PURPOSE
Subependymoma is a slow-growing benign brain neoplasm, classified by the World Health Organization (WHO) as a grade I tumor, which typically presents in middle-aged male adults [1]. It is most often located in the fourth ventricle of the brain and bulges into the ventricular lumen, causing an obstructive hydrocephalus. It may also arise from the lateral ventricles, septum pellucidum, or the central canal of the cervical spine. Subependymoma is generally diagnosed as an incidental finding at autopsy [2]. It is homogeneously hyperintense on T2-weighted images, and iso- to hypointense to grey matter on T1-weighted imaging with and without mild enhancement after gadolinium-based contrast administration [1]. The heterogeneity of signal intensity is associated with calcifications and cystic changes. Because of its distinct demarcation from adjacent brain tissue after surgical resection, tumor recurrence is uncommon [2].
Bruns syndrome is a neurological disturbance caused by a lesion in the ventricular system of the brain. It is characterized by attacks of sudden and severe headache accompanied by vomiting, vertigo and gait ataxia [3]. Symptoms occur with changes in head posture as the tumor is displaced, which leads to obstruction of the cerebrospinal fluid (CSF) pathway and transient intracranial pressure elevation due to episodic non-communicating hydrocephalus.
Here, we discuss the case of a patient with a histo-pathological diagnosis of subependymoma, who presented with episodes of Bruns attacks prior to undergoing total surgical resection of the tumor.
CASE DESCRIPTION
A 49-year-old male patient presented to the Department of Clinical Neurology with intractable daily headaches (VAS 2/10) that had lasted for a month with attacks of severe pain (VAS 8/10) upon sudden postural changes. The patient reported episodes of headaches, vertigo, and vomiting, lasting up to 30 minutes, occurring mainly in the morning when rising to an upright posture. The headache intensified during the day. His past history was insignificant for neuro-oncological diseases and associated risk factors. Initial neurological examination and lab findings were unremarkable. None of the neuro-psychological assessment test results indicated any impairment.
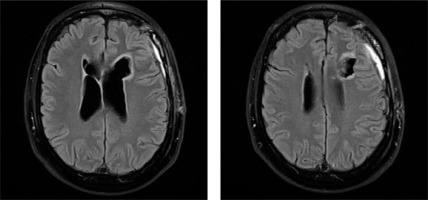
An MRI scan showed an irregular mass in the frontal horn of the left lateral ventricle and a nodular lesion with a cystic appearance in the frontal horn of the right lateral ventricle (Figure I). The maximum size of the cysts was approximately 46 mm, with a solid portion of 43 mm. The tumor was hyperintense with variable contrast enhancement. The tumor mass was shown to compress the foramen of Monro, resulting in an enlargement of the lateral ventricles.
Figure I
Subependymoma. Preoperative MRI image showing an intraventricular tumor arising from the ventricular wall. Axial post-contrast T1 imaging
Following clinical evaluation, a left frontal craniotomy and a complete tumor resection via the transcortical approach were performed. Pathological examination confirmed the diagnosis of a grade I pure subependymoma based on the WHO classification (Figures II-IV). Histological analysis of the resected tumor showed small nodular clusters and nests of ependymal cells embedded in a gliofibrillary matrix without atypical features. No nuclear pleomorphism, hyperchromasia or mitotic figures were seen. Immunohistochemistry was positive for glial fibrillary acidic protein (GFAP), while epithelial membrane antigen (EMA) was negative. The tumor was negative for synaptophysin and neurofilament proteins, but positive for minichromosome maintenance protein complex-2 (MCM-2) in 2-4% of cell nuclei.
Figure II
Hematoxyllin-eosin stain (100 ×) In neuropathological examination the tumor showed at low-power magnification clusters of round-to-oval shaped cells often separated by a matrix and microcystic areas (Figures II and III)
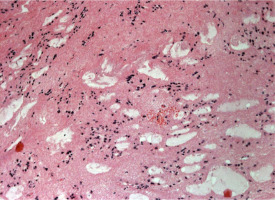
Figure III
Hematoxyllin-eosin stain (100 ×) In neuropathological examination the tumor showed at low-power magnification clusters of round to oval shaped cells often separated by a matrix and microcystic areas (Figures II and III)
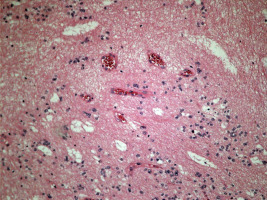
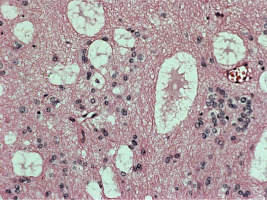
Figure IV
Hematoxyllin-eosin stain (200 ×) Microcysts filled with basophilic myxoid substance were observed at low- power magnification as they typically occur in subependymomas localized near the foramen of Monro
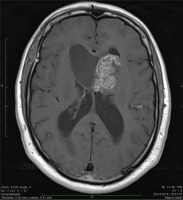
Follow-up imaging obtained 3 weeks after surgery demonstrated an irregular emptying postoperative cavity within the left frontal lobe and moderately dilated ventricular system. On the right side of the ventricle, below the frontal horn, there was an irregular fluid space with a maximum dimension of approx. 11 mm, most likely an infract cavity (Figure V). The patient’s postoperative functioning is currently stable. There are no reported or observed functional impairments or limitations at this time. The patient is displaying normal cognitive and physical abilities, and there have been no indications of any neuro-psychological deficits or abnormalities in their assessment. At the 4-year follow-up after the neurosurgical operation imaging was unremarkable. It is essential to continue monitoring the patient’s progress and conducting regular follow-up assessments to ensure his ongoing well-being.
DISCUSSION
Bruns syndrome is commonly observed in patients with neurocysticercosis and is rarely caused by a tumor [3]. Classic symptoms may not always be present, as observed in a recently reported case of Bruns syndrome in a patient with a third ventricle tumor who initially presented with a sudden disturbance of consciousness. Subependymoma is a rare, non-invasive tumor of an ependymal origin, comprising less than one percent of all central nervous system (CNS) neoplasms and is commonly discovered incidentally during neuroimaging or autopsy [4]. The diverse radiological characteristics and clinical manifestations are dependent on the tumor’s location and size. Patients may present with signs of intracranial hypertension due to obstructive hydrocephalus, epilepsy, or myelopathy [5].
Lateral ventricle tumors and fourth ventricle tumors can be distinguished from each other on the basis of their anatomical location, prevalence, and symptoms. The incidence of lateral ventricular tumors accounts for approximately 0.8% to 1.6% of all intracranial lesions [10]. On the other hand, tumors of the fourth ventricle represent 1-5% of all intracranial lesions [6]. The most common tumor types in both locations are ependymomas [7-9].
Specific symptoms associated with lateral ventricle tumors may include changes in personality/behavior, cognitive impairments, hemiparesis (weakness on one side of the body), and seizures if the tumor irritates the surrounding brain tissue [11-14].
Specific symptoms of fourth ventricle tumors may include cerebellar damage, particularly ataxia, intentional tremor, cerebellar dysarthria, and dysfunction of the cranial nerves (from V to XII) [6]. However, both types of tumors may also exhibit similar symptoms such as hydrocephalus, headache, nausea, and vomiting due to blockage in the ventricles, which hinders the free flow of cerebrospinal fluid and increases intracranial pressure. It is important to note that the manifestation of symptoms can vary depending on factors such as the size, location, growth rate of the tumor, and the individual characteristics of the patient.
Neurocysticercosis and other CNS neoplasms should be included in the differential diagnosis. Differential diagnosis of tumors located within the ventricular system includes ependymoma, subependymal giant cell astrocytoma, central neurocytoma, intraventricular meningioma, choroid plexus papilloma, choroid plexus carcinoma, and secondary metastasis to the brain [2]. Although no radiological features are pathognomonic for a subependymoma, careful evaluation of the patient’s demographic characteristics and clinical presentation are helpful in aiding the diagnosis [1]. Ependymomas are primarily located within the fourth ventricle, and display strong enhancement after contrast media administration, revealing calcifications. Subependymal giant cell astrocytomas are commonly located near the foramen of Monro with calcifications and intense contrast enhancement [2]. These tumors are seen almost exclusively in young patients with tuberous sclerosis.
Differences between tuberous sclerosis complex (TSC) and intraventricular tumors in clinical manifestations can arise due to various factors. TSC affects diverse organs, including the brain, skin, heart, kidneys, and lungs. Consequently, clinical manifestations in TSC encompass a wide array of symptoms, such as seizures, intellectual disability, skin abnormalities (e.g., facial angiofibromas), cardiac abnormalities, renal tumors, and pulmonary complications. In contrast, intraventricular tumors primarily impact brain function and are typically associated with neurological symptoms including headaches, seizures, hydrocephalus (due to obstruction of cerebrospinal fluid flow), cognitive impairment, behavioral changes, motor deficits, or sensory abnormalities. Intraventricular tumors have the potential to grow and cause complications due to their size and location. In contrast, TSC-related symptoms, such as seizures or cognitive impairments, can persist and fluctuate over time, with or without changes in tumor size. There are three main anatomical features associated with TSC that alter the structure of the brain: cortical tubers, subependymal nodules (SENs), and subependymal giant cell astrocytomas (SEGAs).
Cortical tubers generally form on the surface of the brain but may also appear in its deep areas. They are characterized by undifferentiated and dysfunctional cells, comprising both neurons and glial cells; these form dense masses, disrupting the organized structure of the cortex and functional connections between brain regions.
Subependymal nodules (SENs) are common in individuals with TSC. These benign tumors develop along the ependymal lining of the lateral ventricles and vary in size. SENs are composed solely of highly disorganized and dysfunctional glial cells, and they form during early brain development, similarly to cortical tubers. Unlike tubers, SENs can exhibit growth that exceeds the surrounding tissue, leading to their protrusion into the ventricular spaces.
Subependymal giant-cell astrocytomas (SEGAs) are large, benign tumors that arise from SENs. They pose a significant risk due to their potential for continued growth, especially in children and adolescents. Although the reasons are unclear, the development of SEGAs decreases significantly after adolescence. In rare cases, SEGAs can obstruct the flow of cerebrospinal fluid through the narrow passages of the lateral ventricles, leading to hydrocephalus [15, 16].
Central neurocytomas are usually identified in the anterior parts of the lateral ventricles and are often found in patients under 40 years of age. Neurocysticercosis changes observed in neuroimaging scans can reveal cyst morphology, localization, stage, and the presence of inflammation. Live vesicular cysts appear as small, rounded lesions without edema and may display the tapeworm scolex. Degenerative cysts (colloid cysts) exhibit poorly defined borders, edema, and noticeable contrast enhancement. Diffusion-weighted imaging aids in identifying the scolex in colloidal cysticerci. Calcified cysticerci appear as hyper-dense nodules on CT scans. Subarachnoid neurocysticercosis can lead to hydrocephalus and may be associated with the occlusion of ventricular foramina and arachnoiditis. MRI is the preferred imaging modality for assessing intraventricular, cisternal, and spinal cysticercosis. The differences between neurocysticercosis and subependymal tumor are tumor mass appearance, location and growth potential. Subependymal tumors present as solid masses within the ventricular system and do not exhibit cystic features like neurocysticercosis. Subependymal tumors are commonly found in the walls of the lateral ventricles, while the location of neurocysticercosis cysts can vary. Neurocysticercosis cysts generally do not demonstrate significant growth potential once they reach the degenerative stage. Subependymal tumors have the potential for continued growth, especially in children and adolescents [17].
When neuroimaging reveals a pathological mass adhering to the choroid plexus, tumors such as choroid plexus papilloma and carcinomas, secondary CNS meta stases, and meningiomas should be considered [5]. Nevertheless, it is often difficult to differentiate between a benign choroid plexus papilloma and a malignant choroid plexus carcinoma. Blockage of CSF flow may be temporarily relieved by ventriculostomy or ventriculoperito-neal shunts. Emergency surgical removal of the tumor is warranted in patients with Bruns syndrome.
COMMENT
The case report presented Bruns syndrome caused by the obstruction of CSF flow by an intraventricular subependymoma. The patient’s account of symptom onset was well-documented, indicating a clear understanding of the temporal relationship between the clinical manifestations and the day of admission. The reported symptoms, encompassing pain and dizziness, were observed to have a notably brief duration. This concise timeframe can be attributed to the underlying pathophysiology involving the obstruction of CSF flow within the lateral ventricles.
The importance of diagnostic brain imaging cannot be overstated, as it is crucial for patients presenting with paroxysmal vertigo and neurological disturbances. Surgical removal of the subependymoma is the preferred treatment approach, leading to a favorable prognosis. Regular follow-up visits are necessary to monitor the patient’s recovery and assess any potential complications. A baseline postoperative MRI is typically performed within the first few weeks after surgery to evaluate the extent of tumor resection and assess the postoperative changes. In the initial postoperative period, follow-up MRI scans are often performed at intervals of 3 to 6 months to monitor for any signs of tumor recurrence or residual tumor growth. Once the patient’s condition stabilizes and there is no evidence of recurrence, the frequency of follow-up visits and imaging studies may be decreased. Typically, annual or biennial MRI scans are recommended during the first few years after surgery.
In addition to imaging, regular clinical evaluations are essential to monitor the patient’s neurological status and overall well-being. WHO Performance status can be used in assessment. It is crucial to educate subependymoma patients about the signs and symptoms of potential recurrence or complications. The follow-up strategy should be tailored to each patient’s specific circumstances, including their age, overall health, tumor characteristics, and surgical outcomes. The frequency and duration of follow-up visits may vary based on these factors.