PURPOSE
Psychomotor agitation (PMA) is described as increased motor activity (e.g. excessive gesturing) and emotional activation [1]. PMA is usually associated with a variety of psychiatric conditions, among which schizophrenia and bipolar disorder are the most prevalent [2]. Acute PMA requires timely identification, appropriate assessment and management to minimize anxiety for the patient and reduce the risk of escalation to aggression and violence. Recent studies suggest that PMA constitutes up to 10% of all emergency psychiatric interventions [3].
While the thalamus is made up of a series of nuclei which are classically known for their role as a sensory relay, it is also an important contributor in motor activity, emotion and memory control [4]. In most patients, it is supplied by several branches of the posterior cerebral artery (PCA) and posterior communicating artery. The artery of Percheron (AOP) is an anatomical variant present in only 4% to 12% of the population [5]. AOP originates only from one PCA and supplies simultaneously the right and left thalami [6]. One uncommon type of ischemic stroke is the occlusion of the AOP, leading to infarction of the bilateral paramedian thalami and mesencephalon [7].
In this case report we describe a rare instance of acute psychomotor agitation being the main symptom of bilateral thalamic ischemia caused by the occlusion of AOP.
CASE DESCRIPTION
A 59-year-old female with no past medical history was transported to the hospital at 8:26 AM, an hour and 36 minutes after she was found by her husband with altered mental status (slight confusion), loss of consciousness and a secondary paresis of the left limbs. She was last seen well the night before those symptoms occurred. According to the interview she has been smoking for 40 years. So far she has not been cardiologically treated or diagnosed. Due to the COVID-19 restrictions she recently lost her job as a shop assistant and had significantly lowered mood, though she did not see a specialist. Approximately an hour and a half from the onset of the symptoms her husband decided to call emergency medical services and the patient was admitted to the hospital emergency department (ED). During the first neurological examination she was conscious, and in proper, slightly delayed verbal contact. Following this there was significant variation in the patient’s temporal and spatial orientation, which significantly fluctuated during her stay at the ED. Psychomotor agitation was comprised of excessive motor activity – aimless wandering dominated. No verbal or physical aggression was observed. In a neurological examination no focal neurological deficit was observed (within the cranial nerves, motor, sensory and gait function). The patient’s metabolic panel was normal, including a normal blood glucose level. The patient was admitted in accordance with the “acute stroke code” in which computed tomography (CT) of the head and CT angiography (CTA) are routinely performed. In this case, the clinical picture did not clearly suggest a stroke diagnosis; however, the neurologist decided not to restrict the examination protocol only to a plain CT of the head. To perform the neuroimaging examination, the patient had to be administered midazolam 4 mg i.v. The imaging was completed 28 minutes after the patient’s arrival at the ED, and showed no evidence of intracerebral pathology (Figure 1). After excluding acute drug intoxication, transfer to a psychiatric unit was considered. However, a detailed CTA examination showed short stenosis of the proximal left PCA with preserved distal blood flow (Figure 2). A decision was made to perform magnetic resonance imaging (MRI) of the head to find out whether the observed left PCA stenosis was symptomatic or not. The MRI showed increased diffusion-weighted imaging (DWI), decreased apparent diffusion coefficient (ADC) and a negative FLAIR signal (DWI/FLAIR mismatch) within the periventricular white matter consistent with paramedian thalamic nuclei without midbrain involvement. Based on MRI findings, hyperacute (less than 4.5 h) bilateral ischemic infarction, consistent with the AOP occlusion, was diagnosed (Figure 3).
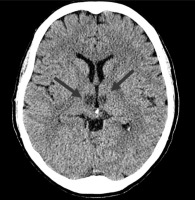
Figure 2
Computed tomography angiography: short stenosis of the proximal left posterior cerebral artery in 3D (A) and in front view (B)
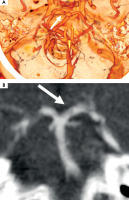
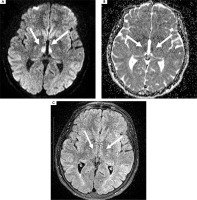
Figure 3
Magnetic resonance imaging of the head. Bilaterally increased diffusion-weighted imaging (A), decreased apparent diffusion coefficient signal within the thalamus (B). No visible changes seen on fluid-attenuated inversion recovery (C)
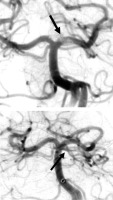
The patient qualified for intravenous thrombolytic treatment (total dose of rtPA – 54 mg) and mechanical thrombectomy (MT). Both procedures were performed in a time window for reperfusion therapies and resulted in complete recanalization of the left PCA and restoration of the blood supply (Figure 4). Due to the psychomotor agitation, MT was performed under general anesthesia. Eight hours after the MT, the patient was restored to proper mental and motor status. The period from onset to recanalization was covered with complete memory loss and the patient did not realize what happened.
Figure 4
Digital subtraction angiography showing thrombus within the left posterior cerebral artery (A) and preserved blood flow within the posterior cerebral artery and Perchron artery (B)
The patient was kept in the neurointensive care unit and the follow-up CT of the head performed the next day revealed hypodense ischaemic lesions in the medial parts of both thalami, with no signs of intracranial hemorrhage (Figure 5). On the third day of hospitalization a 2-dimensional echocardiogram with bubble study was done, which was negative. In echocardiography, normal myocardial contractility and mild regurgitation of bicuspid, tricuspid and aortic valves was found. Twenty-four hour Holter monitoring did not show any abnormal cardiac rhythms. Eventually, after nine days of hospitalization the patient was discharged in good general condition, with no neurological deficit. Antiplatelet therapy, absolute prohibition of smoking and an outpatient psychiatric consultation were recommended.
COMMENTS
Psychomotor agitation always needs an appropriate approach and thorough examination as it may evolve into severe aggression, thus preventing proper diagnosis [8]. There is also a discussion within the literature about what the word “agitation” actually represents, as there are various states that belong to that category. It has been established that it concerns patients with cognitive impairment presenting one of the following behaviours: excessive motor activity, verbal aggression, or physical aggression [9]. Medical professionals should remember that agitation is not always a sign of an underlying psychiatric disorder, especially when it concerns older patients. There are many factors that may cause that state, including intracranial hemorrhage, hypoxia, hypoglycemia, hyperglycemia, infection and intoxication [10]. The diversity of those contributors is definitely an impediment in finding a correlation between clinical manifestations and the neurobio-logical character of those changes. However, some advances in that matter have been reported. The amygdala and its innervation attached to other areas (for example the hypo thalamus) is said to regulate stress hormone release and that may play a substantial role in the occur-rence of acute agitation [11].
Bilateral thalamic stroke is a rare cause of stroke that occurs in less than 0.5% of all cerebral infarctions [12]. It is usually described by a triad of symptoms (altered mental status, vertical gaze palsy, and memory impairment) [13], but can also be manifested solely by the altered mental status – and under this name many different symptoms may occur, as the literature shows. Several studies reported hypersomnia, reduced self-awareness or even status epilepticus as the first sign of bilateral thalamic ischemia [7]. However, in the literature there are only few examples of psychomotor agitation among the symptoms of bilateral thalamic stroke [14], as was the case in the report given here.
From a psychiatrist’s point of view, bilateral thalamic strokes are often hard to diagnose because their symptoms may indicate other diseases, thus they are called “stroke chameleons”. There are many different kinds of such chameleons – dizziness, vertigo, headache, movement disorders and altered mental status [15]. The patho-physiology of altered mental status in the course of ischemic stroke is not entirely known. As for apathy, it was found that it occurs more often in pontine infarcts [16]. Strokes in a similar area – the ventral pons and lateral cerebellum – were also associated with a higher prevalence of poststroke agitation and aggression [17]. Interestingly, Botez et al. reported that agitation and aggression are more frequent in patients with PCA infarcts, which is in line with our report [18].
From a neurologist’s point of view psychomotor agitation with no focal neurological deficit and with a normal brain CT scan is usually an indication of the need for an urgent psychiatric consultation. However, agitation may be the main clinical manifestation of several “organic” conditions including encephalitis, Parkinson’s disease or Alzheimer’s disease [2]. Bilateral thalamic stroke usually presents with altered mental status and focal neurological dysfunction, with apathy or drowsiness as the unusual symptoms. In this case, there was an unusual situation of agitation being the main symptom of the bilateral thalamic stroke, caused by the occlusion of AOP. The diagnosis of thalamic stroke in this case was made somewhat accidentally, as a result of PCA stenosis, which prompted the neurologist to urgently extend the diagnosis to include DWI/MRI. Only a rapid diagnosis of acute stroke made it possible to use effective therapy.
As was stated above, agitation can be associated with many different medical conditions and, unfortunately, lead to the overlooking of a condition that requires rapid intervention.