Introduction
Despite advances in pancreatic surgery with numerous prevention strategies, technological innovations, and development of safety protocols, postoperative pancreatic fistula (POPF) is a common complication associated with significant morbidity and mortality. POPF can originate from the pancreatic remnant after distal pancreatectomy (DP) or enucleation, as well as from the pancreatic anastomosis after head resections or drainage procedures [1–4]. The reported incidence varies between 3% after pancreatic head resection and 30% after DP. Identified risk factors for POPF are the soft gland texture of the pancreas, a small pancreatic duct diameter, intraoperative blood loss, and red blood cell (RBC) and fresh frozen plasma (FFP) transfusion needs [5].
By 2005, reporting of POPF in the scientific literature was characterised by 28 different definitions. Almost a decade later, the grading system was redefined to allow for more precise comparisons of surgical quality; Grade A POPF is called a “biochemical leak” because it has no clinical importance and is no longer referred to as a true pancreatic fistula, Grade B requires drains to be either left in place for more than 3 weeks or to be repositioned through endoscopic or percutaneous procedures, and Grade C POPF requires reoperation or may lead to single or multiple organ failure and/or mortality attributable to the pancreatic fistula [2, 6]. Clinically relevant postoperative pancreatic fistula (CR-POPF) carries a significant mortality risk of up to 25%. CR-POPF is both a clinical and biochemical definition, encompassing increased amylase levels in surgical drains and a deviation from the normal postoperative course, including complications such as bleeding, infection, organ failure, and mortality [6].
Early predictive markers have been tested in their ability to identify POPF, including C-reactive protein (CRP), serum amylase, drain lipase and amylase, and intraoperative acidosis, providing controversial findings [7–9]. Decreased mean platelet volume/total platelet count was a strong predictor for CR-POPF following PD in 105 patients in a recent study [10]. Direct and indirect interactions between platelets and pathogens have been described, as platelets collect and bundle bacteria and assist in neutrophil recruitment and stimulation via cytokines [11, 12].
Aim
This study suggests that an increase in platelet levels may be associated with the development of POPF and could serve as a potential predictive marker to guide therapeutic decisions.
Material and methods
Patient cohort
Two hundred patients underwent pancreaticoduodenectomy for any reason between 1.1.2018 and 31.12.2022 in two academic centres for pancreatic surgery (Table I). Exclusion criteria were patients < 18 years old and splenectomy as part of the operation. In all patients, preoperative workup included a pancreatic protocol abdominal computed tomography (CT), to exclude metastases and confirm surgical resectability. A total of 133 patients were eligible for inclusion in the study. Biliary stenting via endoscopic retrograde cholangiopancreatography was performed in patients presenting with cholangitis or serum total bilirubin higher that 10 mg/dl.
Table I
Demographic characteristics (N = 133)
Patients’ data were retrieved from a prospectively maintained database for analysis. Collected data included preoperative data, operative details, and postoperative morbidity. For all patients, full blood count was available pre-operatively and on postoperative days 1 to 9. The study was approved by the institutional board and Ethics Committee of University General Hospital Attikon, Athens, Greece (D’CEIR, EBD391/31-05-2023).
Statistical analysis
We conducted a comprehensive statistical analysis to assess the association between continuous variables and the presence and severity of POPF in our dataset. Ratios of postoperative platelet values (nominator) to preoperative platelet values (denominator) were assessed for postoperative days 1–9. The ratios were analysed using logistic regression models to examine their relationship with POPF, clinically significant POPF, and POPF grading. Each continuous variable was dichotomised based on its median value, and logistic regression models were fitted to evaluate the association between the dichotomised variables and the outcomes of interest. The statistical significance of the association was assessed using Wald tests, and odds ratios (ORs) with 95% confidence intervals (CIs) were calculated to quantify the strength and direction of the association. Additionally, c2 tests were performed to assess the association between dichotomised continuous variables and POPF grading, providing further insights into the predictive value of these variables for POPF severity. Statistical significance was determined at the conventional threshold of p < 0.05.
The analysis was conducted using RStudio (version 2023.12.1) and the following packages: cutpointr, broom, and nnet.
Results
A total 133 patients were included in the study. Fifty-one were female and 82 were male. The most common type of disease was pancreatic ductal adenocarcinoma (67%). Median (IQR) duration of the operation was 360 min (320–395 min). Median (IQR) length of stay was 11 (9–17.5) days (Table I).
Out of the 133 patients, POPF occurred in 69 (52%) patients with clinically significant POPF in 41 (31%); POPF grade A or biochemical leak (BL) in 28 (21%), POPF grade B in 29 (22%), and POPF grade C (reoperation or mortality) in 12 (9%).
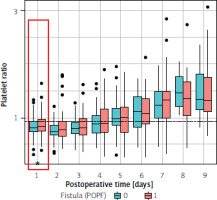
In the univariate unadjusted analysis, the ratios of postoperative platelet values (nominator) to preoperative platelet values (denominator) were assessed for postoperative days 1–9 (Table II A). A higher ratio on postoperative (post-OP) day 1 was significantly associated with POPF (logOR = 0.73; 95% confidence intervals [CI]: 0.04–1.43; p = 0.037). Longer hospitalisation and the presence of diabetes mellitus (DM) was also significantly associated with POPF. No association was observed between post-OP platelet changes and clinically significant POPF. In the multivariate analysis, a higher post-OP ratio on day 1 remained statistically significantly associated with POPF development (Table II B). Other predictors, including sex, smoking status, jaundice status, type of disease, operation duration, transfusions, and neoadjuvant therapy (NAT), were not found to have a significant impact on the likelihood of developing POPF. Results are summarised in Table II B, including the coefficients, p-values, and confidence intervals in the multivariate model. Figure 1 shows the boxplots of platelets and their association with POPF for post-OP days 1–9.
Table II
A – Univariate analysis (POPF), B – multivariate analysis (POPF)
A
B
No significant association was found between platelet changes and POPF grading or clinically significant POPF.
Discussion
This was a retrospective observational study in patients who underwent pancreaticoduodenectomy for any cause from 1.1.2018 to 31.12.2022 in two surgical centres for pancreatic surgery in Athens, Greece. We found that platelet increase on postoperative day 1 was associated with an increased likelihood of POPF. No significant association between platelet changes and POPF grading was found.
Platelets were traditionally thought to play an important role in haemostasis, thrombosis, and wound healing, but lately supporting literature accentuates platelets’ origin from a lower vertebrate cell type and underlines other interesting functions. Thus, platelet involvement is thought to interplay between haemostasis, thrombosis, inflammation, and development of cancerous tumours [12–15].
Platelets are thought to be an extension of the cellular immune system. They possess the ability to engulf foreign particles, recognise pathogens, and release cytokines and chemokines in response to invasion of microbes. They also play a key role in the inflammatory process by adhering to von Willebrand factor, forming platelet-leucocyte complexes, and bringing leucocytes to sites of damaged endothelium, while releasing thromboxane and CD40 ligand in circulation, which act as proinflammatory mediators [16]. Other factors that promote immunomodulation and are released by platelets are CD154, TREM-1 ligand, and P- and E-selectin [17]. On the other hand, there is evidence that proteins of the complement system are activated by platelets, and vice versa [18]. Finally, platelets are involved in a complex DNA structure with proteolytic activity – the neutrophil extracellular trap (NET) – which can trap microbes and contribute to its phagocytosis and lysis [19]. Interactions have been studied in disseminated intravascular coagulation (DIC), acute kidney injury (AKI), and septic cardiomyopathy [17–22].
Several biochemical markers have been tested before to predict POPF after pancreatic surgery. The drain fluid amylase levels on postoperative day 1 have been tested multiple times and with controversial findings. In a study by Chang et al. involving 6087 patients, a cut-off higher than 720 U/l predicted CR-POPF (HR = 4.6, p = 0.001). [20] This cut-off point for drain fluid amylase levels on postoperative day 1 was found to be higher (1883 U/l) in a recent prospective study [9]. Other researchers have tried to collate specific predictors in drain fluid measurements, such as the lipase-to-amylase ratio. The latter has shown an excellent ability to predict CR-POPF on postoperative days 3 and 5 (p < 0.05). [8] Serum amylase ≥ 120 on postoperative day 1 has also been reported to predict POPF [7]. Recently a combined pancreatic leak prediction score (CPLDS) was developed, which incorporates the main pancreatic duct diameter, body mass index (BMI), and comorbidity-polypharmacy score. CPLDS was predictive of POPF in univariate (p = 0.005) and multivariate (p = 0.036) analysis [21]. In another retrospective study with 1368 patients, high BMI, small pancreatic duct diameter, soft pancreatic texture, pathology other than ductal adenocarcinoma or chronic pancreatitis, minimally invasive resection, and male sex were all associated with CR-POPF [20–27]. Other studies report an elevated serum CRP level from POD 1 to POD 3 as a highly predictive factor for subsequent clinically relevant POPF [28–30].
Few studies have evaluated the potential role of platelets as predictors of POPF or CR-POPF. In a recent study by Lale et al., patients who underwent pancreaticoduodenectomy for periampullary diseases were divided into two groups, one with CR-POPF and a second with no POPF. According to their multivariate analysis, the CR-POPF group had a mean platelet volume/total platelet count ratio < 28.9 (p < 0.001) [10]. More recently, Ma et al. conducted a retrospective analysis in 232 patients evaluating the relationship of inflammation related indices of full blood count including neutrophil count, platelet count, lymphocyte count, systemic immune-inflammation index, platelet-to-lymphocyte ratio, and neutrophil-to-lymphocyte ratio. In that study, platelet count was associated with postoperative haemorrhage. Clinical postoperative pancreatic fistula was predicted by systemic immune inflammation index with an area under the curve (AUC) of 0.72, neutrophil-to-lymphocyte ratio (AUC = 0.71), and neutrophil count (AUC = 0.70). Previous studies did not draw the same conclusion. In 2016, neutrophil-to-lymphocyte ratio, platelet-to-lymphocyte ratio, and red cell distribution width-to-platelet ratio were calculated on postoperative day 1 and 3 and did not show predictive value for postoperative pancreatic fistula, whereas intraoperative acidosis was associated with postoperative higher serum amylase level [26]. On the contrary, elevated neutrophil-to-lymphocyte ratio and platelet-to-lymphocyte ratio were associated with clinically relevant postoperative pancreatic fistula after multivariate analysis in a study published in 2022 [27]. Finally, in a study from 2025, an association between higher platelet count and CR-POPF was found, among many other biochemical measurements [31].
In our study an interesting correlation was found between diabetes mellitus and a decreased likelihood of developing POPF. The latter seems unexpected, since DM is generally associated with complications, infections, and decreased wound healing. Similarly, however, other studies have found that the absence of DM is an independent risk factor for POPF [22, 23]. On the other hand, another study found that DM was significantly associated with grade C POPF on multivariate logistic regression (odds ratio = 1.41 95% confidence interval: 1.06–1.87, p = 0.02) [24].
This study has several limitations that need to be considered. Its retrospective design and relatively small sample size (133 patients) may introduce selection bias and limit statistical power. Being a two-centre study in Greece, the findings may not be generalisable to a broader population. Additionally, the lack of an external validation cohort warrants further investigation of the findings, although to our knowledge there is not another cohort study that investigates the use of platelets as a predictive tool for POPF. Due to limited access to other variables at the time of data acquisition, a direct comparison to established predictive biomarkers like drain fluid amylase or CRP was not performed.
Conclusions
Postoperative platelet count changes may serve as an early predictive marker for POPF following pancreaticoduodenectomy. A higher platelet count ratio on the first postoperative day was significantly associated with POPF, highlighting a potential role for platelet dynamics in postoperative risk assessment. Further prospective studies with larger cohorts and direct comparisons to established biomarkers are needed to confirm the clinical utility of platelet-based markers. If validated, this simple and widely available blood test could aid in early risk stratification and guide postoperative management strategies.