Introduction
Inflammatory bowel diseases (IBD), encompassing ulcerative colitis (UC) and Crohn’s disease (CD), are chronic, immune-mediated disorders of the gastrointestinal tract characterized by a relapsing–remitting clinical course and substantial disease burden [1–4]. Clinically, they present with a broad spectrum of manifestations, most commonly abdominal pain, diarrhea, low-grade fever, and features of malabsorption. The global incidence of these disorders shows a sustained upward trend, particularly among young and professionally active populations, underscoring their growing public health relevance [5]. Despite extensive global research efforts, a definitive disease-modifying therapy remains unavailable. Moreover, currently applied management strategies frequently fail to maintain long-term efficacy owing to secondary loss of response, highlighting the persistent need for novel therapeutic approaches.
The pathogenesis of IBD is multifactorial, involving genetic, immunological, and environmental factors. A growing body of evidence highlights the pivotal role of dysbiosis in the development and clinical course of these diseases, as reflected by the distinct composition of the gut microbiota in IBD patients compared with healthy controls [6–8]. These observations have intensified the scientific interest in the gut microbiome and its potential as a therapeutic target. Short-chain fatty acids (SCFAs), particularly butyric acid, have been shown to play an essential role in maintaining intestinal homeostasis, and their supplementation has been proposed as a strategy to support both the induction and maintenance of remission in IBD [9, 10].
Evidence from experimental models indicates that butyrate contributes to colonocyte energy metabolism, reinforces intercellular junctions, stimulates mucin and antimicrobial peptide expression, and supports mucosal healing. These effects involve immunomodulatory mechanisms, including activation of G-protein–coupled receptors (e.g. GPR41) and inhibition of histone deacetylases, resulting in attenuation of NF-kB- and STAT-dependent pro-inflammatory signaling pathways and modulation of cytokine profiles. Clinically, sodium butyrate (SB), particularly in microencapsulated formulations designed to optimize colonic delivery, has emerged as an adjunctive therapy in IBD, with strongest evidence in UC and more limited data in CD [9, 10]. Although current clinical data are constrained by small sample sizes, formulation heterogeneity, and dosing variability, the accumulating mechanistic and clinical evidence supports SB as a biologically rational adjunct in IBD. Further high-quality, adequately powered trials are warranted to clarify its therapeutic role and to determine whether optimized colonic butyrate delivery may represent a future avenue for personalized IBD management.
Butyric acid and its role in intestinal disorders
SCFAs, primarily acetate, propionate, and butyrate, are fundamental regulators of intestinal homeostasis. They represent approximately 83% of the SCFAs pool within the colon, with their relative distribution remaining stable at 60:25:15, respectively [11]. Butyrate is produced mainly through anaerobic fermentation of non-digestible carbohydrates, including resistant starch, inulin, insoluble fiber fractions, sugar alcohols, and endogenous substrates such as mucins and human milk oligosaccharides. The most efficient dietary sources of these substrates comprise resistant-starch–rich foods such as oats, wheat bran, high-amylose products, including partially processed grains and seeds, raw potatoes, and green bananas. However, these foods may exacerbate mucosal irritation and are therefore commonly restricted in the diets of patients with IBD. Such dietary limitations can further reduce colonic butyrate generation, contributing to a state of functional butyrate deficiency [12]. This effect is compounded by the dysbiosis characteristic of IBD, marked in particular by a reduced abundance of Firmicutes – particularly species within the Clostridium coccoides and Clostridium leptum – which are among the most important microbial producers of SCFAs. Their depletion leads to reduced luminal butyrate availability, a perturbation believed to be mechanistically relevant to both the initiation and persistence of mucosal inflammation in IBD [13].
Potential mechanisms of the beneficial effects of butyrate
The vast majority of SCFAs produced in the colon are metabolized locally by colonocytes, where they function not only as the predominant energy source but also as key modulators of metabolic, immunological, and anti-inflammatory pathways; only a minor fraction is excreted in feces [11]. Among these SCFAs, butyrate is particularly crucial, providing 70–80% of the colonocyte’s energy demands [14]. Beyond its metabolic role, it exerts diverse regulatory effects on epithelial barrier integrity, gut microbial ecology, and intestinal motor and sensory functions, collectively contributing to the attenuation of chronic inflammation and the alleviation of clinical manifestations [15]. A comprehensive understanding of the complex, multidirectional mechanisms through which butyrate mediates these effects is therefore fundamental to evaluating its therapeutic potential (Figure 1).
Figure 1
Potential beneficial role of butyric acid in UC C4H8O2 – butyric acid, Na+ – sodium ion, Cl– – chloride ion, K+ – potassium ion, TLR – Toll-like receptor; IKKα, IKKβ, and IKKγ – subunits of the IκB kinase (IKK) complex, NF-κB complex – nuclear factor kappa B complex, p50 – NF-κB p50 subunit, p65/c-Rel – NF-κB p65/c-Rel subunits, IκB – inhibitor of NF-κB, P – phosphorylation of IκB, Ub – ubiquitination of IκB, TNF-α – tumor necrosis factor.
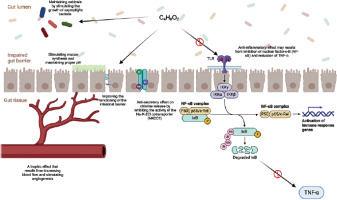
Anti-inflammatory effects of butyrate
The potent anti-inflammatory properties of butyrate stem from its ability to modulate immune responses and support intestinal epithelial barrier integrity. At the molecular level, butyrate exerts broad anti-inflammatory effects by downregulating the expression of pro-inflammatory cytokines and genes involved in inflammatory signaling pathways. A central mechanism of its immunomodulatory action involves the activation of G protein–coupled receptors (GPR41, GPR43, and GPR109A) expressed on T lymphocytes, macrophages, and intestinal epithelial cells [16]. Receptor activation promotes regulatory T-cell (Treg) differentiation and activity, increases the production of anti-inflammatory cytokines such as interleukin-18 (IL-18), and helps maintain the Th17/Treg balance, thereby attenuating the pro-inflammatory responses characteristic of active IBD [17, 18].
Another well-established mechanism is the epigenetic effect of butyrate mediated through the inhibition of histone deacetylase (HDAC). HDAC inhibition suppresses the expression of pro-inflammatory genes – including cytokines, chemokines, and adhesion molecules – while enhancing the transcription of genes involved in epithelial repair and regeneration [19]. It also contributes to downregulation of the nuclear factor-kB (NF-kB) pathway and blockade of interferon-g/signal transducer and activator of transcription (IFN-g/STAT) signaling in intestinal epithelial cells, thereby limiting the propagation of inflammatory cascades [20, 21].
Collectively, these effects reduce the production of key pro-inflammatory mediators, including interleukin-1b (IL-1b), tumor necrosis factor-a (TNF-a), IL-2, IL-8 and IL-23A, while modulating the expression of regulatory cytokines such as IL-10. In parallel, they decrease the expression of Toll-like receptor 4 (TLR4) and adhesion molecules, including intercellular adhesion molecule and vascular cell adhesion molecule-1 (VCAM-1), ultimately restricting the recruitment of inflammatory cells to the intestinal mucosa [22].
The anti-inflammatory effects of butyrate are further reinforced by its antioxidant properties. Butyrate enhances endogenous antioxidant defenses including antioxidant enzymes, thereby reducing oxidative stress and modulating cytokine expression in intestinal epithelial cells [22] (Table I).
Table I
Anti-inflammatory and immunomodulatory mechanisms of butyrate in the intestinal tract
[i] IL – interleukin, TLR-4 – toll-like receptor, TNF-a – tumor necrosis factor a, COX-2 – cyclooxygenase-2, MCP-1 – monocyte chemoattractant protein-1, CXCL – C-X-C chemokine receptor, TGF-b – transforming growth factor b, SOD-2 – superoxide dismutase 2, NF-kB – nuclear factor kappa-light-chain-enhancer of activated B cells, STAT1 – signal transducer and activator of transcription 1, PPAR-g – peroxisome proliferator-activated receptor g, TEER – transepithelial electrical resistance, IgA – immunoglobulin A
Effects on the intestinal epithelium and mucosal barrier
The intestinal barrier – comprising the epithelium, mucus layer, and tight-junction complexes – serves as the primary defense against pathogen and antigen translocation into the lamina propria. Adequate energy supply to colonocytes is essential for maintaining epithelial integrity and supporting proliferation, differentiation, and repair during active inflammation [23]. Butyrate deficiency impairs colonocyte metabolism, intensifies oxidative stress, and disrupts epithelial integrity. Evidence indicates that butyrate upregulates the expression of tight junction–associated proteins including claudins, occludin, and zonula occludens-1, thereby strengthening the mucosal barrier and reducing the excessive intestinal permeability characteristic of IBD (‘leaky gut’) [24]. Butyrate also stimulates mucin production in goblet cells (particularly mucin 2 (MUC2)), the principal component of the protective mucus layer, and enhances the synthesis of secretory immunoglobulin A (IgA) and antimicrobial peptides. Collectively, these actions limit bacterial and toxin translocation into the lamina propria, thereby reducing immune activation and attenuating mucosal inflammation [16, 22, 25].
Effects on the gut microbiota
Butyrate also modulates the composition and function of the gut microbiota. Exogenous supplementation promotes the expansion of butyrogenic bacteria, thereby enhancing endogenous SCFA production and improving the luminal environment [26]. By lowering the luminal pH, butyrate inhibits the growth of pathogenic and opportunistic microorganisms, supporting the restoration of microbial balance [23]. Studies have shown that butyrate supplementation increases microbial diversity and promotes the growth of anti-inflammatory bacteria such as Faecalibacterium prausnitzii [27]. These effects collectively facilitate the re-establishment of eubiosis, which is essential for maintaining immunological homeostasis. A summary of the key anti-inflammatory and immunomodulatory mechanisms of butyrate within the intestine is presented in the Table I [22].
Trophic effects
In the context of IBD pathogenesis, the trophic properties of butyrate are particularly relevant, as they accelerate mucosal regeneration and promote healing of inflammatory lesions. This derives from butyrate’s ability to stimulate deoxyribonucleic acid (DNA) synthesis in intestinal epithelial cells, enhancing their proliferative and reparative capacity [15, 28]. Additionally, butyrate increases local blood flow through effects on arteriolar smooth muscle and activation of vascular receptors, improving tissue oxygenation and nutrient delivery [29]. It also promotes angiogenesis and restoration of the mucosal microcirculation within damaged tissue [30], ultimately facilitating faster ulcer healing and re-establishing intestinal barrier integrity [31].
Regulation of visceral sensitivity and gastrointestinal motility
Butyrate also plays a role in modulating gut–brain axis functions. Acting on the enteric nervous system, it influences gastrointestinal motility and visceral sensitivity. Animal studies show that butyrate administration in healthy subjects induces visceral hypersensitivity without inflammatory changes [32]. Similar findings have been reported in the trinitrobenzenesulfonic acid–induced colitis model, where butyrate exacerbated hyperalgesia without affecting inflammation severity [33]. In contrast, human studies demonstrate that local administration of physiologically relevant doses of butyrate increases intestinal wall compliance while reducing visceral pain perception [34]. Moreover, butyrate has also been shown to improve colonic smooth muscle contractility and neurotransmitter metabolism, ultimately supporting enhanced colonic motility [35].
Inhibition of neoplastic transformation
A growing body of evidence indicates that butyrate can inhibit neoplastic transformation in colonic epithelial cells. This effect is primarily mediated through HDAC inhibition, leading to activation of tumor suppressor genes and induction of apoptosis in cancer cells [36–39]. These actions contribute to cell-cycle arrest and limit the proliferation of dysplastic cells [30]. This phenomenon, known as the ‘butyrate paradox,’ reflects that butyrate supports physiological proliferation and regeneration of normal colonocytes, yet in dysplastic or malignant cells activates epigenetic pathways that suppress their growth [11, 36–38]. Butyrate also engages additional mechanisms relevant to mutagenesis. It reduces oxidative stress and the generation of reactive oxygen species, thereby lowering the risk of mutation formation [40]. By modulating the tumor microenvironment, butyrate attenuates inflammatory responses and the production of pro-inflammatory cytokines, reducing dysplastic progression [41]. In addition, it strengthens intestinal barrier integrity, limiting epithelial exposure to luminal pro-carcinogenic factors [14].
Given the broad spectrum of beneficial effects attributed to butyrate, maintaining adequate intraluminal levels is critically important. In healthy individuals, a balanced diet typically meets physiological requirements; however, in patients with IBD, demand increases due to intensified mucosal repair. Direct supplementation with free butyric acid is limited by its instability, strong odor, and lower bioavailability. For this reason, its salt form – SB – is used, providing greater stability, a solid form, and markedly reduced olfactory intensity [11]. Orally administered butyrate is, however, rapidly absorbed in the upper gastrointestinal tract, thereby limiting its delivery to the distal segments of the intestine, which are the predominant sites of inflammatory involvement in IBD [11]. To enhance colonic bioavailability, microencapsulation techniques have been developed, embedding SB within lipid microgranules dispersed in a triglyceride matrix [12]. This formulation enables controlled distal release and improves stability, therapeutic efficacy, and patient acceptability [13].
Extensive research has explored the role of butyrate in IBD pathogenesis. In the 1980s, Roediger demonstrated that butyrate serves as the primary energy source for colonocytes and that its deficiency compromises epithelial integrity and repair – findings that helped establish early mechanistic concepts of the disease [28, 41]. These observations prompted subsequent experimental work and clinical trials evaluating butyrate-based therapies in IBD [42]. Early investigations focused largely on topical administration (rectal enemas) owing to the limited bioavailability and suboptimal efficacy of conventional oral preparations.
Therapeutic use of SB in ulcerative colitis
The first open-label, non-randomized clinical trials evaluating rectal enemas containing butyric acid or mixed SCFAs were conducted in the early 1990s. Although these studies included only small groups of patients (9–12), they were the first to demonstrate the efficacy of this therapeutic approach, showing meaningful clinical improvement and reductions in disease activity [42–48]. However, the results of subsequent controlled trials involving larger patient populations did not consistently confirm a clear advantage of these formulations over placebo [49, 50]. Notably, one study reported that a SCFA mixture (acetate, propionate, SB) administered as rectal enemas resulted in clinical improvement in patients experiencing relatively short disease exacerbations (< 6 months), provided that sufficient mucosal contact time was ensured [51]. Among patients who initially failed to respond to placebo, switching to SCFA enemas resulted in clinical and histological improvement in 65% of cases [51]. Another trial demonstrated that in patients with distal UC, a combination therapy with rectally administered mesalamine and butyrate was significantly more effective than rectal mesalamine alone. In this population, butyrate enemas were also shown to significantly reduce NF-kB activity in lamina propria macrophages, which was associated with decreased inflammatory activity and lower disease activity index scores [52]. In a subsequent multicenter trial published in 2003, topical butyrate as an adjunct to mesalamine improved clinical and endoscopic outcomes in patients with refractory distal UC, supporting its role as an effective and well-tolerated add-on therapy [53].
The first study evaluating the efficacy of oral SB in inducing remission in UC was published in 2000. Unprotected SB delivered in pH-dependent, polymer-coated capsules (4 g/day), when combined with mesalamine, enhanced treatment efficacy in patients with mild-to-moderate disease activity and demonstrated an excellent safety profile [54].
Favorable outcomes of a combined therapy with butyrate and mesalamine were also confirmed in a large, multicenter, open-label trial, involving over 200 UC patients with an incomplete response to mesalamine therapy. In this study, a modified-release formulation containing SB and inulin (0.9 g butyrate and 0.75 g inulin per day) was used. The combination regimen significantly reduced disease activity, as reflected by improvements in clinical symptoms and endoscopic appearance [55]. Although the lack of randomization and blinding limits definitive conclusions, the study provided valuable clinical evidence supporting the feasibility and potential benefit of oral butyrate as an add-on therapy and underscored the need for further trials.
More than a decade later, further development of this approach led to research using more advanced delivery systems. In 2020, a study evaluating microencapsulated sodium butyrate (MSB) as an add-on therapy to mesalamine for the maintenance of remission in UC was published. Despite several limitations (small sample size, lack of randomization, observational design), the results suggested that MSB may help sustain clinical and biochemical remission while improving quality of life and alleviating symptoms such as abdominal pain [56].
In 2022, an analysis of SCFA profiles in patients with IBD further characterized alterations in individual SCFA concentrations according to disease activity (remission vs active disease). The investigators found that patients with active UC – particularly those with high disease activity – had significantly lower butyrate levels compared with individuals in remission and healthy controls. A negative correlation was also observed between C-reactive protein (CRP) levels and C4 concentrations [57].
In a recent randomized, placebo-controlled trial conducted by Firoozi et al., the effects of MSB supplementation were evaluated in patients with active mild-to-moderate UC. The study demonstrated that MSB at a dose of 600 mg/day may serve as an effective adjunct therapy in this population, reducing inflammatory markers (fecal calprotectin (FC), hs-CRP), modulating the expression of circadian clock genes (CRY1, CRY2, PER1, PER2, BMAL1, CLOCK), and improving both sleep quality and overall quality of life [58].
Most recently, a multicenter, double-blind, randomized, placebo-controlled trial further evaluated MSB as an add-on therapy for induction of remission in adults with mild-to-moderate UC. In this study, MSB administered at 600 mg/day for 8 weeks demonstrated a significant therapeutic benefit, with 51% of patients achieving clinical improvement, 31% clinical remission, and 42% biochemical remission. Endoscopic improvement was observed in 25% of treated patients. Notably, among individuals achieving clinical remission, changes in fecal butyric acid (C4) strongly correlated with improvements in both endoscopic response and total Mayo score, underscoring the role of butyrate availability in mucosal healing [59].
In parallel, growing evidence indicates that the concentration of butyrate present in the intestinal lumen may be clinically relevant beyond its direct therapeutic effects. In a multicenter prospective study, fecal butyric acid (C4) concentration was identified as a strong, non-invasive predictor of endoscopic remission in UC, with significantly higher levels observed in patients achieving complete mucosal healing. Fecal C4 demonstrated excellent predictive performance and showed no significant correlation with FC, suggesting that it reflects distinct pathophysiological processes related to epithelial integrity and inflammatory control [60].
Taken together, these findings support the concept that fecal butyrate may serve as a complementary biomarker of mucosal healing and treatment response, and further position MSB as a promising therapeutic adjunct in UC, warranting continued investigation in larger and longer-term clinical trials.
Therapeutic use of SB in Crohn’s disease
To date, only a limited number of studies have evaluated the use of SB in CD. In an observational study by Di Sabatino et al., oral butyrate supplementation was assessed in patients with mild-to-moderate ileocecal disease. Administration of an enteric-coated formulation (4 g/day for 8 weeks) resulted in a clinical response in 9 of 13 patients (69%), including remission in 7 (53%) and partial improvement in 2. A statistically significant reduction in CDAI (Crohn’s disease activity index), NF-kB activity, and mucosal levels of the pro-inflammatory cytokine IL-1b was also observed [61].
Further evidence comes from a randomized, placebo-controlled trial by Facchin et al. In this study, 60-day supplementation with MSB (1800 mg/day) in patients with IBD (19 with CD, 30 with UC) favorably modulated the gut microbiota by increasing the abundance of SCFA-producing bacteria (Lachnospiraceae spp. in UC; Butyricicoccus spp. in CD). Additionally, a subjective improvement in quality of life was reported, more pronounced among patients with UC (p = 0.0284), despite the absence of significant changes in clinical disease activity (pMS, HBI) or FC levels [62].
This line of investigation was extended in the first multicenter, prospective, randomized, placebo-controlled trial conducted in a pediatric population. The study evaluated oral SB as an adjunctive therapy in children and adolescents (6–18 years) with newly diagnosed, colon-involving IBD (CD and UC). A total of 72 patients were enrolled and randomized to receive MSB (150 mg twice daily) or placebo for 12 weeks. At the end of treatment, the primary endpoint was not met, as no statistically significant differences were observed between groups in remission rates, median disease activity scores (PCDAI, PUCAI), or FC levels. The authors discussed several factors that may potentially account for the lack of clinical effect, including insufficient butyrate dosing, the pH-dependent microencapsulation technology (potentially affecting the site of release), the absence of defined therapeutic targets for distal colonic butyrate concentrations, and the relatively short follow-up period.
Despite the negative clinical outcome, the study provided important methodological insights into SCFA supplementation in IBD and underscored the need to optimize formulation strategies and to identify patient subgroups more likely to benefit from such interventions [63].
Taken together, the currently available clinical data suggest that SB supplementation in CD remains an experimental therapeutic approach. While mechanistic and microbiota-modulating effects are biologically compelling, robust evidence supporting its clinical efficacy in CD is still lacking and warrants confirmation in adequately powered, well-designed randomized trials.
Summary
SB is increasingly recognized as a promising adjunctive therapy in the management of IBD. Its broad biological actions – reinforcement of epithelial barrier integrity, modulation of immune and inflammatory pathways, and beneficial effects on gut microbial composition – provide a strong mechanistic rationale for its clinical application. By targeting key components of IBD pathophysiology, SB may complement existing therapeutic strategies rather than replace them. Current evidence suggests that butyrate supplementation may improve disease-related outcomes, particularly in UC, by attenuating inflammatory activity, supporting mucosal healing, and potentially enhancing responsiveness to treatment.
Beyond its therapeutic effects, emerging data indicate that fecal butyrate may serve as a clinically relevant, non-invasive biomarker of mucosal healing and endoscopic remission in UC. Its concentration appears to reflect pathophysiological processes related to epithelial integrity and inflammatory control, providing information complementary to established inflammatory markers.
However, the evidence base remains limited by the relatively small number of randomized controlled trials, heterogeneity in study design, and variability in formulations and dosing regimens. These limitations preclude definitive conclusions regarding its efficacy across different IBD phenotypes. Future research should therefore focus on well-designed clinical trials to more precisely define the therapeutic value of SB, establish optimal dosing strategies, and identify patient subgroups most likely to benefit from supplementation. In addition, further exploration of its potential synergy with established therapies, including biologics and small-molecule agents, as well as comprehensive assessment of long-term safety, will be essential. Overall, SB represents a biologically plausible, safe, and promising therapeutic adjunct in IBD management. While current data are encouraging, its precise role in clinical practice requires further definition and awaits further clarification through next clinical investigation and a more personalized approach to treatment select.













