1. Guideline methodology
The clinical practice guidelines for management of Clostridium difficile infections in adults were developed by the task force of the Board of the Polish Gastroenterology Society. This document summarises recommendations for improved diagnosis and treatment of C. difficile infections (CDI) in adults. The definition of CDI includes symptoms, usually diarrhoea, and a positive stool test for C. difficile toxin, or detection of a toxin-producing C. difficile strain, or a typical endoscopic image, or pathology study confirming pseudomembranous colitis. In addition, for proper diagnosis, treatment, patient care, and prevention, the guidelines include recommended methods of infection control, patient management, and infection prevention. The panel includes systematic rating of strength and quality of evidence in compliance with the GRADE system (evaluation of rating, recommendation development, and assessment) (Tables I and II). A detailed description of the methods, background, and summary of evidence in support of each recommendation is provided in the full text of the guidelines.
Table I
Strength of recommendation statement according to the GRADE rating system
Table II
Quality of evidence according to GRADE
1.1. Scope and aims of the guidelines
1.1.1. Aims
Clostridium difficile is still the most common cause of healthcare-associated diarrhoea. It became the most important pathogen in hospitalised patients for several reasons. The main purpose of the guidelines is to identify the best methods for diagnosis and management, as well as the most effective, sufficient, and safe treatment regimens for CDI patients. We expect that application of the guidelines will translate into greater disease awareness, help reduce costs of differential diagnosis, and will contribute to appropriate CDI management.
1.1.2. Health problems discussed in the guidelines
The following CDI-associated health questions are covered in the guidelines:
What is the aetiology of CDI?
What has been the epidemiological situation of CDI in recent years?
What are the clinical symptoms of CDI?
What are the diagnostic criteria for CDI?
Which clinical tools should be considered in differentia diagnosis?
What is the proper management of CDI (including therapy, patient monitoring, isolation, and supportive care)?
1.1.3. Target patient population of the guidelines
The guidelines discuss management of adult patients (over 18 years of age) of both sexes with symptoms suggestive of CDI, with the diagnosis confirmed based on current criteria, regardless of the type or severity of symptoms. In addition, recommendations for managing CDI also include patients with recurrent infection.
1.2. Development of recommendation statements
The clinical practice guidelines are recommendation statements aimed at optimising patient care, based on the systematic review of evidence as well as evaluation of benefits and losses of alternative medical care [1].
The practice guidelines were developed by a multidisciplinary panel of experts in epidemiology, diagnosis, infection control, and treatment of adult CDI patients. Only original studies (optimally prospective, randomised, double-blinded, placebo-controlled studies) were incorporated for the development of the guidelines. In the absence of such studies, the evidence of lower quality was considered, including observational and retrospective studies (excluding case series and case reports), as well as systematic reviews and meta-analyses. The source data were found in the following electronic databases: PubMed, NCBI, Cochrane Library, Researchgate, and GoogleScholar, as well as in the recommendation statements and guidelines published on the websites of international scientific societies. The literature review and analysis were limited to articles published in either English or Polish.
The evaluation of the quality of evidence and the strength of recommendation statements was supported by the Grading of Recommendations Assessment, Development, and Evaluation system (GRADE) (Tables I and II). Delphi voting was implemented for the final decision making. The acceptance level for each recommendation statement was assessed by the expert panel on a five-point scale (A–E) and determined by the agreement level (Table III). Each recommendation statement was discussed taking into consideration the scientific evidence used for its formulation, covering the relationship between the guidelines and the scientific data. Based on the acceptance level, the consensus between experts was considered high when 80% or more of voters chose A or B categories. When the results were below 80%, the agreement level was deemed low.
Table III
The agreement level of votes
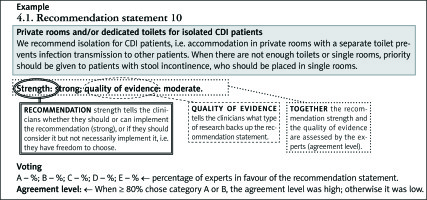
1.2.1. Recommendation statement interpretation
The graphic interpretation scheme is shown below. Each recommendation statement consists of three parts regarding its strength, the quality of evidence, and the distribution of the expert votes.
2. Epidemiology
2.1. Recommendation 1
Recommended definition of CDI
To monitor infection sites and to increase comparability between analyses, the widely available standard definitions of primary infection should be used, including: (1) healthcare-onset (HO) CDI – the site of primary infection is in a healthcare facility, e.g. hospital; (2) community-onset, healthcare facility-associated (CO-HCFA) CDI – infection occurring in a non-hospital environment following contact with healthcare facilities; and (3) community-associated (CA) CDI – infection with no relation to a hospital stay.
(Good practice recommendation)
Voting:
A – 83.3%; B – 16.7%; C – 0%; D – 0%; E – 0%.
Agreement level: HIGH.
2.2. Recommendation 2
Recommendation on minimal supervision for institutions with limited resources
Implement HO-CDI surveillance in all hospitals to identify increased risk or outbreaks of CDI in the healthcare facility.
(Recommendation: weak; quality of evidence: low)
Voting:
A – 50%; B – 33.3%; C – 16.7%; D – 0%; E – 0%.
Agreement level: HIGH.
2.3. Recommendation 3
Recommendation on the best way to express CDI incidence and prevalence
Express HO-CDI prevalence as the number of cases per 10,000 man-days of treatment. Express CO-HCFA prevalence as the number of cases per 1000 patient admissions.
(Good practice recommendation)
Voting:
A – 83.3%; B – 16.7%; C – 0%; D – 0%; E – 0%.
Agreement level: HIGH.
2.1.1./2.2.1./2.3.1. Discussion
Clostridiumdifficile infection is a major cause of antibiotic-associated diarrhoea. It is responsible for 15–25% of all cases, with a considerable increase in the number of CDI cases in the 21st century [2]. In 2016 a report was issued evaluating 7711 CDI cases from 20 countries. The number of healthcare-associated (HA) CDI cases was 5756 (74.6%), and the number of community-associated (CA) CDI cases was 1955 (25.4%). In the hospital, from which the analysed data were obtained, men constituted 44.9% of patients, with the average age of 75.0 years. The incidence of HA CDI was 2.4 cases/10,000 man-days. As for 126/593 (21.2%) of the hospital surveillance period, no cases of HA CDI were observed. The median in-hospital incidence of HA CDI was 2.9 cases per 10,000 man-days. The average incidence of CDI was the highest in reference hospitals (5.8 cases/10,000 man-days; 95% CI 3.6–7.8 cases/10,000 man-days). It was also the lowest in community hospitals (2.8 cases/10,000 man-days; 95% CI: 2.1–3.5 cases/10,000 man-days; p = 0.001). Estonia, Lithuania, and Poland reported the highest incidence of HA CDI.
The incidence of CA CDI was 0.8 cases/10,000 man-days, with the highest rates being reported by Estonia, France, and Poland [3].
Clostridium difficile is a Gram-positive, obligate anaerobe and a spore-forming bacterium, which was first identified in 1935 [4]. The most common route of transmission is faecal-oral. At the beginning of the 21st century, an increase in CDI incidence was observed due to the rapidly spreading NAP1 strain (North American Pulse Field Type 1) otherwise known as ribotype 027 or B1/NAP1/027. This strain caused both an increased risk of developing hospital-based endemic foci and a regional increase in CDI incidence. The endemic strain B1/NAP1/027 is hypervirulent, characterised by an increased ability to form spores, high resistance to fluoroquinolones, and it more often causes infections with severe clinical course [5, 6].
Over the last decade, one of the most serious epidemiological problems was the increase in C. difficile infection rate in hospitals. Based on Kronika Epidemiologiczna (pol. the epidemiological chronicle), in 2016 C. difficile infections accounted for 28.3% of all registered outbreaks of hospital-acquired infections. There were 8716 cases reported, of which 540 were fatal [7].
According to the data from the National Institute of Public Health – National Institute of Hygiene in Poland, in 2017 the incidence of CDI was 30.2 per 100,000 inhabitants. There were 11,667 reported cases. Hospital stay was required in 88.1% of patients [8]. In the years 2011 to 2013, clinical studies were conducted in 13 hospitals in Poland, proving that PCR ribotype 027 CDIs predominate (62%). The second most common aetiological factor is ribotype 176, responsible for 14% of CDIs [6].
Risk factors of CDI
CDI infections are significantly more common in patients with risk factors, including: antibiotic therapy, age over 65 years, hospitalisation for more than 4 weeks or stay in a long-term medical care facility, comorbidities, diseases associated with immunosuppression or chemotherapy, and use of proton pump inhibitors. Chronic diseases that promote CDI infection include: chronic kidney disease, dialysis, cystic fibrosis, diabetes, stroke, malnutrition, alcoholism, chronic heart disease, chronic lung disease, primary and secondary immunity disorders – including HIV, cancer, and non-specific inflammatory bowel diseases [4]. Almost every antibiotic used can be a risk factor for the development of CDI; even the drugs used to treat CDI – vancomycin and metronidazole – are a risk factor for the disease. Antibiotics were divided into three groups, depending on the degree of risk of CDI induction (Table IV) [5].
Table IV
Antibiotics and the risk of CDI
Clostridiumdifficile colonisation and infection
Clostridiumdifficile transmission in a healthcare facility is most likely the result of faecal-oral spread or direct contact with a contaminated environment. The asymptomatic colonisation rate with C. difficile is 3–26% among adult in-patients. The rate of asymptomatic C. difficile presence in stool in adults is < 2%. Research suggests that individuals with asymptomatic colonisation for a prolonged time have a reduced risk of developing CDI. Long-term colonisation may be dependent on the increase of anti-A and anti-B toxin serum antibodies [9].
The main routes of C. difficile transmission include: hands of the medical staff and a polluted environment, an inadequately cleaned room previously occupied by a CDI patient, and contaminated thermometers and other medical equipment.
Different forms of CDI are summarised in Table V [10–12].
Table V
Severity of CDI
| Severity | SHEA and IDSA criteria [10] | Criteria by Zar et al. [11] | Criteria accepted by the National Program for Antibiotic Protection according to McDonald et al. [12] |
|---|---|---|---|
| Severe | Leukocytosis ≥ 15,000/mm3 and/or creatinine ≥ 1.5 × greater than before the infection onset | At least 2 of the following: – age ≥ 60 – temperature ≥ 38.3°C – albumin ≤ 2.5 g% – leukocytosis ≥ 15 000/mm3 and/or – pseudomembranes on endoscopy and/or – treatment in ICU | Within 30 days since CDI diagnosis: – treatment in ICU due to complications (e.g. shock, catecholamine infusion) or – surgery due to complications (toxic megacolon, perforation, ineffective medical treatment) |
| Fulminant | Leukocytosis > 50,000/mm3 | – | – |
| Severe complicated | Ileus, perforation, or shock | – | Toxic megacolon, perforation, ineffective medical treatment, or death |
2.4. Recommendation 4
Recommendation on CDI surveillance in the face of high endemic or epidemic rates
Classify data based on patient location in order for appropriate control measures when CDI incidence exceeds average rates and/or it is impossible to limit the disease spread, or an outbreak occurs.
(Recommendation: weak; quality of evidence: low)
Voting:
A – 83.3%; B – 16.7%; C – 0%; D – 0%; E – 0%.
Acceptance level: HIGH.
2.4.1. Discussion
Special supervision is required in patients with diarrhoea, toxic megacolon, or severe ileus [13], as well as in those testing positive or with pseudomembranes on endoscopy or microscopy. The occasional infection is a new first episode of symptoms, i.e. no symptoms were present within the preceding 8 weeks; positive test result – enzyme-linked immunosorbent assay (EIA) or nucleic acid amplification assay (NAAT). The current infection is the onset of symptoms and a positive stool test following an episode, with a positive test result within the previous 2 to 8 weeks [14].
Surveillance of specific communities in the event of an outbreak in non-hospitalised individuals should be considered to determine the incidence trends in the given population. Adequate classification of patients by place of residence can improve infection control.
3. Symptoms, differential diagnosis, and diagnostic criteria
3.1. Recommendation 5
Preferred patient population for testing, most suspected of CDI
Patients with new, unexplained diarrhoea with ≥ 3 loose stools within 24 h are the preferred target population for CDI testing.
(Recommendation: weak; quality of evidence: very low)
Voting:
A – 50%; B – 50%; C – 0%; D – 0%; E – 0%.
Acceptance level: HIGH.
3.1.1. Discussion
Testing for C. difficile infection should include individuals with an unexplained new episode of three or more loose stools within 24 h. The optimal number of loose stools required for CDI testing depends on the likelihood of infection (high vs. low) and potential confounding factors such as an underlying disease and surgical interventions. Potential confounding factors may contribute to an increased risk of antibiotic-associated diarrhoea. When a patient presents with diarrhoea that cannot be attributed to any disorder, testing for C. difficile infection is recommended. Testing should also be conducted when diarrhoea persists despite discontinuation of treatment, e.g. with laxatives.
Some conditions and medical interventions relating to the diarrhoea itself, such as irritable bowel syndrome (IBS) or enteral feeding, are associated with an increased risk of CDI. In clinical practice, it is difficult to exclude CDI in patients with new-onset or severe diarrhoea. The clinical criteria for the diagnosis of CDI have changed along with increasing awareness of C. difficile infection. It is worth mentioning that the number and frequency of loose stools justifying CDI testing have decreased over the last 40 years. In 1974, Tedesco et al. defined diarrhoea as > 5 stools a day [15]. In 2013, Johnson et al. defined diarrhoea as > 3 loose stools or watery bowel movements within 24 h [16]. Clinicians can improve the test accuracy only by examining patients suspected of C. difficile infection.
3.2. Recommendation 6
The most effective methods (in terms of positive and negative predictive values) to identify patients at risk of a clinically significant C. difficile infection based on stool testing
Use a stool toxin test as part of a multi-step algorithm, i.e. 1. glutamate dehydrogenase (GDH) and toxin; 2. GDH plus toxin, confirmed by nucleic acid amplification test (NAAT); or 3. NAAT plus toxin. This algorithm is recommended rather than NAAT alone for all samples sent to the laboratory when there are no local guidelines for stool sampling.
(Recommendation: weak; quality of evidence: low)
Voting:
A – 66.6%; B – 16.7%; C – 16.7%; D – 0%; E – 0%.
Acceptance level: HIGH.
3.2.1. Discussion
Clostridiumdifficile infection should be suspected in patients with diarrhoea (≥ 3 loose stools within 24 h) of unexplained origin. Many laboratory studies can be implemented in CDI diagnosis. Those methods are based on direct detection of bacteria as well as one or both major toxins (A and B) in stool. Table I lists those methods in order of decreasing analytical sensitivity. Toxicological cultures include selective medium or its variant and anaerobic incubation for several days. Later the organism is identified using several methods such as laser desorption, matrix-assisted ionisation mass spectrometry. The characteristic ‘horse barn’ smell often heralds the presence of C. difficile. After pathogen identification, testing for toxins should be performed to confirm infection with the toxin-producing strain. Toxicological culture is one of the gold standard methods, to which other studies are compared.
Another reference method is a cell culture cytotoxicity neutralisation assay (CCNA) [17]. It allows for direct detection of toxins in stool. First, stool filtrate is prepared and administered to an appropriate monolayer cell line such as Vero cells or human fibroblasts. After incubation, the cells are observed for cytopathic effect (CPE). The control study is carried out simultaneously using neutralising antibodies to Clostridium sordellii or C. difficile to confirm that CPE is caused by the toxins produced by C. difficile and not by other substances in the stool. Incubation is continued for a maximum of 48 h, but positive results can often be obtained after overnight incubation. This method is time-consuming and lacks standardisation; however, it is characterised by high sensitivity and specificity. CCNA has been supplanted by faster antigen and molecular testing methods. Enzyme immunoassays (EIAs) detect proteins in the tested material using monoclonal or polyclonal antibodies linked to an enzyme. At first, the tests only detected toxin A; however, with time both toxins A and B started to be detected. Toxins A and B and hydrolytic enzymes including hyaluronidase, chondroitin sulfatase, gelatinase, and collagenase are virulence factors of C. difficile. Toxin A is an enterotoxin stimulating production of cytokines, chemokines, and substance P. In addition, toxin A stimulates neutrophil dipeptidase production in the intestinal lumen, leading to disruption of connections between enterocytes and their apoptosis. Toxin B is a cytotoxin causing apoptosis of enterocytes damaged by toxin A. Both toxin A and toxin B inactivate Rho GTPase (guanosine triphosphate hydrolase) leading to depolymerisation of actin fibres and damage to the skeleton of the colon epithelial cells, resulting in pseudomembrane formation. They consist of neutrophils, fibrin, mucus, and enterocyte residues. Microscopically, pseudo-membranes form of yellowish-white deposits on the inflamed colonic mucosa.
The most frequently used method in CDI diagnosis is based on direct detection of C. difficile toxin in a stool sample. In selected patients, e.g. with inflammatory bowel disease, it is recommended that CDI be tested for at every disease exacerbation. The most commonly used method is the enzyme immunoassay (EIA) [17, 18], which gives quick results, i.e. after 1–2 h. The sensitivity is 75–85% and specificity 95–100%. EIAs use monoclonal or polyclonal antibodies to detect C. difficile toxins, and many commercial tests are available. Due to poor performance of EIA tests, GDH (glutathione dehydrogenase) immunoassays and molecular tests for detecting toxin genes have been developed [19, 20]. Glutamate dehydrogenase immunoassays detect the metabolic enzymes present in all C. difficile isolates. The antigen detected by the GDH test is not specific only to C. difficile toxin-producing strains, it is also present in non-toxin-producing C. difficile bacteria. The combination of two tests allows for a faster result and better sensitivity compared to the EIA tests alone [20]. In 2009, the nucleic acid amplification test (NAAT) was used to detect CDI. The test is based on the PCR (polymerase chain reaction) method and has a higher sensitivity (80–100%) and specificity (87–99%) compared to the EIA test. The NAAT test has limitations in the form of high costs of implementation and difficulties in interpretation.
Table VI shows the available tests for CDI detection in order of decreasing sensitivity.
Table VI
Available tests for CDI detection in order of decreasing sensitivity [1]
| Test | Sensitivity | Specificity | Detected substance |
|---|---|---|---|
| Bacterial cultures | High | Low* | C. difficile vegetative forms or spores |
| Nucleic acid amplification test | High | Low/moderate | C. difficile nucleic acid (toxin genes) |
| Glutamate dehydrogenase | High | Low* | C. difficile common antigen |
| Cytotoxicity neutralisation test on a cell culture | High | High | Free toxins |
| Immunological tests detecting toxins A and B | Low | Moderate | Free toxins |
The optimal diagnostic method for CDI is hard to achieve.
Patients may also be carriers of toxin-producing strains without clinically apparent disease. Diarrhoea in hospitalised patients is common, and C. difficile is responsible for less than 30% of cases. Based on current understanding, it is difficult to indicate which method is the most useful in CDI detection. Tables VII and VIII compare the available diagnostic tests for CDI [21–27].
Table VII
Evidence for the recommended diagnostic tools for C. difficile infection diagnosis
| Evidence for diagnostic tools | Design | Number of participants | Limitations | Quality of evidence (GRADE) | References, first author |
|---|---|---|---|---|---|
| GDH and NAATs have the highest sensitivity, but low PPV in asymptomatic patients; all tests had high NPV regardless of symptoms | Observational study, patient interview and clinical assessment | 150 | Low number of participants; only basic lab tests were assessed in real time | Dubberke [21] | |
| Toxins – negative, NAAT – positive, untreated patients showed no side effects. The CDI relapse was more common when both NAAT and toxins were positive rather than when only NAAT was positive (31% vs. 14%; p = 0.03) | Retrospective observational study | 128 | Low number of participants | Kaltsas [22] | |
| No difference in positive evaluation for EIA toxicity between patients with mild and severe disease (49% vs. 58%; p = 0.31) | Observational study, prospective study, retrospective assessment of patient data | 299 | Single-centre study | Humphries [23] | |
| Complications were more common with positive NAAT and GDH/EIA/CCNA compared to isolated positive NAAT (39% vs. 3%; p < 0.001) | Prospective cohort study; observational | 1321 | Only some samples were tested using the gold standard | Longtin [24] | |
| Patients with positive CCNA or GDH/EIA showed higher all-cause mortality compared to patients with positive NAAT or TC (p = 0.001) | Multi-centred observational study | 12420 | Limited clinical data | Planche [25] | |
| Patients testing positive for toxins with EIA showed longer duration of diarrhoea, more CDI-related complications, greater CDI-related mortality compared to patients testing negative for toxins but with positive PCR test result (8.4% vs. 0.6%, p = 0.001) | Prospective single-centre trial, observational, cohort | 1416 | Single-centre study | Polage [26] |
Table VIII
Evidence for recommended diagnostic tests for CDI. It is preferred to use NAAT alone or as a part of a multi-step algorithm (i.e. GDH plus toxin supported by NAAT or NAAT plus toxin) rather than testing for toxins when there are established institutional procedures for stool sampling
| Evidence for diagnostic tests | Study | Number of participants | Limitations | Quality of evidence (GRADE) | References, first author |
|---|---|---|---|---|---|
| PCR was more sensitive (93.3%) than EIA (73.3%) for toxin detection and direct cytotoxicity test (76.7%), when used in patients meeting clinical criteria for CDI | Observational; patient interviews | 350 | Peterson [27] | ||
| Using clinical diagnosis as reference, PCR was more sensitive than CCNA and GDH (99.1% vs. 51% 83.8%). Clinically confirmed in 91.5% of cases | Prospective double-centre study | 1051 | Various order of tests; limited statistical analysis; limited patient observation | Berry N, Sewell B, Jafri S, et al. Real-time polymerase chain reaction correlates well with clinical diagnosis of Clostridium difficile infection. J Hosp Infect 2014; 87: 109-14 |
[i] CCNA – cell cytotoxicity neutralisation assay, CDI – Clostridium difficile infection, EIA – enzyme immunoassay, GDH – glutamate dehydrogenase, NAAT – nucleic acid amplification test, NPV – negative predictive value, PCR – polymerase chain reaction, PPV – positive predictive value, TC – toxin-producing strain culture.
3.3. Recommendation 7
The most sensitive method of CDI diagnosis in stool in patients at risk of CDI based on clinical presentation
If stool samples were obtained from patients who were at risk of CDI based on clinical presentation, use NAAT or a multi-step algorithm to test: 1. GDH plus toxin; 2. GDH plus toxin and NAAT; and 3. NAAT plus toxin. Those methods are preferred over toxin detection itself, when there are established institutional criteria for stool sampling.
(Recommendation: weak; quality of evidence: low)
Voting:
A – 0%; B – 100%; C – 0%; D – 0%; E – 0%.
Acceptance level: HIGH.
3.3.1. Discussion
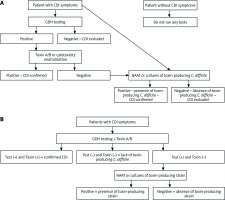
If patients are carefully examined for clinical symptoms of CDI (at least three loose stools within 24 h, with a history of exposure to antibiotics), then the best way of further evaluation is a multi-step clinical algorithm. GDH plus toxin or GDH plus toxin/NAAT or NAAT plus toxin. A two- or three-step algorithm has greater diagnostic accuracy compared to single-step testing. Recommended diagnostic algorithms are shown on Figure 1.
3.4. Recommendation 8
The role of repeated testing. Should repeated testing be considered in asymptomatic patients, including post-treatment control test?
During the same episode of diarrhoea, stool tests should not be repeated for 7 days. Stool testing should be avoided in asymptomatic patients. Stool testing can be performed for epidemiological studies in asymptomatic patients.
(Recommendation: strong; quality of evidence: moderate)
Voting:
A – 83.3%; B – 16.7%; C – 0%; D – 0%; E – 0%.
Acceptance level: HIGH.
3.4.1. Discussion
The issue of retesting for CDI is strictly linked to the accuracy of the testing method. The use of suboptimal specificity tests means that multiple repetitions are associated with a high risk of false positives. In the absence of CDI suspicion, do not re-test. The use of highly sensitive testing strategies (two-stage or three-stage algorithms) means that individual tests have a very high negative predictive value (usually > 99%).
3.5. Recommendation 9
The use of lactoferrin or other biological markers in stool to increase efficacy of CDI diagnosis compared to detecting toxin-producing C-difficile strains
There is insufficient evidence to recommend using biological markers to enhance diagnosis.
(No recommendation)
Voting:
A – 100%; B – 0%; C – 0%; D – 0%; E – 0%.
Acceptance level: HIGH.
3.5.1. Discussion
In the recent years, there has been an increase in the number of biomarkers detected in stool used to differentiate inflammatory and non-inflammatory diarrhoea. Those include lactoferrin, calprotectin, as well as IL-8 and IL-1β [28].
Lactoferrin is an iron-binding glycoprotein found in neutrophils, and its concentration in the stool is proportional to the neutrophil count in stool [29]. Calprotectin is a calcium-binding protein found in the neutrophil cytosol [30, 31]. Secretion of intestinal cytokines such as IL-8 and IL-1β was also evaluated. Although the markers are used in the diagnosis of irritable bowel syndrome, their usefulness in the diagnosis of CDI has not been established [32].
4. Infection control and prevention
4.1. Recommendation 10
Isolation of CDI patients
Should separate rooms and toilets be used for isolated CDI patients?
Isolation recommendations for patients with CDI – placing patients in separate rooms with separate toilets protects against disease transmission to other patients. When there are not enough toilets and single rooms, priority should be given to patients with stool incontinence, who should be placed in separate rooms.
(Recommendation: strong; quality of evidence: moderate)
Voting:
A – 100%; B – 0%; C – 0%; D – 0%; E – 0%.
Acceptance level: HIGH.
4.1.1. Discussion
Patients with confirmed or suspected CDI should be isolated. This is a preventive measure against infection spread in the ward. Additional precautions are often implemented, especially when CDI rates are high. The ‘control packet’ strategy for infection control is used to effectively control major CDI foci. It includes hand washing, isolation, environmental decontamination, and antibiotics [33–35]. The basis for CDI prevention and control are properly designed hospital rooms and hand washing products. Single rooms with private toilets are preferred. The effect of single rooms on CDI and other bacterial transmission rates was investigated when the ICU of one of the hospitals was closed for renovation [36]. It was established that the CDI rate decreased by 43%. When patient grouping is necessary due to a lack of single rooms, the patients should be given individual lockers for storing private items, which reduces transmission.
4.2. Recommendation 11
Recommendations for cohorting infected or colonised patients with the same pathogen
If cohorting is necessary, it is recommended to group patients colonised or infected with the same pathogen. CDI patients should not be grouped if there is discordance with respect to other drug-resistant pathogens such as MRSA – methicillin-resistant Staphylococcus aureus or vancomycin-resistant Enterococcus.
(Recommendation: strong; quality of evidence: moderate)
Voting:
A – 83.3%; B – 16.7%; C – 0%; D – 0%; E – 0%.
Acceptance level: HIGH.
4.3. Recommendation 12
Should hospital staff wear gloves and gowns when caring for CDI patients?
Hospital staff should wear gloves (recommendation: strong; evidence quality: high) and gowns (recommendation: strong; evidence quality: moderate) when entering rooms with CDI patients and when caring for them.
Voting:
A – 100%; B – 0%; C – 0%; D – 0%; E – 0%
Acceptance level: HIGH.
Voting:
A – 83.3%; B – 16.7%; C – 0%; D – 0%; E – 0%.
Acceptance level: HIGH.
4.3.1. Discussion
Preventive measures include gowns and gloves when caring for CDI patients. The hands of the hospital staff may be contaminated with C. difficile spores, especially when they do not wear gloves and are exposed to faeces [37]. In addition, when using gloves, remove them carefully so as not to contaminate your hands and forearms. It is recommended that protective gowns be worn due to the risk of contamination of uniforms. Contaminated uniforms can be a source of C. difficile transmission.
4.4. Recommendation 13
When should isolation be established?
Safety precautions should be established for patients suspected of CDI while waiting for the results of C. difficile testing, when the results cannot be obtained on the same day.
(Recommendation: strong; quality of evidence: moderate)
Voting:
A – 83.3%; B – 16.3%; C – 0%; D – 0%; E – 0%.
Agreement level: HIGH.
4.4.1. Discussion
If there is a delay in test results, safety measures should be introduced similar to those for confirmed CDI. Patients with suspected CDI should be subjected to preventive safety measures while waiting for CDI confirmation [38].
4.5. Recommendation 14
How long should the isolation last?
Safety measures restricting contact with the patient should be continued up to at least 48 h after resolution of diarrhoea.
(Recommendation: weak; quality of evidence: low)
Voting:
A – 50%; B – 33.3%; C – 16.7%; D – 0%; E – 0%.
Agreement level: HIGH.
It is appropriate to extend safety measures and restrict the contact with the patient until discharge if the CDI incidence rate is high despite introduction of standard infection control measures.
(Recommendation: weak; quality of evidence: low)
Voting:
A – 33.3%; B – 50%; C – 16.7%; D – 0%; E – 0%.
Agreement level: HIGH.
4.5.1. Discussion
According to British recommendations, safety measures should be continued up to 48 h after resolution of diarrhoea [39]. The average time to diarrhoea resolution is 4.2 days. There is no evidence for reduced CDI rate in response to prolongation of safety measures.
4.6. Recommendation 15
Which hand washing method is recommended (assuming glove use) during care for isolated CDI patients?
In the case of high CDI rates or an outbreak, routine hand washing with soap and water is the preferred method instead of alcohol-based products. Hand washing with soap and water before and after contact with CDI patients shows superior efficacy in removing C. difficile spores.
(Recommendations: strong; quality of evidence: moderate)
Voting:
A – 66.6%; B – 16.7%; C – 16.7%; D – 0%; E – 0%.
Agreement level: HIGH.
Hand washing using water and soap is preferred when there is direct contact with faeces or probably contaminated skin region, e.g. perineal area.
(Good practice recommendation)
Voting:
A – 83.3%; B – 16.7%; C – 0%; D – 0%; E – 0%.
Agreement level: HIGH.
4.6.1. Discussion
The hands of the medical staff are often the source of C. difficile infection. A prospective study on 27 CDI patients showed that C. difficile can often contaminate various body regions including the groin, chest, abdomen, forearms, and hands. Contamination of chest or abdominal skin persisted after resolution of diarrhoea [40]. Patients with CDI symptoms are the main source of disease transmission. Their faeces contain a large number of spores, which are transferred/excreted to the environment. Contamination of skin, clothes, bedsheets, and surroundings have been appropriately called faecal veneer [40, 41]. The hands of the medical personnel are an important vector of spore transmission onto vulnerable patients [41]. Patients may also become contaminated with spores due to direct contact with contaminated surfaces or appliances. If patient isolation in a single room with a separate bathroom is impossible, then hand washing is of upmost importance to prevent the disease transmission between patients, medical staff, and visitors [42]. It was proven that C. difficile can be isolated from hands and faeces of asymptomatic medical staff. Moreover, spores were also found in sewage, soil, and patients’ houses. It is epidemiologically significant that C. difficile can survive for up to 5 months on the floor.
4.7. Recommendation 16
The role of the patient’s bath in CDI prevention
Patients should be encouraged to wash their hands and take a shower in order to decrease the amount of C. difficile spores on the skin.
(Good practice recommendation)
Voting:
A – 100%; B – 0%; C – 0%; D – 0%; E – 0%.
Agreement level: HIGH.
4.7.1. Discussion
The patient can transport C. difficile spores on his or her contaminated hands. Hand washing and disinfection are important to prevent CDI recurrence. Patients should take a bath or shower regularly to decrease the number of spores on their skin [43].
4.8. Recommendation 17
Recommendations for cleaning non-critical equipment and accessories after their use by CDI patients
Use disposable equipment if possible. Ensure that reusable equipment is thoroughly cleaned and disinfected, especially with use of sporicidal disinfectants compatible with the equipment.
(Recommendation: strong; quality of evidence: moderate)
Voting:
A – 83.3%; B – 16.7%; C – 0%; D – 0%; E – 0%.
Agreement level: HIGH.
4.9. Recommendation 18
The role of manual final disinfection using sporicidal disinfectants active against C. difficile in isolated CDI patients
Cleaning the rooms using sporicidal disinfectants should be considered in combination with other measures preventing CDI endemic infections in the case of an epidemic or if there is evidence of recurring CDI in the same room.
(Recommendation: weak; quality of evidence: low)
Voting:
A – 33.3%; B – 66.7%; C – 0%; D – 0%; E – 0%.
Agreement level: HIGH.
4.11. Recommendation 20
The role of automatic terminal sporicidal disinfection against C. difficile in isolated CDI
Until now there have only been a few studies recommending the use of automatic terminal disinfection in CDI prevention.
(Non-recommended)
Voting:
A – 16.7%; B – 66.6%; C – 0%; D – 0%; E – 16.7%.
Agreement level: HIGH.
4.12. Recommendation 21
The role of everyday sporicidal disinfection
For CDI prevention, everyday cleaning using dedicated sporicidal disinfectants should be considered, especially in the case of an epidemic, pandemic, or recurrent cases of CDI.
(Recommendation: weak; quality of evidence: low)
Voting:
A – 16.7; B – 66.6%; C – 16.7%; D – 0%; E – 0%.
Agreement level: HIGH.
4.12.1. Discussion
Everyday cleaning using sporicidals decreases contamination with C. difficile. Available disinfectants include the following: hypochlorite-based solution (500 ppm of chlorine), quaternary ammonium compounds, whitening tissues with 0.55% active chlorine, and peracetic acid-based products [1, 44].
4.13. Recommendation 22
The necessity and usefulness of identification of asymptomatic carriers of C. difficile with further isolation of C. difficile carriers
There is a lack of evidence for compulsory screening of asymptomatic carriers for their isolation or implementation of safety measures.
(Not recommended)
Voting:
A – 50%; B – 16.7%; C – 0%; D – 33.3%; E – 0%.
Agreement level: LOW.
4.14. Recommendation 23
The role of proper antibiotic therapy in control of CDI rate
In order to minimise the risk of CDI development, the frequency and duration of high-risk antibiotic therapy should be limited. Moreover, the number of prescribed antibiotics should be limited as well.
(Recommendation: strong; quality of evidence: moderate)
Voting:
A – 100%; B – 0%; C – 0%; D – 0%; E – 0%.
Agreement level: HIGH.
The national program of rational antibiotic use should be implemented.
(Good practice recommendation)
Voting:
A – 100%; B – 0%; C – 0%; D – 0%; E – 0%.
Agreement level: HIGH.
4.14.1. Discussion
In the case of a CDI epidemic it is important to implement supervision of the used antibiotics. The program of antibiotic rational use should be implemented. The frequency and duration of antibiotic therapy may have an impact on CDI development. The use of a considerable number of antibiotics (about four) is a significant risk factor of CDI progression, while the number of C. difficile infections increases with the number of prescribed antibiotics [1]. CDI risk is associated with increased dosage, the number of antibiotics, and the duration of therapy. Adjusted hazard ratio (HR) of patients receiving two, three, four, or five antibiotics was, respectively, 2.5 (95% CI: 1.6–4.0), 3.3 (95% CI: 2.2–5.2), and 9.6 (95% CI: 6.1–15.1) times higher compared to patients with one drug. It is important to minimise the duration of drug administration and verify the validity of antibiotic therapy. Hospitals should implement antibiotic management programs. Benefits from these programs are as follows: improvement of the treatment results, decrease of adverse events such as CDI, and improvement of sensitivity to antibiotics [45, 46].
4.15. Recommendation 24
Decrease the frequency and duration of antibiotic therapy
In order to decrease the risk of CDI, the frequency and duration of high-risk antibiotic therapy and the number of prescribed antibiotics should be minimised.
(Recommendation: strong; quality of evidence: moderate)
Voting:
A – 100%; B – 0%; C – 0%; D – 0%; E – 0%.
Agreement level: HIGH.
4.16. Recommendation 25
Targeted antibiotic therapy for CDI control
The type of antibiotics used should reflect the local epidemiological situation and C. difficile strains. Reduced used of fluoroquinolones, clindamycin, and cephalosporins (except for surgical antibiotic prevention) should be taken into consideration.
(Recommendation: strong; quality of evidence: moderate)
Voting:
A – 100%; B – 0%; C – 0%; D – 0%; E – 0%.
Agreement level: HIGH.
4.17. Recommendation 26
The role of proton-pump inhibitors (PPI) use reduction in CDI prevention
There is an epidemiological association between PPI and CDI. If there are no definite indications for PPI therapy, it should be discontinued. There are insufficient data to indicate cessation of PPI therapy as a CDI prevention measure.
(Non-recommended)
Voting:
A – 83.3%; B – 16.7%; C – 0%; D – 0%; E – 0%.
Agreement level: HIGH.
4.17.1. Discussion
Many studies reported the associations between PPI and CDI [47, 48]. Despite clinical data indicating a constantly increased risk of CDI in patients receiving PPI therapy, the impact of PPI on CDI incidence is still not clearly established. Many clinical trials were characterised by data heterogeneity, the influence of unidentified confounding factors, or lack of dose-dependent effect. Because of an unclear association between PPI therapy and CPI development, PPI therapy with no clear indications should be discontinued.
4.18. Recommendation 27
Usefulness of probiotics in primary CDI prevention
At the moment there are insufficient data about the role of probiotics in primary CDI prevention, and thus no clear recommendation can be given.
(Not recommended)
Voting:
A – 50%; B – 16.7%; C – 16.7%; D – 16.7%; E – 0%.
Agreement level: LOW.
4.18.1. Discussion
The literature gives contradictory information about the influence of probiotics in C. difficile prevention.
Lactobacillus plantarum 299 v (Lp299v) colonises healthy individuals’ digestive tract mucosa following oral administration. Its excellent adhering properties are a result of binding to mannose of the mucosal cells. This strain, due to its adherence, colonises rectal mucosa and is able to survive for up to 11 days after the supplementation is stopped, which prevents adherence of other pathogenic bacteria. Moreover, studies showed that Lp299v stimulates production of mucin by intestinal cells [49]. Lp299 is vulnerable to antibiotics such as ampicillin, erythromycin, and trimethoprim/sulfamethoxazole [50, 51]. Studies including patients receiving antibiotic therapy supplemented by either Lp299v or genomically similar L. plantarum 299 showed high safety profile and lack of significant side effects [47, 52].
Saccharomyces boulardii is a probiotic that was shown to decrease the infection recurrence in patients with severe or recurring CDI [53, 54]. Studies on animal models showed that this probiotic causes an increase in antibodies against toxin A, which subsequently decreases the intensity of CDI symptoms. Moreover, S. boulardii has an anti-inflammatory effect due to suppression of the nuclear transcription factor κB (NF-κB), and it reduces IL-8 production, which is a mediator of inflammatory response in CDI [55].
Products containing S. boulardii are available on the Polish market; some of them contain S. boulardii exclusively, others contain additional probiotics and prebiotics (e.g. inulin).
Clinical studies also assessed the efficacy of Lactobacillus acidophilus and Bifidobacterium bifidum in patients receiving antibiotics in CDI prevention. The proportion of CDI patients was higher in the group of patients receiving placebo compared to those on probiotic supplementation. On the other hand, patients receiving the probiotic were characterised by more frequent C. difficile colonisation without toxin presence in stool [56].
Probiotics may be useful in CDI prevention if they are administered (without previous history of CDI) during antibiotic therapy [57, 58]. There have also been reports on infection caused by probiotic microorganisms [58]. Previous scientific reports showed limited success in CDI decrease in high-risk patients who received systemic antibiotics. Due to a lack of reliable studies and insufficient data, there are no recommendations for probiotic administration in CDI prevention [51].
5. Treatment
5.1. Recommendation 28
Supportive treatment in CDI
The antibiotic that triggered the infection should be discontinued as soon as possible because it may affect the risk of CDI recurrence.
(Recommendation: strong; quality of evidence: moderate)
Voting:
A – 83.3%; B – 16.7%; C – 0%; D – 0%; E – 0%.
Acceptance level: HIGH.
Empirical CDI antibiotic therapy should be implemented when a significant delay in laboratory confirmation is expected or in the case of fulminant CDI.
(Recommendation: weak; quality of evidence: low)
Voting:
A – 33.3%; B – 66.7%; C – 0%; D – 0%; E – 0%.
Acceptance level: HIGH.
5.2. Recommendation 29
The safest and most effective treatment regimen for the first CDI episode
Treatment of the first CDI episode should have high efficacy and cause symptom relief and resolution within a month after the therapy. Vancomycin or fidaxomicin are the drugs of choice for the treatment of the first CDI episode. Vancomycin 125 mg four times a day PO for 10 days or fidaxomicin 200 mg twice daily for 10 days are recommended.
(Recommendation: strong; quality of evidence: high)
Voting:
A – 100%; B – 0%; C – 0%; D – 0%; E – 0%.
Acceptance level: HIGH.
Metronidazole should only be used for the first episode of non-severe CDI, and only in the face of limited access to vancomycin and fidaxomicin. The metronidazole regimen is 500 mg three times a day PO for 10 days.
(Recommendation: weak; quality of evidence: high)
Voting:
A – 83.3%; B – 16.7%; C – 0%; D – 0%; E – 0%.
Acceptance level: HIGH.
Due to potential neurotoxicity of metronidazole, treatment exceeding 10 days should be avoided.
(Recommendation: strong; quality of evidence: moderate)
Voting:
A – 100%; B – 0%; C – 0%; D – 0%; E – 0%.
Acceptance level: HIGH.
5.2.1. Discussion
Treatment of the first episode of CDI involves vancomycin orally at the dose of 125 mg four times a day or fidaxomicin 200 mg twice daily. The therapy should last for 10 days. If vancomycin or fidaxomicin is not available and the infection is mild, you can give metronidazole 500 mg three times a day PO for 10 days. Prolonged metronidazole therapy can be dangerous because of its neurotoxic effect. Treatment regimens and comparison of treatment efficacy of the first episode are presented in Tables IX and X [10, 16, 59–64].
Table IX
Treatment of C. difficile infection in adults according to IDSA and SHEA guidelines [10]
Table X
Efficacy comparison of various regimens/treatment option for C. difficile infection according to IDSA and SHEA guidelines [10]
| Evaluated outcome | Number of participants (Number of studies) | Patient percentage | Relative risk (95% CI) | P-value | Quality of evidence | First author [References] |
|---|---|---|---|---|---|---|
| Direct comparison of metronidazole with vancomycin | ||||||
| Resolution of diarrhoea after 10 days of treatment | RCT before 2000: 156 (2) | 95 (MTR) 98 (VAN) | RR: 0.97 (0.91–1.03) | 0.4 | Teasley [59] Wenisch [60] | |
| RCT before 2000: 687 (3) | 75 (MTR) 85 (VAN) | RR: 0.89 (0.82–0.96) | 0.002 | Zar [11] Johnson [61] | ||
| Overall RCT: 843 (5) | 78 (MTR) 87 (VAN) | RR: 0.89 (0.85–0.96) | 0.0008 | High | ||
| Resolution of diarrhoea after treatment without CDI relapse within a month | RCT before 2000: 156 (2) | 85 (MTR) 84 (VAN) | RR: 1.0 (0.9–1.2) | 1.0 | Teasley [59] Wenisch [60] | |
| RCT after 2000: 687 (3) | 59 (MTR) 70 (VAN) | RR: 0.84 (0.74–0.94) | 0.002 | Zar [11] Johnson [61] | ||
| Overall RCT: 843 (5) | 63 (MTR) 73 (VAN) | RR: 0.87 (0.79–0.96) | 0.003 | High | ||
| Direct comparison of vancomycin and fidaxomicin | ||||||
| Resolution of diarrhoea after 10 days of treatment | 1105 (2) | 88 (FDX) 86 (VAN) | RR: 1.0 (0.98–1.1) | 0.36 | High | Louie [62] Cornely [63] |
| Resolution of diarrhoea after treatment without CDI relapse within a month | 1105 (2) | 71 (FDX) 57 (VAN) | RR: 1.2 (1.1–1.4) | < 0.0001 | High | Louie [62] Cornely [63] |
| Direct comparison of faecal transplant and vancomycin | ||||||
| Resolution of diarrhoea after treatment without CDI relapse by 56 days after treatment | 29 (1) | 81 (FMT) 31 (VAN) | RR: 2.6 (1.1–6.2) | 0.01 | Moderate | van Nood [64] |
[i] RCT – randomised controlled trial, RR – relative risk, CI – confidence interval, MTR – metronidazole, VAN – vancomycin, FDX – fidaxomicin, FMT – faecal microbiota transplant. RR was calculated relative to vancomycin, i.e. RR < 1 favours vancomycin, RR > 1 favours the other treatment option compared to vancomycin.
5.3. Recommendation 30
The best management of fulminant CDI
Fulminant CDI is defined as a severe, complicated disease characterised by hypotension or shock, ileus, or acute toxic megacolon. Vancomycin PO is the treatment of choice for fulminant CDI. The vancomycin dosage is 500 mg PO four times a day.
(Recommendation: strong; quality of evidence: moderate)
Voting:
A – 100%; B – 0%; C – 0%; D – 0%; E – 0%.
Acceptance level: HIGH.
When CDI is complicated by ileus, vancomycin can be administered rectally. The dose of vancomycin is 500 mg in 100 ml saline every 6 h as a retention enema.
(Recommendation: weak; quality of evidence: low)
Voting:
A – 83.3%; B – 16.7%; C – 0%; D – 0%; E – 0%.
Acceptance level: HIGH.
Metronidazole should be administered intravenously together with oral or rectal vancomycin, especially if there is intestinal obstruction. The metronidazole dose is 500 mg IV every 8 h.
(Recommendation: strong; quality of evidence: moderate)
Voting:
A – 83.3%; B – 0%; C – 16.7%; D – 0%; E – 0%.
Acceptance level: HIGH.
5.3.1. Discussion
Fulminant infection complicated by toxic megacolon, ileus, or shock should be treated with vancomycin 500 mg PO four times daily. If intestinal obstruction is present, vancomycin can be administered rectally at the dose of 500 mg in 100 ml 0.9% NaCl every 6 h. Vancomycin therapy should be enhanced by intravenous metronidazole 500 mg three times daily.
5.4. Recommendation 31
Surgical treatment for CDI
We recommend surgical treatment for critically ill patients. The preferred method is subtotal rectal-sparing colectomy.
(Recommendation: strong; quality of evidence: moderate)
Voting:
A – 66.7%; B – 33.3%; C – 0%; D – 0%; E – 0%.
Acceptance level: HIGH.
Loop ileostomy with colonic lavage followed by vancomycin lavage is an alternative approach that may lead to better treatment outcomes.
(Recommendation: weak; quality of evidence: low)
Voting:
A – 50%; B – 33.3%; C – 16.7%; D – 0%; E – 0%.
Acceptance level: HIGH.
5.4.1. Discussion
If medical treatment proves ineffective, surgical treatment should be considered, including subtotal rectal-sparing colectomy [65].
5.5. Recommendation 32
The most effective and safe treatment regimen for the first CDI recurrence
For the first CDI relapse, prolonged oral vancomycin therapy is recommended with a gradually reduced and pulsatile regimen instead of a second 10-day vancomycin cycle. The dose is 125 mg four times a day for 10–14 days, later reduced to two times a day for a week, then once a day for a week, and finally once every two or three days for 2 to 8 weeks.
(Recommendation: weak; quality of evidence: low)
Voting:
A – 83.3%; B – 0%; C – 16.7%; D – 0%; E – 0%.
Acceptance level: HIGH.
Alternatively, the first CDI relapse can be treated with fidaxomicin for 10 days instead of the standard 10-day vancomycin regimen if vancomycin was used in the initial therapy.
(Recommendation: weak; quality of evidence: moderate)
Voting:
A – 83.3%; B – 0%; C – 16.7%; D – 0%; E – 0%.
Acceptance level: HIGH.
Vancomycin in the standard 10-day regimen should be given when metronidazole was used for treatment of the first episode of CDI. Metronidazole treatment should not be repeated.
(Recommendation: weak; quality of evidence: low)
Voting:
A – 83.3%; B – 0%; C – 16.7%; D – 0%; E – 0%.
Acceptance level: HIGH.
5.6. Recommendation 33
The most effective and safe treatment regimen of the second or subsequent CDI recurrence
Patients with a second or subsequent CDI relapse should be treated with oral vancomycin in a pulsatile regimen. The dose of vancomycin is 125 mg PO four times a day for 10–14 days, two times a day for a week, once a day for a week, then once every two or three days for 2 to 8 weeks.
(Recommendation: weak; quality of evidence: low)
Voting:
A – 83.3%; B – 0%; C – 16.7%; D – 0%; E – 0%.
Acceptance level: HIGH.
OR
Patients with a second or subsequent CDI relapse should be treated with the standard vancomycin oral regimen, i.e. 125 mg four times daily followed by 400 mg rifaximin PO three times daily for 20 days.
(Recommendation: weak; quality of evidence: low)
Voting:
A – 50%; B – 33.3%; C – 16.7%; D – 0%; E – 0%.
Acceptance level: HIGH.
Alternatively, patients with a second or subsequent relapse of CDI may be treated orally with fidaxomicin 200 mg twice daily for 10 days.
(Recommendation: weak; quality of evidence: low)
Voting:
A – 66.6%; B – 16.7%; C – 16.7%; D – 0%; E – 0%.
Acceptance level: HIGH.
5.6.1. Discussion
For treatment of CDI relapses, prolonged use of vancomycin in gradually reduced doses is recommended. The preferred regimen is 125 mg four times a day for 10–14 days, then 125 mg twice daily, then 125 mg once a day, and 125 mg every 2 to 3 days for 2 to 8 weeks. It is also justified to treat relapses with fidaxomicin 200 mg twice daily for 10 days instead of vancomycin. The vancomycin standard 10-day regimen should be used if the first episode was treated with metronidazole [1].
Subsequent relapses (second or later) require prolonged vancomycin or fidaxomicin course. Vancomycin 125 mg four times a day for 10–14 days, then twice a day for 7 days, then once a day for 7 days, then once a day every 2 to 3 days for a period of 2 to 8 weeks. It is also possible to prescribe rifaximin α 400 mg three times a day for 20 days after a standard 10-day vancomycin course. Fidaxomicin is recommended for a relapse at a dose of 200 mg twice a day for 10 days.
Rifaximin αtherapy is new and was first introduced in the guidelines for CDI treatment in 2018. In the study by Garey et al. in 2011, it was shown that the use of rifaximin reduced the CDI recurrence significantly more often [66]. Treatment regimens and comparison of treatment efficacy for the first episode are presented in Tables IX and X.
5.7. Recommendation 34
Faecal microbiota transplant for CDI treatment
Faecal microbiota transplant is recommended for CDI treatment in patients with multiple recurrences, in whom targeted antibiotic therapy proved ineffective.
(Recommendation: strong; quality of evidence: moderate)
Voting:
A – 50%; B – 50%; C – 0%; D – 0%; E – 0%.
Acceptance level: HIGH.
5.7.1. Discussion
Antibiotic therapy used in CDI patients is associated with the risk of relapse. Some patients experience multiple relapses. Promising treatment outcomes in those cases have been obtained following faecal microbiota transplant (FMT). FMT involves administration of faecal filtrate obtained from a healthy donor to the patient’s intestines, with the aim of curing CDI and restoring the missing components of the intestinal microbiota. The effect can be observed after administration of one or two doses through a colonoscope or by rectal infusion. Curative factors in FMT therapy include not only live bacteria, but also bacterial components, antimicrobial compounds of bacterial origin, e.g. bacteriocins or bacteriophages, which contribute to normalisation of intestinal microbiota after CDI. FMT can be administered by rectal enema, through an NG tube, or in the form of freeze-dried capsules. The capsules can potentially become popular due to their low invasiveness and simple administration. Patients undergoing FMT achieve remission of the CDI-related diarrhoea, which is not always the result of reinfection but can stem from post-infectious irritable bowel syndrome. Such postinfectious diarrhoea is associated with other microbiota abnormalities. The use of FMT can be extended to dysbiosis caused by other bacteria responsible for enteritis [67].
The effectiveness of FMT is still subject to research, and the effectiveness of FMT was lower in magnitude in randomised controlled trials compared to non-randomised studies [68, 69]. Because the use of FMT is still new, the long-term effects of FMT therapy are not yet known.
FMT is an effective treatment option in infections caused by the highly virulent 027 C. difficile ribotype, which is also highly resistant to antibiotics. The treatment consists of vancomycin for four days at the dose of 4 × 125 mg PO followed by FMT [70].
Donor preparation for FMT involves taking thorough medical history, obtaining blood samples, testing for hepatitis A, B, and C viruses, and HIV infection. Collected faecal samples are examined for the presence of parasites, toxins A and B of C. difficile, Helicobacter pylori antigen, and bacteria including Salmonella, Shigella, Campylobacter, Escherichia coli 0157:H7, as well as Yersinia enterocolitica. In addition, the tests for cytomegalovirus, Epstein-Barr virus, and rotaviruses are carried out [71].
Quraishi et al. in their meta-analysis of 37 studies, including seven randomised controlled studies, showed that FMT was more effective than vancomycin treatment (RR = 0.23, 95% CI: 0.07–0.80) for relapsing and refractory CDI. The clinical effect in all studies was 92% (95% CI: 89–94%). Significantly higher efficacy was observed if FMT was performed in the lower gastrointestinal tract compared to the upper segment – 95% (95% CI: 92–97%) vs. 88% (95% CI: 82–94%), p = 0.02. There were no differences between fresh and frozen FMT 92% (95% CI: 89–95%) vs. 93% (95% CI: 87–97%), respectively, p = 0.84. Subsequent administration of FMT cycles after failure of the first FMT resulted in a further increase in treatment efficacy. Donor testing was consistent, but there was variability in recipient preparation and FMT volume. The above analysis proves that FMT, regardless of the route of administration, is an effective treatment for recurrent and refractory CDI [72].
Similarly, in Poland Grzesiowski et al. [73] obtained the curative rate of 55/62 (88.7%) in recurrent CDI (rCDI); 76.5% of cures occurred after the first course of treatment, 14.5% after the second, and the remaining 9% after the third infusion through the nasogastric tube. The results by Grzesiowski are particularly important because they indicate that in most cases CDI treatment should be planned with implementation of FMT cycles (not just a single transplant), bringing the expected curative rate to nearly 100%.
Research on faecal filtrate transfer (FFT) is underway. FFT is a clear brown liquid with no intense odour. Preparation of FFT consists of filtering faecal suspension through air pressure filtration systems; however, such filtrate has a reduced microbiota composition compared to FMT [67].
5.8. Recommendation 35
Treatment of patients requiring continuation of targeted antibiotic therapy against primary infection or repeated antibiotic therapy shortly after finishing CDI treatment
There are currently insufficient data on patients requiring continuous antibiotic therapy for the primary infection or requiring repeated antibiotic therapy shortly after discontinuing CDI treatment to recommend prolongation of treatment against C. difficile, or treatment using non-standard therapy, or resuming C. difficile treatment.
(Not recommended)
Voting:
A – 33.3%; B – 50%; C – 16.7%; D – 0%; E – 0%.
Acceptance level: HIGH.
5.8.1. Discussion
A new form of CDI treatment is the administration of bezlotoxumab. It is a human monoclonal antibody that binds to C. difficile toxin B, neutralises it, and prevents damage to the cells of the colonic mucosa. To date, too few studies have been conducted to confirm its effectiveness, and phase III trials are ongoing [74]. According to the data from the summary of product characteristics, the drug should be administered during antibiotic therapy for CDI [75]. The novel mechanism of action of the drug does not directly affect the colon microbiome, and bezlotoxumab therapy may be an alternative treatment for recurrent C. difficile infection in patients at risk [76].
Research on C. difficile vaccine are still ongoing. Active vaccination may be, in the future, a form of CDI prevention. There are currently three products in the clinical phase trials [77]. In addition, they are also being studied with biotherapeutics, i.e. products containing toxin-non-forming C. difficile strains. There is also an oral β-lactamase (SYN-004) being studied that is supposed to protect the colonic microbiota by reducing the antibiotic concentration in the intestinal lumen. SYN-004 – ribaxamase is an oral β-lactamase that has been designed for administration together with intravenous β-lactam antibiotics [78].