: R18-143).
Results
Median follow-up times were 71 months (range, 19-92) and 11 months (range, 2-33) for the BT and RARP groups, respectively.
Salvage BT group
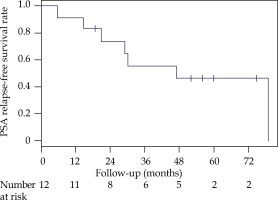
The median time to biochemical failure was 47 months, and the BRFS rates following salvage BT were 56% (95% CI: 33-94%) and 46% (95% CI: 25-88%) at 3 and 5 years, respectively (Figure 1).
The frequency of GU toxicity is shown in Table 3. Changes in urine frequency and urgency were observed, along with hematuria, dysuria, and urethral stricture. Four patients (33%) developed acute grade 2, and one patient (8%) developed acute grade 3 GU toxicity. Of the four patients who developed acute grade 2 GU toxicity, three patients had their symptoms resolve, while one patient developed grade 3 urethral stricture. This patient received a urethral bougie for urethral stricture and subsequently developed grade 3 urinary incontinence. The patient who initially developed acute grade 3 urethral strictures received a urethral bougie, which resolved obstructive symptoms and precluded the occurrence of urinary incontinence. No patients developed grade ≥ 2 GI toxicity.
Salvage RARP group
Three patients (50%) out of a total of six experienced PSA failure, with a median follow-up of 11 months. Pre-salvage MRI staging and pathological outcomes are presented in Table 4. On pre-salvage MRI, no patients were suspected of having lesions that were of stage T3 or higher. However, three patients (50%) had stage pT3a lesions and two patients (33%) had stage pT3b lesions. The frequencies of GU toxicity are shown in Table 3. Four patients (67%) developed acute grade 2 urinary incontinence, while no patients developed grade 3 GU toxicity. Of the four patients who developed acute grade 2 urinary incontinence, two patients experienced a resolution of symptoms. No patients developed grade ≥ 2 GI toxicity.
Table 4
Magnetic resonance imaging (MRI) staging and pathological outcomes
| MRI T staging | |
| 1c | 3 |
| 2a | 2 |
| 2c | 1 |
| Pathological stage | |
| pT2a | 1 |
| pT3a | 3 |
| pT3b | 2 |
| Surgical Gleason score | |
| 4 + 3 | 5 |
| 4 + 5 | 1 |
| Surgical margins | |
| Negative | 2 |
| Positive | 4 |
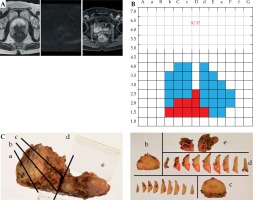
A representative case is presented in Figure 2. This patient was a 70-year-old man with low-risk prostate cancer, who received BT but still progressed to PSA failure after 127 months. The PSA doubling time was 14.5 months. No recurrent lesions were identified on the pre-salvage MRI. A pathology examination subsequent to TTMB revealed that 9 of 37 cores were positive and GS 3 + 4. Biopsy cores targeting seminal vesicles (SVs) were also positive. This relapse was then treated with RARP. The pre-salvage treatment PSA was 2.56 ng/ml. The final pathology following RARP indicating an adenocarcinoma with Gleason score 4 + 3, ly1, V0, pn1, sv1, pT3b, and N0. Post-RARP PSA never decreased to < 0.02 ng/ml, and hormone therapy was initiated at 12 months after RARP.
Fig. 2
A patient treated with robot-assisted laparoscopic prostatectomy (RARP). A) No recurrent lesions were apparent on pre-salvage magnetic resonance imaging (MRI) (T2-weighted, diffusion-weighted, and Gd-enhanced imaging). B) Sites of transperineal template-guided mapping biopsy (TTMB) are represented in blue or red. Pathology following TTMB revealed 9 of 37 cores to be positive (red areas). The biopsy cores targeting seminal vesicles (SVs) were also positive. C) Gross appearance of a RARP specimen sliced by a standard method. Tumors are encircled in red. The final pathology from RARP confirmed the presence of SV invasion
Discussion
In this study, BRFS rates were 56% (95% CI: 33-94%) and 46% (95% CI: 25-88%) at 3 years and 5 years, respectively, following salvage BT, with 33% of patients experiencing grade 2 GU toxicity and 17% developing grade 3 GU toxicity. Several studies have reported outcomes after salvage BT, but the available data on salvage low-dose-rate BT with a median follow-up greater than 5 years are limited [6,7,8,9,10]. In a series of 49 patients treated with salvage BT, Grado et al. reported a 5-year BRFS rate of 34%, with a median follow-up of 64 months [6]. In a series of 17 patients, Beyer et al. described a 5-year BRFS rate of 53%, with a median follow-up of 62 months [7]. In a study with 37 patients, Burri et al. reported a 5-year BRFS rate of 65% and a cumulative incidence of adverse events (grade ≥ 3) of 11%, with a median follow-up of 86 months [8]. In a series of 31 patients, Moman et al. described a 5-year BRFS rate of 20%, with a grade 3 GU toxicity rate of 19% and a grade 3 GI toxicity rate of 6%, and a median follow-up of 9 years [9]. In a study including 69 patients, with a median follow-up of 5 years, Vargas et al. reported that 89.9% of patients received androgen suppression as a part of their salvage treatment, with a 5-year BRFS rate of 73.8% for non-castration resistant prostate cancer (CRPC) patients and 22% for CRPC patients, and a grade 3 GU toxicity rate of 8.7% [10]. Our findings for PSA control are similar to those reported in the literature.
Historical data at our institution showed that the 7-year actuarial toxicity rates after 125I BT for ≥ grade 3 GU and GI toxicity were 2% and 0.3%, respectively, in the initial curative treatment setting [12]. The frequency of GU toxicity in this study was to some extent higher, which might imply that it is difficult to maintain a low-risk of GU toxicity in whole-gland BT, while it is possible to keep the risk of GI toxicity low. Peters et al. suggested that RV100 (the volume of rectum receiving 100% of the prescribed dose) less than 0.35 cc was necessary for avoiding severe GI toxicities in salvage BT [13]. The median RV100 in this study was 0.2 cc, which may be one reason for fewer adverse events.
While previous studies have used a dose of 120-145 Gy in salvage 125I BT [7,8,14,15,16,17], no optimal prescription dose for salvage BT has been established. In this study, the prescribed radiation dose was 110 Gy of 125I, and the median prostate D90 was 131 Gy (range, 110-149 Gy), which was rather low compared to the dose used in previous studies. Nguyen et al. reported a promising 4-year BRFS rate of 70% after 125I salvage BT, with a prescribed dose of 137 Gy [14]. However, the actuarial rate of grade 3/4 GU or GI toxicity was 30% at 4 years. Rose et al. reported an increase in late GU/GI toxicity, with a higher D90 [15]. Crook et al. reported that V100 was the only factor associated with late grade 3 GU/GI toxicities, suggesting that there are regions that should be protected from the full dose in the salvage setting [16]. In the context of this trade-off relationship, priority is given to focal therapy and partial prostate boost, after specifying a lesion site as a strategy for enhancing the usefulness of treatment. Aaronson et al. reported on this approach, delivering 144 Gy to the recurrent lesion sites identified by MRI, while concurrently delivering 108 Gy to the remaining prostate gland. They analyzed 24 patients and reported a 3-year BRFS rate of 89.5%. Toxicities included one grade 3 rectal hemorrhage, five instances of grade 2 gross hematuria, one grade 2 urethral stricture, one grade 2 urinary incontinence, and two instances of grade 1 hematochezia [17].
In this study, no patients were suspected of having T3 or higher stage lesions based on pre-salvage MRI. Nevertheless, multifocal lesions were apparent in specimens drawn from patients treated with salvage RARP. However, all patients in the study underwent TTMB, which identified multiple positive cores among patients in the salvage RARP group. As such, the results of TTMB may be useful for discriminating multifocal lesions from unifocal lesions and warrant further evaluation in future studies.
As an alternative side effect mitigation measure, Hepp et al. reported on a successful case, in which a polyethylene glycol hydrogel spacer was found to reduce the rectal dose in the salvage setting [18]. This strategy is promising and could lead to improved safety profiles for salvage treatment in the future.
In our study, three patients out of six (50%) experienced PSA failure following salvage RARP, with a median follow-up of 11 months. With pre-salvage MRI, no patients were suspected of having T3 or higher stage lesions; however, three patients (50%) had pT3a and two patients (33%) had pT3b stage lesions. These results demonstrated that it is difficult to evaluate T stage accurately after definitive radiotherapy.
In the past studies on salvage prostatectomy, it was reported that pathological stage T3 cases were not uncommon. In a multi-institute cohort of 404 patients undergoing salvage radical prostatectomy, the rate of seminal vesicle invasion (SVI) and extraprostatic extension (EPE) was 30% and 45%, respectively [19]. Takeda et al. examined 77 patients with local recurrence after radiotherapy and reported that the percentage of patients with a pre-salvage clinical classification of T3 was 10%. However, 42% of the surgical specimens in this study showed SVI, and 58% showed EPE [20].
A highly precise method of detecting SVI in preoperative biopsy samples has been reported [21], which may be of particular importance given the utility of biopsy including SV. SVI has been reported to be a poor prognostic factor for BRFS [21]. Therefore, it is considered useful to provide accurate diagnoses before counseling patients about expectations following salvage treatment.
This study has some limitations. It is a retrospective study with a relatively small number of patients. In addition, in the RARP group, the follow-up period was fairly short, because salvage RARP was introduced to Japan only recently. Furthermore, the treatment selection criteria and pre-salvage image evaluation were not uniform. Hence, although the findings reported here remain useful, as we report on long-term outcomes of salvage BT, further investigation is required. To the best of our knowledge, our study is the largest case series of salvage BT and salvage RARP reported from Japan so far, and is therefore worth reporting. Further research is needed in order to identify the specific patient population that is most likely to benefit from salvage treatment by considering clinical factors in addition to the imaging findings and TTMB results.
Conclusions
Whole-gland salvage BT is an effective treatment with an acceptable toxicity profile, although the frequency of GU toxicity is slightly higher than that observed during the initial treatment. Longer follow-up is necessary to evaluate the oncologic outcomes of salvage RARP and BT. The final pathology data from RARP implies that there is a room for improvement in diagnoses determined by MRI in the pre-salvage setting.