Introduction
In liver surgery, the most important factors affecting morbidity and mortality are intraoperative blood loss and septic complications [1]. Various techniques have been used to prevent intraoperative blood loss. The oldest and most commonly used of these techniques is the Pringle manoeuvre (PM). The PM was first discovered in 1908 by J.H. Pringle, and it was proven that temporary clamping of the hepatoduodenal ligament reduces intraoperative blood loss [2]. The PM causes portal, superficial, and inferior mesenteric venous stasis, which leads to a decrease in splanchnic blood flow [3]. Experimental studies have shown that splanchnic hypoperfusion secondary to the PM causes intestinal mucosal injury and bacterial translocation (BT) [4–7]. Intestinal mucosal injury and BT have been shown to be predisposing factors in the development of serious clinical conditions after trauma, major surgery, and shock, such as sepsis, and multiorgan failure [8, 9].
Immunonutrition is a nutritional product containing high energy and protein, enriched with certain nutrients, mostly arginine, glutamine, omega-3 fatty acids, and nucleotides. The use of these products 5–7 days prior to various surgical procedures has been shown to reduce postoperative infectious complications [10]. It has also been shown to reduce BT in experimental obstructive jaundice and intestinal obstruction models [11, 12].
Aim
In this study, it was planned to investigate the effects of preoperative immunonutritional support against development of BT due to PM, which is commonly used in liver surgery.
Material and methods
The study protocol was made with the approval of Kocaeli University Experimental Animal Ethics Committee. Forty Wistar albino rats weighing 200–250 g were used in the study. Rats were housed in a 12-hour light/dark cycle at constant temperature in accordance with the standards of the National Institutes of Health Publication Guide for the Care and Use of Laboratory Animals (NIHPGC). Rats were randomly sampled and divided into four groups with 10 rats in each group (Table I). All groups were fed for 5 days according to their characteristics. Groups 1 and 2 were fed with standard rat diet and water, and groups 3 and 4 were supplemented with 1 g/kg/day amino acid containing enteral nutritional supplement (22% protein, 51% carbohydrate, 24% fat, also containing L-arginine 14 g/l, L-glutamine 10 g/l) and fed with a paediatric orogastric feeding probe in addition to standard rat diet and water.
Table I
Groups (n = 10)
Surgical procedure
All subjects, kept under standard laboratory conditions, were allowed to drink water while being fasted on the day of operation at the end of the fifth day. Anaesthesia was induced by administering intramuscular 100 mg/kg ketamine hydrochloride and 10 mg/kg xylazine hydrochloride combination to all subjects in the group.
After anaesthesia, the anterior abdominal wall was cleansed with povidone-iodine and then the hepatoduodenal ligament was released by exploration of the abdomen through a 2 cm upper abdominal incision, as standard. The hepatoduodenal ligament was occluded for 30 min in the subjects of groups 2 and 3 using a vascular clamp. At the end of 30 min, reperfusion was provided, and the sac was closed with 3/0 Vicryl. The hepatoduodenal ligament was explored with laparotomy in group 3 and 4 subjects. After 30 min of waiting, the abdomen was closed with 3/0 Vicryl. All subjects in the groups underwent relaparotomy 30 min after the closure of the abdomen. The portal vein was cannulated with a gauge catheter and a 2-ml blood sample was taken. Samples from the spleen and terminal ileum were taken for microbiological examination. All surgical procedures were performed using sterile instruments in aseptic conditions. After the procedure, the rats were sacrificed by cardiac incision.
Microbiological analysis
Microbiological analyses of the study were carried out in Kocaeli Derince Training and Research Hospital Microbiology Laboratory. During microbiological analysis, tissue samples from the meso of small intestine were placed in sterile tubes containing 2 ml Brain Heart Infusion Broth (RTA Laboratories, Turkey). After homogenisation, direct seeding was carried out for qualitative evaluation and for quantitative evaluation, 100 μl was inoculated into 5% Sheep Blood Agar (RTA Laboratories, Turkey ) and Eosine Metilen Blue Agar (RTA Laboratories, Turkey), and then the plates were incubated for 24 h at 37°C. Incubation of plates without reproduction was extended to 48 h. Tissue samples taken from the spleen were collected into sterile tubes with 2 ml Brain Heart Infusion Broth (RTA Laboratories, Turkey). After homogenisation, direct seeding was carried out for qualitative evaluation and for quantitative evaluation, 100 μl was inoculated into 5% Sheep Blood Agar (RTA Laboratories, Turkey) and Eosine Metilen Blue Agar (RTA Laboratories, Turkey), and then the plates were incubated for 24 h at 37°C. Incubation of plates without reproduction was extended to 48 h. Two-millilitre blood samples taken from the portal vein were incubated in a BacT/ALERT®3D (bioMERIEUX, France) automated blood culture device after being inoculated into BacT/ALERT®3D (bioMERIEUX, France) blood culture bottles.
Reproducible microorganisms were identified by a VITEK®2Compact (bioMERIEUX, France) automated identification and antibiogram instrument.
Statistical analysis
Statistical evaluation was performed with IBM SPSS 20.0 (SPSS Inc., Chicago, IL, USA) package programme. Categorical variables were given as frequencies (percentages). Differences between groups were assessed by Monte Carlo χ2 analysis. P < 0.05 was considered sufficient for statistical significance in two-way tests.
Results
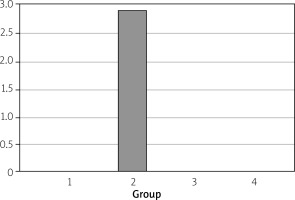
During the experiment, all subjects tolerated the intervention, and no subjects were lost within the estimated period or left out of the study. Growth in blood cultures were detected in three samples (30%) of group 2 (standard feed + PM), while no growth was detected in the other groups. In the statistical analysis of all groups, the proliferation in blood cultures of group 2 was statistically higher than in the other groups (p < 0.001) (Table II). All of the proliferation in blood cultures occurred within the first 48 h after inoculation. Enterococcus faecalis growth was seen in two samples of portal vein culture of group 2 and Leuconostoc pseudomesenteroides growth was seen in one sample (Figure 1).
Table II
Distribution of microorganism species isolated from blood cultures
| Microorganism species | Group 1 | Group 2 | Group 3 | Group 4 |
|---|---|---|---|---|
| Enterococcus faecalis | 0 | 2 (20%) | 0 | 0 |
| Leuconostoc pseudomesenteroides | 0 | 1 (10%) | 0 | 0 |
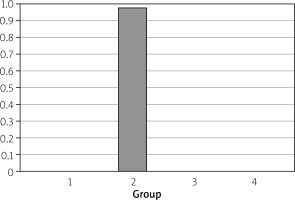
Growth in mesenteric lymph node specimens was 10% in group 2, and no growth was observed in other groups (Figure 2). When compared with other groups, there was no statistically significant difference, but there was a proliferation in group 2 (p = 1.00). Enterococcus faecalis had proliferated in the mesenteric lymph node samples of a rat from group 2. No spleen samples were found to have any growth; no statistical comparison was made for this reason.
Discussion
BT is defined as the passage of microorganisms or their products (endotoxin), which are localised in the intestinal organs to extraintestinal areas that are normally sterile, such as mesenteric lymph nodes, other intestinal organs, and bloodstream, by crossing the intestinal mucosal barrier [13]. There are different opinions about the clinical importance of BT. There are opinions suggesting that BT can be considered as a physiological event in normal healthy individuals [14]. However, contrary to this view, O’Boyle et al. [15] conducted a study in which BT was found in 15% of patients undergoing laparotomy, and 45% of the patients with BT had postoperative sepsis, which was reported to be 19% in the group without translocation. In addition, other studies supporting this view have reported that BT is a predisposing factor in the development of postoperative infectious complications in patients undergoing major abdominal surgery [16, 17].
PM, which is commonly used for intraoperative bleeding control in liver surgeries, has been shown to lead BT in mesenteric lymph nodes and portal blood specimens in various experimental and clinical studies [18–21]. Our study also showed that PM causes bacterial growth in portal blood and mesenteric tissue cultures in rats. Although there is sufficient information in the literature about the clinical conditions that cause BT, information about the anatomic pathway (lymphatic-hematogenous) that is effective in BT development and when it first developed is insufficient [22]. In our study, it was observed that there was growth in more rats in the portal blood cultures taken at the first hour after PM application, compared to the mesenteric culture samples. In a study done to explain the anatomic pathway in BT, it was shown that portal blood cultures in the early period had a higher rate of growth than mesenteric cultures, and the hematogenous pathway was the main pathway of BT [23].
There are few experimental studies to prevent BT after PM. In these few experimental studies, it has been shown that the use of prophylactic antibiotics, preoperative bowel cleansing, and the use of low-dose dexamethasone prevent BT after PM [18, 24].
It has been reported that the use of enteral immunonutrition products containing glutamine, arginine, nucleotide, and omega-3 fatty acids in different combinations and doses prior to surgery regulates the inflammatory response in the early period and strengthens the immune response and bowel function [25]. Glutamine has been shown to inhibit intestinal atrophy by increasing epithelial cell proliferation, to decrease bacterial adhesion to the intestinal mucosal surface by increasing levels of secretory immunoglobulin A and mucin, and to prevent BT by helping to preserve the integrity of the intestinal wall [26]. In a study conducted by Zou et al. in a severe acute pancreatitis model, enteral immunonutrition was reported to enhance the intestinal mucosa barrier system and the immune system [27]. In an obstructive jaundice model, the use of enteral immunonutrition products in preoperative and postoperative periods has been shown to reduce BT and intestinal villus atrophy [11]. Although histopathological examination was not included in our study, preoperative immunonutrition support was shown to prevent BT after PM, according to microbiologic studies.
Conclusions
In this study, based on microbiological findings, it was shown that PM leads to BT, and preoperative immunonutrition support prevents BT due to PM. However, uncertainties remain about how much BT is responsible for the development of postoperative septic complications. In light of our study and previous studies, the effect of immunonutritional support on BT and infectious complications in patient groups who undergo a surgical technique with a high risk of BT, such as PM, and other groups with risk of BT (burn, trauma, pancreatitis, cirrhosis) should be investigated in future clinical trials.