INTRODUCTION
Renal cell carcinoma (RCC) accounts for over 90% of primary renal malignancies and is among the most lethal urologic cancers [1]. Approximately 25% of patients present with metastatic disease at diagnosis, most commonly involving the lungs, lymph nodes, bones, liver, contralateral kidney, adrenal glands, and brain. Cutaneous metastases are rare, with reported prevalence ranging from 1% to 6.3% [1, 2].
We report the case of a middle-aged man who developed rapidly progressive cutaneous metastases of RCC, involving the face, scalp, and chest, shortly after discontinuing targeted therapy, despite a history of radical nephrectomy 3 years earlier.
CASE REPORT
A 56-year-old man with a 3-year history of clear cell RCC presented with multiple asymptomatic red raised lesions over the face, scalp, and trunk of 3 weeks’ duration. He denied pain, bleeding, or pruritus. On further questioning, he reported constitutional symptoms including fatigue, significant weight loss, exertional breathlessness, and abdominal discomfort.
The patient had undergone radical right nephrectomy 3 years earlier. Subsequently, he remained in remission while receiving targeted therapy with oral cabozantinib 40 mg daily for 1 year, followed by combination therapy with lenvatinib 12 mg and everolimus 5 mg for 6 months. His medical history was also significant for diabetes mellitus and hypertension, both well controlled with medications.
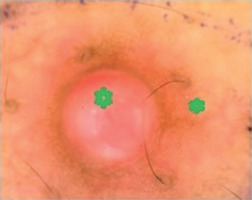
Cutaneous examination revealed multiple erythematous, non-tender, smooth, shiny nodules ranging from 0.5 to 1.5 cm in diameter over the scalp, face, and back (Figures 1 A–C). Dermoscopy demonstrated arborizing and tortuous vessels located around central lacunae, overlying the translucent nodules (Figure 2). Clinical and dermoscopic differential diagnoses included vascular tumors such as cherry angioma, pyogenic granuloma, Kaposi sarcoma, and bacillary angiomatosis.
Figure 1
Clinical photographs showing multiple non-tender, firm, reddish angiomatous nodules measuring 0.5–1.5 cm on the scalps (green arrows) (A), face (black arrows) (B) and back (black arrows) (C)
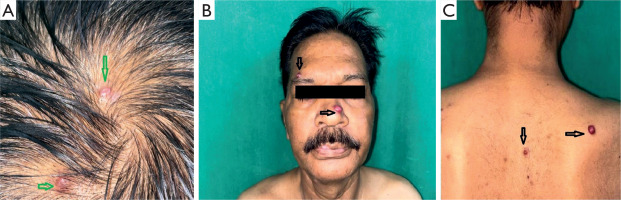
Figure 2
Dermoscopy showing arborizing and tortuous vessels (green stars) surrounding central lacunae over translucent nodules
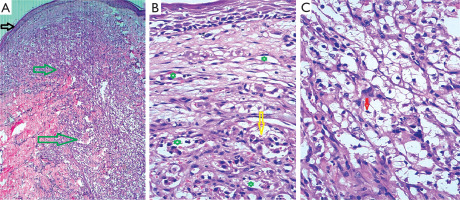
Routine hematological and biochemical investigations were unremarkable. Serological tests for human immunodeficiency virus (HIV), hepatitis B, and hepatitis C were negative. A punch biopsy from a nodule on the back showed an attenuated epidermis with dermal neoangiogenesis and diffuse infiltrative sheets of atypical clear cells involving the full thickness of the dermis (Figures 3 A, B). The cells were large with clear cytoplasm and centrally located nuclei showing focal atypia (Figure 3 C). Occasional clusters of clear cells were observed within vascular lumina.
Figure 3
Photomicrographs showing attenuated epidermis (black arrow) with pan-dermal interstitial sheets of clear cells (green arrows) (H&E, ×4) (A). Increased dermal vascularity (neoangiogenesis) (green stars), with intravascular clusters of atypical cells (yellow arrow) (H&E, ×20) (B). Clear cells with centrally placed cleaved nuclei (red arrow) showing an interstitial growth pattern (H&E, ×20) (C)
Clinicopathological correlation supported a diagnosis of cutaneous metastasis of clear cell RCC. Further evaluation with fluorodeoxyglucose positron emission tomography-computed tomography (FDG PET-CT) revealed metastases involving multiple organs, including the lungs, liver and lumbar vertebrae. The patient was advised continuation of lenvatinib and everolimus therapy; however, his clinical condition deteriorated rapidly and he died 2 months after the diagnosis.
DISCUSSION
Renal cell carcinoma (RCC) accounts for approximately 2–3% of malignancies affecting the adult population. Clear cell subtype is the most common, representing around 70–80% of cases. Inactivating somatic mutations or deletions in the von Hippel–Lindau (VHL) tumor suppressor gene located on chromosome arm 3p are implicated in up to 45% of clear cell RCC cases [2]. Clear cell RCC most frequently occurs in men between the ages of 50 and 70 years, consistent with the demographic characteristics of the presented patient.
Around 25–30% of patients present with metastatic disease at the time of diagnosis. The lungs, bones, liver, and brain are the most common sites of metastasis. Cutaneous metastases are rare, occurring in only 1–6% of cases, and usually indicate advanced systemic disease. RCC frequently spreads via the inferior vena cava or through the vertebral venous system (Batson’s plexus), enabling hematogenous dissemination to distant sites including the head and neck region. Porter et al. reported that metastatic RCC most often presents as a solitary cutaneous nodule, whereas multiple lesions were observed in this case [3]. The angiomatous clinical appearance of metastatic RCC is attributed to increased tumor vascularity mediated via vascular endothelial growth factor (VEGF) signaling and tumor-associated neoangiogenesis.
Due to their highly vascular nature, cutaneous metastases of RCC may clinically mimic angiomatoid conditions, including infectious entities such as bacillary angiomatosis as well as vascular tumors such as pyogenic granuloma, cherry angioma, and Kaposi sarcoma [4–6]. Histopathological examination remains the gold standard for distinguishing these entities, as summarized in Table 1.
Table 1
Clinicopathological and immunohistochemical comparison of angiomatous skin lesions
Certain tumors with clear cell morphology should also be considered in the differential diagnosis. Metastatic RCC typically demonstrates diffuse dermal infiltration by clear cells with abundant cytoplasm and centrally located nuclei, often accompanied by prominent vascular stroma and extravasated erythrocytes. Intracytoplasmic glycogen and lipid droplets are frequently present. In contrast, eccrine acrospiroma (nodular hidradenoma) is usually multilobular and characterized by dense hyalinized stroma and ductal differentiation. Sebaceous carcinoma may also contain clear cells but typically demonstrates sebocytic differentiation with scalloped nuclei and lacks the prominent vascular stroma characteristics of RCC [6]. Immunohistochemistry is vital in distinguishing these entities, with metastatic RCC typically expressing markers such as RCC-Ma, CD10, and PAX8. Immunohistochemical analysis was not performed in our case due to financial constraints.
In recent years, targeted therapies including multikinase inhibitors (MKI) such as cabozantinib and lenvatinib have become a cornerstone in the management of metastatic RCC. These agents inhibit pathways involving VEGF and platelet-derived growth factor (PDGF), thereby reducing tumor angiogenesis and progression. In case of disease progression or therapeutic resistance, mammalian target of rapamycin (mTOR) inhibitors such as everolimus may be employed.
Although targeted therapies can prolong survival, durable remission remains uncommon. Discontinuation of therapy after achieving complete response (CR) or sustained partial response (PR) has been reported in selected patients, particularly in those experiencing significant toxicity or expressing a preference to stop treatment [7–9]. Johannsen et al. reported that among 36 patients who discontinued targeted therapy after achieving CR or no evidence of disease (NED), 67% experienced recurrence within a median of 7 months; however, most responded to reintroduction of therapy [7, 8]. In contrast, our patient developed rapidly progressive recurrence after discontinuation of treatment.
Cutaneous metastases are associated with poor prognosis, particularly when multiple lesions are present. Reported 5-year survival rates do not exceed 8% [9]. Voog et al. demonstrated that the risk of mortality is higher within the first year following the appearance of skin metastases [10]. In our patient, widespread visceral metastases affecting the lungs, liver, and vertebrae were detected and the patient died 2 months after the diagnosis.
Although cutaneous metastases from RCC have been described previously, multifocal angiomatous skin lesions associated with rapidly progressive systemic dissemination remain uncommon [11]. This case highlights the importance of considering metastatic RCC in the differential diagnosis of vascular-appearing skin lesions, particularly in patients with a history of RCC. Early histopathological evaluation is essential for prompt diagnosis and appropriate treatment decisions.
CONCLUSIONS
Cutaneous metastases of renal cell carcinoma are uncommon and usually indicate advanced disease with poor prognosis. Because of their vascular appearance, these lesions may clinically mimic benign or infectious vascular conditions such as cherry angiomas, pyogenic granuloma, Kaposi sarcoma, or bacillary angiomatosis. Early recognition and histopathological confirmation are crucial for accurate diagnosis. Awareness of this rare manifestation may facilitate timely oncologic evaluation and management.







