INTRODUCTION
Carpal tunnel syndrome (CTS) is a common upper limb nerve entrapment syndrome which carries signifi- cant economic and social burden to the affected indivi- duals. Its main feature is a reduction of the space inside the carpal tunnel thus interfering with the spatial rela- tionship between its contents, causing symptoms distal to the lesion site. This paper aims to present the recent find- ings in the scope of CTS treatment, and propose a direc- tion for further clinical trials.
METHOD AND RESULTS
PubMed was searched for articles published between January 1, 2016 and March 23, 2021 using the phrase “Car- pal tunnel syndrome” as the MeSH phrase. The search result was filtered by selecting the conditions “meta- analysis”, “systematic review”, and “randomized con- trolled trial”. We searched for epidemiology, risk factors, etiology, clinical features and treatment. All full-text, English language articles were included. Altogether 229 articles were identified, of which 72 were selected with the aim to give a comprehensive review of current CTS treatment methods.
EPIDEMIOLOGY
The annual incidence of CTS is ranging from 428 in women and 182 in men per 100,000 adults [1] with the prevalence of 3-5% in general population, raising up to 6% in women aged over 40 [2]. CTS severity may vary greatly between patients, from mild symptoms such as transient paresthesia or dysesthesia to nocturnal pain, sleep disturbance, muscle weakness and constant pain. Electrodiagnostic studies are conducted to confirm the diagnosis, with ultrasound imaging adding value to electrodiagnostic tests [3]. Tensor diffusion magnetic reso nance imaging (MRI) is also a valuable option for CTS diagnosis [4].
PATHOGENESIS
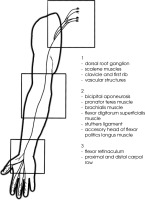
Studies have shown ischemia, edema, intraneural and extraneural fibrosis of the affected nerve in CTS [5]. A prolonged exposition to these factors changes axonal transport and consequently the excursion of the nerve may become impaired [6]; eventually demyelination and axon degeneration may occur. The entire process of de- myelination alongside with changes in the quantity and regulation of ion channels is implicated with spontaneous ectopic generation of action potentials [7, 8]. These sites are called AIGS (abnormal impulse generating sites). Neuroinflammation can occur beyond the site of com- pression. It is also present in the spinal root ganglia and may explain why symptoms occur not only at the ana- tomical sites of nerve innervation but also in other areas innervated by the nerves originating from the same nerve root. The bilateral presentation of symptoms occurs in 60% [9] to 74% [10] patients with CTS, which may be caused by the inflammatory response of glial cells that can spread to the spinal cord and the contralateral dorsal root ganglion (DRG) [11]. This distant response may be associated with the changes present in the central ner- vous system observed in CTS [12]. In more severe cases of neuropathic pain, the extraterritorial spread of symp- toms is more prevalent (Figure 1).
PHYSIOLOGY AND PATHOPHYSIOLOGY
Dorsal root ganglion
The dorsal root ganglion (DRG) is a crucial struc- ture for sensory transduction and modulation, includ- ing pain transmission [13], because it houses the somata of the primary sensory neurons (PSNs). Each DRG con- sists of up to 15,000 PSNs cell bodies and approximately eight-fold more glial cells [14]. In contrast to periphe ral nerves, the DRG are not provided with a blood-nerve barrier, which makes them prone to irritation by bio- chemical agents.
The connective tissue of the nerve
The endoneurium envelops a singular axon and pro- vides it with constant endoneural environment and fluid pressure gradient. The perineurium envelops a few to a few hundred axons making a fascicle. The epineurium’s main goals are to biomechanically protect the nerve from mechanical deformation due to compression or elonga- tion, and allow for the gliding of the nerve trunk among the surrounding tissues and the gliding of fascicles inside the nerve trunk. The main goal of the mesoneurium is to provide enough mobility between the nerve trunk and the surrounding tissues. It also supports the extrinsic blood vessels and attaches the nerve trunk to the sur- rounding tissues.
Axonal transport
Axonal transport is an energy-dependent system that transports many necessary substances synthetized in the cell somata to the terminal parts of the axon (ante- rograde) and vice versa (retrograde). These substances include neurotransmitters and cytoskeleton components. The prolonged (eight hours) compression of just 20 mmHg can reduce fast anterograde axonal transport and two- hour compression of 30-50 mmHg effects in a block of axonal transport, which is reversible in 24 hours. In this case, two hours of 200 mmHg compression was re- versible within three days [15]. Retrograde transport is also dose-dependent and can be blocked or limited via nerve compression from 20 mmHg [16].
Blood supply
Each fascicle is segmentally supplied by branches from epineural vessels. Two diffusion barriers, i.e., blood- nerve barriers, are located in the perineurium and the endoneurium, providing a homeostatic endoneural envi- ronment for uninterrupted axonal transport and nerve impulse conduction. Damage to these barriers, e.g., mechanical compression, may cause intraneural patho- physiological changes and impair nerve function [17]. A nerve elongation of 8% results in a slowing of intraneu- ral blood flow while 15% elongation causes complete ar- rest of intraneural blood flow. A return of blood flow is dependent on time and severity of hypoperfusion [18, 19].
Compression and elongation
Nerve conduction is limited by 70% in the case of one hour 6% elongation of the nerve while a complete conduc- tion block occurs with a 12% one-hour elongation [20]. If the nerve is no longer elongated, then nerve conduction should return to normal but it is dependent on the dose of elongation [21]. Two-hour compression by pressures of 150 to 200 mmHg produces a transient increase in pre- cursors to neuropeptide Y (CPON – C-terminal flanking peptide of neuropeptide Y) in DRGs. This supports the con- cept that local nerve compression activates the entire neu- ron [22]. Chronic compression results in the prominent upregulation of CPON and other neuropeptides [22, 23]. Intraneural structural changes, such as hypoperfusion and demyelination of the nerve, occur on the borders of com- pression similar to tourniquet compression.
Pressure gradient
The pressure gradient is the specific ratio of pressure between the tunnel of the nerve (PT), capillary pressure (PC), arterial pressure (PA), venous pressure (PV), and endoneural pressure (PF). According to Sunderland, the normal gradient is PA > PC > PF > PV > PT [24]. In the case of even mild compression of 20 mmHg, the intraneural pressure can rise due to impaired axonal transport and blood flow. In healthy people, the pressure inside the carpal tunnel is approximately 2.5 mmHg in the neutral position. It is 30-50 mmHg in the neutral po- sition in CTS patients. This increases to 90-110 mmHg in wrist carpal and dorsal flexion and can reverse the pres- sure gradient. The nocturnal occurrence of symptoms, common in CTS, is associated with a lower blood pres- sure in arteries and veins because the tissue pressure for nerve fiber viability is 30 mmHg below the diastolic blood pressure.
Nerve innervation
Connective tissue of peripheral nerves is innervated by nervi nervorum. These thin, unmyelinated fibers termi- nate in free endings in the connective tissue of the nerve sheath. They have been shown to contain neuropeptides such as substance P and calcitonin gene-related pep- tide (CGRP) [25]. These fibers respond to a nociceptive stimuli such as pressure applied on the nerve trunk, in- creased intrafascicular pressure, and increased tension. In the case of nerve trauma, the nervi nervorum can cause neurogenic inflammation due to release of CGRP and substance P.
RISK FACTORS
CTS is considered to be an idiopathic condition, how- ever there are factors which increase the risk of its occur- rence. These can be divided into four categories: extrin- sic factors altering volume of tunnel contents, extrinsic factors altering the cross section of the tunnel, intrinsic factors and neuropathic factors [26] (Table 1).
Table 1
Carpal tunnel syndrome risk factors
CONSERVATIVE TREATMENT
Acupuncture and acupressure
Acupuncture and acupressure are the supportive therapies based on Chinese medicine. Acupuncture uses needles to alleviate symptoms while pressure is applied to the affected areas in acupressure, without the use of needles. Electroacupuncture uses electrical current that passes between two needles. PC7 or the Daling point is especially considered when managing CTS; it was found to cause an increase of endomorpin-1, beta-endorphin, serotonin, dopamine, and enkephalin levels in the brain tissues and plasma [27]. Acupressure was found to be more effective compared to the placebo and splints re- garding the functional status and nerve conduction studies [28]; however, another study suggests that acupuncture with splinting does not produce better results than splinting alone [29] or has no effect compared to the placebo or sham intervention [30]. Acupuncture was shown to be more effective than ibuprofen in improving the functional status and better than splinting alone [31]; acupuncture with splint was found to be better than ce- lebrex with splint in improving symptoms and distal motor latency [32]. The basic science of acupuncture is contrary to the Western medical knowledge as it is root- ed in the traditional Chinese medicine. Further research should continue to develop our knowledge of the mecha- nisms of acupuncture treatment in relation to contempo- rary medical knowledge.
Extracorporeal shock wave therapy
Extracorporeal shock wave therapy (ESWT) is a tran- sient sequence of acoustic pulses with high peak pressure (100 MPa) followed by negative pressure (5-10 Mpa) with energy density between 0.003-0.89 mJ/mm2 [33]. Based on the design of the appliance, ESWT can be classified as focused or radial ESWT. These differ in depth of pene- tration, focusing of energy, and intensity. When it comes to syndrome alleviation and functional recovery in CTS, the radial ESWT was shown to be superior to the focused ESWT [34]. Radial ESWT produces better results for pa- tients with moderate CTS than those with mild CTS [33]. Studies show that ESWT is more effective than sham therapy and wrist splint [35, 36]. No difference was found in comparison with CSI in one meta-analysis [35], how- ever another study showed that ESWT was better than CSI in improving pain and distal motor latency [37]. Regarding the management of nerve injuries, ESWT pro- motes axonal regeneration of peripheral nerves through a cascade of molecular reactions improving conduction velocity and amplitude [38, 39]. ESWT safety remains controversial because of a few animal studies in which damage to myelin sheaths was found after high intensity therapy[36]. ESWT seems to be promising but further re- search is needed, especially considering safety issues.
Hydrodissection
Hydrodissection is a minimally invasive alternative to steroid injection [40]. It is a procedure of injecting hyaluronidases into anatomic spaces to facilitate dissec- tion and adhesiolysis. In the case of CTS, fluid is injected between the median nerve and transverse carpal ligament and underlying tendons. Hyaluronidases can depolarize hyaluronic acid leading to its degradation [41]. The ratio- nale behind this treatment is that hyaluronidase injection may interrupt adhesions and facilitate resorption of ex- cess fluid and blood [42], thus reducing the symptoms. Hyalase + 10 ml saline hydrodissection was shown to be more effective than 10 ml saline hydrodissection alone in improving VAS, functional disability score, and nerve conduction [41]. Another study has shown that 5 ml ultrasound guided saline hydrodissection is more effective than 5 ml ultrasound guided subcutaneous injection [43], proving the relevance of application site.
kinesiology taping
Kinesiology taping is a non-invasive method of treat- ment developed to provide an external, dynamic support and protection to a specific area of the body. The appli- cation of kinesiology taping varies depending on its aim but overall it consists of applying adhesive, elastic tape with certain degree of stretch over a specific area in cer- tain position of the body part [44]. According to Aktürk et al., the application of kinesiology taping in CTS can alleviate symptoms by reducing pressure on the nerve by lengthening the transverse carpal ligament, regulating edema, and improving blood flow and lymphatic circu- lation [45]. Kinesiotaping with splinting was shown to be more effective than splinting alone in symptom reduc- tion, improving functional status score and resulting in electrophysiological improvement in two RCTs [45, 46], but another RCT did not show benefits of KT and splint use over placebo KT, splint or splinting alone [47]. Fur- ther research regarding kinesiotaping should focus on proving this initial hypothesis in connection with MRI or Doppler ultrasound studies.
Corticosteroid injection
Corticosteroid injections (CSI) are a common treat- ment for CTS. The injection can be guided by ultra- sound imaging or by landmarks [48]. RCT found that US-guided injection was superior to landmark injection in improving BCTQ score, functional status [49], symp- tom severity [48], and nerve conduction velocity [50] while other RCTs produced the opposite results [51, 52]. The goal of the procedure is to deliver corticosteroid near the median nerve inside the carpal tunnel. When it comes to the application above or under the median nerve, the effects of the US-guided injection, proximal and dis- tal injections [53] were the same [54]. Injections are rel- atively safe, but they provide only short-term relief [55]. The most frequent adverse effects are pain in the hand or a region of injection and possible infection. Alleviation of symptoms is obtained mainly through reducing ede- ma, and improving spatial relation between the median nerve and tendons [56]. The improvements caused by an injection are short-term (> 6 months) [57]. Compared to other interventions, CSI shows better outcome to splint alone at 6 weeks [58] and injection and splint shows bet- ter outcome than splint alone [59], however the evidence is not homogenous [60]. Adding an injection during sur- gery was proven as effective as surgery alone [61]. CSI should be considered as a supportive treatment, especial- ly as a modality to alleviate severe symptoms.
Platelet-rich plasma injection
The extracted plasma portion contains a high con- centration of platelets and growth factors that may play crucial roles in tissue regeneration [62]. Growth factors are effective in managing inflammation and promoting wound healing. PRP improved the clinical condition of the patients and was beneficial for patients with mild-to-moderate CTS [63, 64]. The in vivo studies of the effects of PRP injection demonstrated improved Schwann cell proliferation, function, and migration. PRP improved symptom severity scale [62, 65] but not func- tional status scale. VAS and nerve conduction studies were no different compared to controls [65]. Basic science research have found that PRP has modulatory effects on neuroinflammatory environment. They assist with tissue healing and promote axon regeneration [62, 65]. This can explain why PRP adjuvant to surgery improves hand grip strength to pre-operative levels in 6 weeks compared to surgery alone [66]. It is also hypothesized that growth factors released after PRP injection might alle viate nerve ischemia by protecting the blood-nerve barrier [63]. Compared to other treatment modalities PRP with splint- ing and 5% dextrose do not provide for a better functional recovery [67, 68], but reduce the cross sectional area of the median nerve [69]. Studies have shown that PRP and splint combination was more effective than splint alone [70]. PRP can be effective as an adjuvant to other therapies, promoting neural tissue regeneration.
Low level laser therapy
Low level laser therapy (LLLT) uses red or near-infrar ed lasers (wavelength 600-1070 nm). The rationale for the use of LLLT in CTS management is its ability to produce bio- physical effects on neural tissue that lead to nerve cell regeneration, peripheral nerve regrowth, and functional improvements [71]. In animal studies, LLLT has shown wound healing stimulatory properties by increasing col- lagen synthesis with utility in reducing edema forma- tion and inflammation [72]. Studies on rats have shown an increase in myelin production and reduction of ret- rograde degeneration of motor neurons. Other possible mechanisms include selective inhibition of nociceptive activation at peripheral nerves and improvement in blood circulation [73]. These effects can be potentially advantageous in CTS patients, but the search for this re- view provided no evidence to support any clinical effect of LLLT in treating CTS [74]; no difference compared to placebo [72, 75] or LLLT combined with splinting to splinting alone [71, 76]. Only one systematic review has shown that LLLT is more effective than placebo in very short term (< 5 weeks), following which the effect tends to subside [77]. LLLT, despite its low cost and low risk of adverse effects should be abandoned as a treatment for CTS.
Splints
Wrist splinting is the most common conservative me- thod used in the treatment of CTS [78]. Splint is an orthotic device applied externally, i.e., to the wrist of a CTS patient. The splint with a metacarpophalangeal unit is more effec- tive than thermoplastic wrist splint in pain reduction and DASH score with no difference in grip strength and pinch strength [78]. It can modify the structural and function- al characteristics of the neuromuscular and skeletal sys- tems [79]. The main rationale behind the use of splints in CTS patients is the prevention of extensive flexion and extension of the wrist that causes an increase in the pres- sure inside the carpal tunnel. Sixty days of using a stretch- ing splint has been known to reduce the need for surgery 2 to 1 compared to stretching and NSAIDs [80]. Splints are also commonly used after surgical retinaculum release where they theoretically provide safety regarding flexor tendon bowstringing to prevent nerve subluxation and thus reduce postoperative pain. However, the lack of or- thosis compared to removable orthosis and nonremovable orthosis produced no change in outcome up to 12 months after mini-open carpal tunnel release [81]. On the other hand, splints can increase the time needed to return to work and can cause unnecessary additional indisposition [81]. Because splints may reduce patient’s activity and perfor- mance of daily tasks, it is important not to rely on them as the only way of treatment and use them as a way to im- prove such issues as sleep disturbance. Gradual return to full functionality should be advised.
Manual therapy and neurodynamics
This review is limited to the methods based on the current standards recommended by the International Federation of Orthopedic Manipulative Physical Thera- pists Incorporated (IFOMPT). Manual therapy can im- prove the local range of motion and mechanosensitivity, reduce spinal cord hyperexcitability, and facilitate modu- lation of the central nervous system pain processing [82]. In various studies, groups that integrated joint mobili- zation performed better than the control group in pain reduction, subjective symptoms, and functional status [83, 84] but no difference was found in 2-point discrimi- nation [85]. Cadaveric studies discovered that neuro- dynamic techniques are effective on fluid dispersion in the median nerve at the level of the carpal tunnel, with no difference between sliding and tensioning [86]. The same results were found at the ankle level regarding the tibial nerve [87]. Nerve mobilizations can also facilitate intra- neural fluid dispersion at the level of the nerve root as demonstrated in the studies by Gilbert et al. Repetitive straight leg raise (SLR) induced a significant increase in longitudinal fluid dispersion in the L4 nerve root of hu- man cadaveric specimen [88]. These results offer a clini- cal mechanism for the reduction of intraneural edema, which is a clinical presentation of CTS. Neurodynamic techniques were found to induce median nerve excursion in vivo [89]. However, complex biomechanical effect in pe- ripheral nerves [90], along with biochemical changes may play a crucial role regarding the therapeutic effect. Cyclic stretching of nerves was found to promote axonal growth and increase activating transcriptor factor-3 (ATF3) [91]. Two RCTs found the neurodynamic techniques to be more effective than sham therapy [92] or no therapy [93] in improving nerve conduction, pain assessment, 2-point discrimination, symptom severity, and functional status, but a systematic review found no conclusive findings due to methodological limitations in the current body of re- search [94]. Manual mobilizations of the carpal bones were found to produce an increase in the carpal tunnel cross- sectional area and anteroposterior diameter in vivo [95] as well as in cadaveric studies [96]. Manual therapy and sur- gery had similar effectiveness when it comes to self-reported improvement of function, symptom severity, and pinch-tip grip force [97]. Although for the purposes of this review we only chose studies following the IFOMPT standards, it is unclear which techniques or their dosage are effective. Manual therapy seems to work well with multifactorial conditions, such as CTS, but the current body of knowl- edge does not provide any clear evidence for the most effective time or area of its application.
Ultrasound and phonophoresis
The ultrasound device converts electrical energy to the piezoelectric acoustic wave, which is then absorbed by tissues of varying impendency to impact heat genera- tion in different tissues. Phonophoresis combines ultra- sound via topical anti-inflammatory drugs. Sound waves enhance cutaneous drug absorption [95]. Ultrasound in CTS may have beneficial effects because it stimulates nerve regeneration, increases nerve conductivity, and reduces inflammatory processes [99]. Analgesic effects may be related to increased tissue metabolism and capillary per- meability and also thermal effects thus increasing the pain threshold [100]. Despite these findings, the studies exa- mined show no difference between US and phonophoresis in the treatment of CTS [98] and no difference between continuous, pulsed, or sham US combined with splinting [101, 102]. US and phonophoresis do not provide any ad- vantage in the treatment of CTS and thus their application should be reconsidered.
CONCLUSIONS
CTS is common condition that affects women more often than men, with variable clinical presentation, often including sensory and motor symptoms distal to the le- sion site. Based on the current body of knowledge, it has been concluded that the conservative CTS treatment is very safe, but the effectiveness of the methods reviewed in this paper differs. Promising first results motivate the need for further research on novel treatment meth- ods but more conclusive and repeated research results are needed to provide a clinical framework for patient alloca- tion and decisions regarding the treatment choices.