Solid pancreatic masses are observed as malignant in 88-95% of cases [1]. Pancreatic ductal adenocarcinoma accounts for nearly 90% of pancreatic neoplasms. Only 10–15% of pancreatic adenocarcinomas are diagnosed at a resectable stage. Moreover, even after resection and adjuvant chemotherapy, the median overall survival remains limited to approximately 54.4 months [2]. Extramedullary plasmacytoma (EMP) is characterized by the proliferation of plasma cells outside the bone marrow, forming a mass lesion. While the exact incidence of pancreatic involvement in plasmacytoma remains unclear, gastrointestinal involvement is reported in approximately 10% of cases [3]. Treatment strategies vary depending on the presence of underlying multiple myeloma (MM) and may include radiotherapy, surgery, or chemotherapy, with generally favourable outcomes [4]. Therefore, determining the origin of pancreatic masses is of paramount importance in guiding treatment decisions and prognostic assessment.
A 65-year-old female patient presented to our clinic with jaundice and pruritus that had developed over the past week. Her medical history included hypertension, diabetes mellitus, coronary artery disease (status post coronary stenting), and hypothyroidism. Additionally, she had been diagnosed with multiple myeloma 6 months prior and was undergoing VCD (bortezomib, cyclophosphamide, dexamethasone) therapy. On physical examination, scleral and cutaneous icterus were observed. Excoriation marks due to pruritus were present on the trunk and lower extremities. Abdominal examination was unremarkable. Laboratory tests revealed elevated levels of total bilirubin (10.2 mg/dl), direct bilirubin (10 mg/dl), alanine aminotransferase (167 U/l), aspartate aminotransferase (54 U/l), alkaline phosphatase (830 U/l), and gamma-glutamyl transferase (1002 U/l). C-reactive protein was elevated at 31 mg/l, whereas the white blood cell count was within normal limits (7.75 × 103/µl). Abdominal ultrasonography demonstrated intrahepatic bile duct dilation, common bile duct enlargement, and a suspicious mass lesion located at the pancreatic head. Magnetic resonance imaging (MRI) confirmed the presence of a mass obstructing the common bile duct (Figure 1); however, it was inconclusive in determining lesion origin. Serum CA19-9 levels, which are typically elevated in pancreaticobiliary malignancies, were within the normal range. Endoscopic ultrasonography (EUS) revealed an approximately 5 × 4 cm oval-shaped, heterogeneous mass. Since no specific imaging feature is pathognomonic for pancreatic malignancies, a biopsy was performed. Multiple samples were obtained from the duodenal bulb using a 19-gauge fine-needle biopsy (FNB) (Figure 2). Histopathological examination revealed clusters of CD138-positive, kappa light chain-positive plasma cells within a proteinaceous matrix (Figure 3). These findings confirmed that the pancreatic head mass was a plasmacytoma. During follow-up, the patient developed cholangitis, which necessitated endoscopic retrograde cholangiopancreatography (ERCP) with plastic biliary stent placement. After successful biliary drainage and cholangitis treatment, the patient was referred to the haematology department for further management.
Figure 1
Dilated intrahepatic and common bile ducts (yellow arrows) and a pancreatic head lesion (red arrow) on MRI
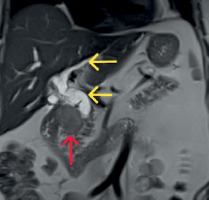
Figure 2
EUS-guided fine-needle aspiration (19G) from an oval, hypoechoic, heterogeneous mass lesion in the pancreatic head
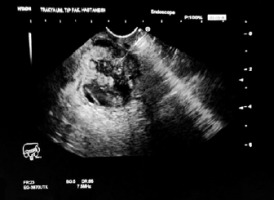
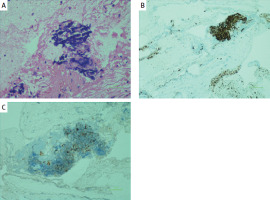
Figure 3
Histopathology findings: A – Lymphoplasmacytic cell infiltration within a proteinaceous matrix (hematoxylin eosin staining, .400); B – CD138 positivity in plasma cells (.100); C – Kappa light chain positivity in plasma cells (.100)
Extramedullary plasmacytomas are clonal plasma cell proliferations occurring in conjunction with or preceding multiple myeloma. Extramedullary involvement of plasma cell neoplasms is classified under four main categories: extramedullary disease, paraskeletal plasmacytoma, solitary plasmacytoma, and plasma cell leukaemia. Extramedullary disease (EMD) is a particularly aggressive variant of multiple myeloma, which is distinguished by the development of soft-tissue plasmacytomas that are the consequence of hematogenous dissemination. Furthermore, in cases of relapsed refractory multiple myeloma, the tissues most commonly affected by extramedullary disease include the liver, kidneys, lymph nodes, central nervous system, breast, pleura, and pericardium [5]. Our patient, diagnosed with extramedullary disease, is currently undergoing treatment for multiple myeloma. However, the site of occurrence was atypical, i.e., the pancreas. Involvement of the pancreas by plasma cell neoplasms is exceedingly uncommon, accounting for less than 0.1% of all pancreatic masses. Based on autopsy investigations, the diagnosis was frequently rendered postmortem, with an incidence rate of 2.3%. Approximately 80% of pancreatic EMPs are situated at the head of the pancreas. The majority are solitary lesions; however, in some instances, two or more simultaneous lesions may arise. Even more infrequently, the lesion may originate in a different abdominal organ but be sufficiently massive to impact the pancreas, particularly the pancreatic tail [6]. The lesion in our case was similarly situated in the head of the pancreas, consistent with existing literature. The differential diagnosis of pancreatic plasmacytoma encompasses adenocarcinoma, lymphoma, neuroendocrine tumours, focal pancreatitis, amyloidosis, and metastasis [6, 7]. Radiological findings are nonspecific. Transcutaneous ultrasonography serves as a valuable screening modality for patients exhibiting signs of obstructive jaundice. Regrettably, the presence of gas in the gastrointestinal tract and the size of the lesions (under 3 cm) may hinder visualisation. High-resolution, dual-phase, contrast-enhanced computed tomography is the principal imaging technique for patients with suspected pancreatic tumours, owing to its non-invasiveness and ease of administration. However, the modality with the highest sensitivity and specificity, which also allows for biopsy sampling without the limitations of computed tomography (CT)-guided techniques – such as the difficulty of biopsy due to the long track required to reach the retroperitoneum and the risk of tumour seeding – is endoscopic ultrasonography (EUS) [8]. The appearance of plasmacytoma on endoscopic ultrasonography is not pathognomonic. In most cases, it presents as a mass-forming lesion with well-defined borders, hypoechoic characteristics, and heterogeneous echogenicity compared to the normal pancreatic parenchyma [9, 10]. Our patient, who presented with jaundice, underwent transabdominal ultrasonography, which revealed a suspicious lesion. MRI was performed to better assess intrahepatic bile duct and common bile duct dilatation and to more effectively rule out obstructive lesions or calculi. Following the detection of a mass in the pancreatic head, an endoscopic ultrasonography (EUS) procedure was performed. EUS revealed an oval-shaped, well-defined, hypoechoic, and heterogeneous mass lesion, consistent with findings reported in the literature, compared to the pancreatic parenchyma. The cornerstone of diagnosis is histopathological evaluation obtained through biopsy. Biopsy samples can be acquired percutaneously, endoscopically, or surgically. The preferred method, when accessible, is EUS-guided biopsy due to its relatively low rate of major complications, reported to be as low as 2.5%. Among minor complications, the most common are pancreatitis and infection [11]. In histopathological evaluation, infiltration of the tissue by clonal plasma cells must be demonstrated. To confirm monoclonality, it is essential to identify monoclonal kappa or lambda light chains, heavy chains, or plasma cell markers such as CD38 and CD138 through immunohistochemical analysis [10]. In our patient, an EUS-guided biopsy was performed from the bulbous region of the duodenum without complications. Immunohistochemical analysis demonstrated abundant plasma cell clusters with CD138 and kappa light chain positivity.
Every pancreatic lesion must be carefully evaluated to ensure an accurate diagnosis because treatment strategies and prognoses vary significantly among different tumour types. EUS-guided biopsy is the most common method for obtaining pancreatic tissue samples. When performed by experienced practitioners, it is the safest technique, with a negligible complication rate.













