INTRODUCTION
Fungal skin infections are common and affect approximately 20–25% of the global population [1]. They represent the most prevalent dermatological disorders worldwide, accounting for 10.09% of skin diseases and contributing significantly to disability-adjusted life years (DALYs) associated with skin conditions [2, 3]. The main etiological agents include dermatophytes, non-dermatophyte moulds, and commensal yeasts, with dermatophytosis representing the most frequent clinical form [4].
Dermatophytosis, also referred to as tinea, involves the skin, hair, and nails [5, 6]. The causative organisms are dermatophytes belonging to three genera: Epidermophyton, Microsporum, and Trichophyton. These fungi colonize the stratum corneum and spread radially, without invading viable tissue.
In addition to dermatophytes, several non-dermatophyte fungi including Aspergillus, Scytalidium dimidiatum and Cladosporium, have increasingly been recognized as causes of dermatomycoses [7].
Despite their high prevalence and potential for chronicity or recurrence, superficial fungal infections are often perceived as minor ailments by healthcare providers, which may lead to underestimation of their impact on patients’ quality of life [8]. The diagnosis of dermatophytosis is typically based on clinical history and the appearance of skin lesions. However, it has been estimated that up to 40% of visual diagnoses may be incorrect [9, 10]. In addition, atypical presentations such as large lesions, “rings within rings”, corticosteroid-modified lesions, and involvement of multiple anatomical sites, can further complicate the diagnosis [11]. In such situations, laboratory investigations provide valuable confirmation.
Direct microscopic examination using a potassium hydroxide (KOH) mount is a widely used diagnostic method. KOH dissolves keratin, enabling visualization of fungal elements under the microscope. However, the reported sensitivity of this technique varies widely, ranging from 44% to 100% [12, 13]. Other diagnostic approaches include Wood’s lamp examination, fungal culture, and histopathological analysis. Although fungal culture is highly specific and useful for species identification, it has lower sensitive and requires a longer turnaround time, which may limit its clinical usefulness [14].
Recent advances in molecular diagnostic, including polymerase chain reaction (PCR) and spectrometric techniques such as matrix-assisted laser desorption/ionization time-of-flight mass spectrometry (MALDI-TOF MS), allow rapid and accurate detection of fungal pathogens. However, their routine use is often limited in resource-constrained settings due to high costs and technical requirements.
Several special stains can improve fungal detection in KOH preparations, including Parker blue-black ink, Swartz-Lamkins stain, Chlorazole-KOH, KOH-acridine orange, Chicago sky blue 6B (CSB), and fluorochromes such as Calcofluor white (CW). Among these, Parker ink (PI) and Chicago sky blue provide enhanced contrast compared with conventional KOH examination, facilitating identification of dermatophytes [15, 16].
Accurate and timely diagnosis is essential for appropriate treatment and for reducing the burden of fungal infections. In addition, diagnostic methods should be cost-effective and widely accessible, particularly in settings with limited healthcare resources. Therefore, this study aimed to evaluate the diagnostic performance of KOH, PI, and CSB staining in patients with suspected dermatophytosis.
OBJECTIVE
To evaluate the diagnostic characteristics (sensitivity, specificity, predictive values, and accuracy) of KOH, PI, and CSB staining in the diagnosis of dermatophytosis.
MATERIAL AND METHODS
Study design
A comparative observational study was conducted over a 1-year-period (November 2020 to November 2021) in the Outpatient Department of Dermatology, Venereology and Leprosy at Government Medical College, Jammu.
Inclusion criteria
Patients with a clinical diagnosis of dermatophytosis confirmed by at least two dermatologists were included in the study, regardless of age or sex, after obtaining informed consent.
Exclusion criteria
Patients with superficial fungal infections caused by yeasts (candidiasis, pityriasis versicolor, and Malassezia folliculitis), suspected clinically or during preliminary examination using KOH, Parker ink, or CSB.
Patients who had received oral or topical antifungal treatment within the previous 4 weeks.
Preparation of KOH and CSB
KOH pellets, PI and CSB dye powder were prepared as working solutions. A 10% KOH solution (w/v) was prepared by dissolving 10 g of KOH pellets in 100 ml of distilled water, whereas a 20% solution was prepared by dissolving 20 g of pellets in 100 ml of distilled water. A 1% CSB solution was prepared by dissolving 1 g of CSB dye powder in 100 ml of distilled water.
Sample collection
The selected skin areas were disinfected with 70% alcohol. Samples were collected by scraping the actively spreading peripheral margins of the lesions using the blunt edge of a No. 20 surgical blade. Hair specimens were obtained using epilation forceps to include the hair shafts. Nail clippings with attached subungual debris were collected using a nail clipper.
All collected samples (skin scrapings, nail clippings, and hair specimens) were divided into three portions and processed using the following methods:
KOH: 10% KOH for scrapings and 20% KOH mount for nail clippings and hair specimens
Parker ink staining with KOH used as the clearing agent
1% CSB staining with KOH used as the clearing agent
When culture was required, specimens were collected on sterile black card paper and inoculated onto Sabouraud dextrose agar (SDA) plates. The plates were incubated at room temperature for 2–4 weeks. If no growth was observed after 4 weeks, the culture was considered negative and discarded. Fungal culture was performed only in cases with diagnostic uncertainty after preliminary microscopy, multisite involvement, or recalcitrant disease.
Duplicate slides were prepared from each sample portion and appropriately labelled. One drop of 10% KOH was added to all the slides; one drop of CSB stain was applied to the second slide, and one drop of Parker ink to the third. Slides were gently covered with a coverslip, and excess stain was removed with blotting paper.
Microscopic examination was performed after 30 min using a light microscope with reduced illumination and lowered condenser to enhance contrast. Slides were initially examined under scanning (4×) and low power (10×) objectives, and suspicious areas were confirmed using the high-power (40×) objective. Slides prepared with KOH and added PI were assessed for fungal elements. A CSB-positive result was defined by the presence of blue-stained hyphae or spores.
Treatment response
Erythema, primary lesions (papules, pustules, and vesicles), and scaling were visually assessed by an independent blinded evaluator and graded as absent, mild, moderate, or severe. The evaluation was repeated 1 month after initiation of treatment.
The overall improvement was assessed using a six-grade ordinal scale:
Grade I – worsening,
Grade II – no improvement,
Grade III – 25% improvement,
Grade IV – 50% improvement,
Grade V – 75% improvement,
Grade VI – 100% improvement.
Pruritus intensity was recorded by the patient using a visual analogue scale (VAS).
Statistical analysis
The sensitivity, specificity, positive predictive value (PPV), negative predictive value (NPV), precision, and accuracy of CSB stain, PI, and KOH mount were calculated using treatment response after antifungal therapy as the reference standard.
Statistical analysis was performed using the compbdt program of R [17]. The Wald test was used to evaluated the null hypothesis, while the McNemar test with Holm correction was applied to assess difference between diagnostic methods. A p-value < 0.05 was considered statistically significant.
RESULTS
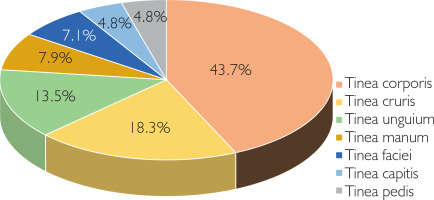
The study included 118 samples obtained from 110 patients with clinical diagnosis of dermatophytosis. Among the participants, 57.3% were male and 42.7% were female, with a mean age of 32.9 years. The most affected age group was 21–40 years, and the mean diseases duration was 4.37 months. The most common clinical presentations were tinea corporis (43.7%), tinea cruris (18.3%), and tinea unguium (13.5%) (Figure 1).
KOH examination detected 87.6% of cases and showed a specificity of 38.5% (Table 1). PI staining demonstrated a sensitivity of 95.2% and specificity of 84.6% (Table 2), whereas CSB staining achieved the highest sensitivity and specificity, reaching 99.1% and 92.3%, respectively (Table 3).
Table 1
Characteristics of potassium hydroxide
Table 2
Characteristics of Parker ink
Table 3
Characteristics of Chicago sky blue
Significant differences were also observed in predictive values and overall diagnostic performance. The positive predictive values (PPV) were 92.0% for KOH, 98.0% for PI, and 99.0% for CSB. Negative predictive values (NPV) were highest for CSB (92.3%), followed by PI (68.8%) and KOH (27.8%). Diagnostic accuracy and precision were also greatest for CSB (98.3% and 99.0%), compared with PI (94.1% and 98.0%) and KOH (82.2% and 92.0%).
When stratified by infection site, the sensitivities for skin infections were 89.4% for KOH, 94.1% for PI, and 98.8% for CSB. For nail infections, the corresponding sensitivities were 78.6%, 85.7%, and 92.9%, respectively. In hair infections, both PI and CSB detected all cases (100%), whereas KOH detected 83.3%.
DISCUSSION
Preliminary clinical assessment often fails to distinguish dermatophytosis from other conditions with similar clinical features. Reported diagnostic sensitivity of clinical examination is approximately 81%, with a specificity of 45% [18]. Although fungal culture is considered the diagnostic gold standard, its sensitivity ranges from 23% to 84.6% [9, 19], and false-negative results may occur. To address discrepancies between the reference method and the diagnostic test under evaluation, treatment has been proposed as an alternative reference standard in some studies [20, 21].
In the present study, the diagnostic performance of KOH, PI, and CSB staining was evaluated using treatment response as the reference standard. Both subjective and objective clinical improvement after 1 month of antifungal therapy was considered indicative of a positive treatment response.
Our findings highlight several limitations of conventional KOH examination, which remains one of the most commonly used diagnostic methods for dermatophytosis. Its relatively low specificity and frequent false-positive results reduce its reliability. In addition, the absence of colour contrast in KOH mounts may hinder identification of fungal elements, particularly in sparse samples or when artifacts resemble hyphae.
Sensitivity reflects the ability of a diagnostic test to correctly identify diseased individuals. Previous studies report KOH sensitivity ranging from 44% to 100%, depending on the sample site, with lower sensitivity observed in nail infections [12, 13, 22]. In the present study, KOH examination demonstrated a sensitivity of 87.6%, whereas PI and CSB achieved sensitivities of 95.2% and 99.1%, respectively. When stratified by infection site, skin infections were detected with sensitivities of 89.4% for KOH, 94.1% for PI, and 98.8% for CSB. For nail infections, the corresponding sensitivities were 78.6%, 85.7%, and 92.9% (CSB). In hair infections, PI and CSB detected all cases (100%), whereas KOH detected 83.3%.
Specificity indicated the ability of a test to correctly identify individuals without disease. False-positives KOH results may occur when artifacts such as fibres or cellular borders are misinterpreted as fungal elements. In our study, the specificity values for KOH, PI, and CSB were 38.5%, 84.6%, and 92.3%, respectively.
Predictive values are influenced by disease prevalence. The positive predictive values (PPV) observed in this study were 92.0% for KOH, 98.0% for PI, and 99% for CSB. Negative predictive values (NPV) were substantially higher for CSB (92.3%) and PI (68.8%) compared with KOH (27.8%), indicating that there staining methods may be more reliable for excluding dermatophytosis.
Diagnostic accuracy and precision were highest for CSB (98.3% and 99%), followed by PI (94.1% and 98%), and KOH (82.2% and 92.0%). The improved colour contrast provided by CSB facilitates visualizations of fungal elements, even at lower magnification and in samples with low fungal burden.
These findings are consistent with previous studies demonstrating the superior diagnostic performance of CSB compared with conventional KOH examination. Although KOH preparation remains rapid and inexpensive, it requires considerable experience and may yield false-negative results. PI improves visibility of fungal structures but still relies on subjective interpretation. In contrast, CSB staining provides clearer visualization and higher sensitivity, suggesting that it may represent a valuable addition to routine diagnostic methods for suspected dermatophytosis.
Limitations of the study
The sample size was relatively small; therefore, the results may not be fully generalizable to larger populations. Predictive values depend on disease prevalence in the studied population. Consequently, the PPV and NPV observed in this study may not be directly applicable to populations with different prevalence rates. The number of patients with nail and hair infections was lower than the number of patients with skin infections, which may have influenced the estimates of diagnostic performance for these sample types. Fungal culture and molecular species identification were not performed in all cases. This may have resulted in missed detection of Trichophyton indotineae, a species increasingly associated with terbinafine resistance and poor treatment response.