INTRODUCTION
Atopic dermatitis (AD) is a chronic and recurring inflammatory dermatosis characterized by epidermal barrier dysfunction, leading to pruritus and eczematous lesions with age-dependent distribution patterns [1]. Updated epidemiological data on the prevalence and incidence of AD across different age groups and geographic regions provide insight into the global disease burden. The onset of AD occurs predominantly in early childhood, with approximately 80% of cases developing within the first years of life. Spontaneous remission is observed in nearly 60% of individuals during adolescence [2]. In the United States adult population, three population-based studies published in 2013, 2015, and 2019 reported AD prevalence rates of 10.2%, 7.2%, and 7.3%, respectively. Among children aged 5, 9, and 15 years, prevalence rates were reported as 15.0%, 15.1%, and 14.5%, yielding an average prevalence of approximately 14.8% across these age groups. Notably, the prevalence among children aged 0–5 years was substantially higher, reaching 24%. In the United Kingdom, the prevalence of AD was reported to be 12.3% among individuals aged 17 years and younger. Epidemiological trends suggest that the incidence and prevalence of AD have reached a plateau in Europe and North America, whereas a rising trend has been observed in other regions, particularly in parts of Asia. Studies conducted in China, Taiwan, and Malaysia reported prevalence rates of approximately 12.94% among children aged 1–7 years, 10.7% among those aged 6–8 years, and 13.4% among children aged 1–6 years, respectively [3]. In Indonesia, based on hospital visit data from patients aged 0-18 years treated at Dr. Wahidin Sudirohusodo Hospital and Hasanuddin University Hospital in South Sulawesi between 2022 and 2023, the prevalence of AD was reported to be 0.15% and 0.5%, respectively.
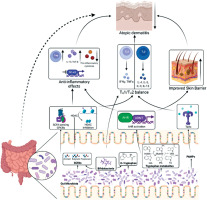
The pathophysiology of AD involves complex interactions between genetic mutations affecting epidermal barrier integrity and skin dysfunction, leading to alterations in lipid composition and immune dysregulation [2, 4, 5]. Increasing evidence links the gut microbiota to both disease development and the maintenance of immune homeostasis [6]. The gut microbiota is considered a key regulator of gut-skin axis mechanisms, capable of modulating skin function and inflammatory responses in individuals with AD [4, 7]. The pathophysiology of AD has been proposed to involve three major gut-skin axis pathways: immunological, metabolic, and neuroendocrine pathways [8]. Given the pivotal role of the gut microbiota in immune regulation and skin homeostasis, this article aims to elucidate the mechanisms of the gut-skin axis in AD.
MECHANISM OF THE GUT-SKIN AXIS
The gut microbiota can influence skin condition through bidirectional communication known as the gut-skin axis [2, 4]. Commensal gut bacteria produce various metabolites that directly affect the development of the immune system [9]. The interaction between Th1 and Th2 cells is essential for maintaining Th1/Th2 homeostasis, disruption of which may lead to chronic inflammation and autoimmune or allergic conditions. Previous studies in humans and animal models have demonstrated that intestinal microbes and their metabolic products play a significant role in the proliferation and differentiation of T and B cells, thereby inducing both cellular and humoral immune responses [10].
Intestinal barrier integrity prevents bacteria, toxins, and neurotransmitters from translocating from the gut into the bloodstream and subsequently to the skin [11]. Disorders of the intestinal tract are characterized by gut microbiome imbalance, reduced mucus layer thickness and immunoglobulin A secretion, and increased intestinal permeability [2]. Gut microbiome dysbiosis may influence AD symptoms through three main mechanisms: immunological, metabolic, and neuroendocrine pathways. Modulation of the gut-skin axis has emerged as a potential therapeutic strategy in AD, with probiotics and fecal microbiota transplantation showing promising results in several recent studies [12].
IMMUNOLOGICAL PATHWAY
The gut microbiome and its metabolites are primarily recognized by Toll-like receptors 2 and 4 (TLR2 and TLR4), which are implicated in the pathogenesis of AD, while interactions between bacteria and TLRs contribute to the maintenance of systemic immune homeostasis [6, 13]. Binding of gut microbiome pathogen-associated molecular patterns (PAMPs) to TLR2 helps maintain Th1/Th2 cell balance and supports skin homeostasis by enhancing the expression of tight junction proteins, such as claudins, and promoting antimicrobial peptide differentiation within the epidermis [13].
Gut microbiome dysbiosis may lead to translocation of microbes and their metabolites into the circulation and influence pathological skin manifestations through TLR signaling pathways. This process involves activation of thymic stromal lymphopoietin (TSLP), which induces the release of Th2-associated cytokines, including IL-4, IL-5, IL-13, and IL-22 [14]. Increased intestinal permeability in patients with AD enhances skin inflammation due to the systemic dissemination of toxins, poorly digested food components, and gut microbes via the bloodstream, including to the skin surface. Consequently, Th2-mediated inflammatory responses are amplified, leading to immune dysregulation and disease exacerbation [12] (Figure 1).
METABOLITE PATHWAY
The gut microbiome produces large amounts of metabolites, including short-chain fatty acids (SCFAs) and tryptophan derivatives, which can enter the circulation, disseminate throughout the body, and affect distant organs such as the skin, thereby maintaining its structure and integrity [15]. Gut microbiome dysbiosis disrupts intestinal epithelial integrity, a condition commonly referred to as leaky gut syndrome. Consequently, intestinal mucus production decreases and gut permeability increases [16]. This process may lead to the translocation of toxins, neurotransmitters, gut microbial pathogens, and immune mediators into the systemic circulation, ultimately affecting the skin in patients with AD [11].
Intestinal commensal microbiota produce SCFAs through the fermentation of indigestible carbohydrates, including acetate, propionate, and butyrate [17]. Soluble fibers, such as pectin, beta-glucan, and inulin, are more readily fermented by intestinal microbes than insoluble fibers, such as cellulose and lignin; therefore, increased intake of soluble fiber may enhance SCFA production. Several bacterial species, including Bifidobacterium spp., Lactobacillus spp., Clostridium butyricum, Faecalibacterium prausnitzii, and Akkermansia muciniphila, contribute to SCFA production through the fermentation of carbohydrates and proteins [17].
SCFAs exert anti-inflammatory effects by binding to G protein-coupled receptors and inhibiting histone deacetylases, thereby reducing nuclear factor kappa B pathway activation and promoting regulatory T cell differentiation as well as interleukin-10 and transforming growth factor beta production. Butyrate is metabolized by epidermal keratinocytes into long-chain and very-long-chain fatty acids, which serve as ceramide precursors [18]. Additionally, butyrate enhances the expression of filaggrin, loricrin, involucrin, keratin 1/keratin 10 [13], and transglutaminase-1 [18], promoting keratinocyte differentiation, strengthening epidermal barrier function, and preventing allergen penetration and sensitization. In patients with AD, gut microbiome dysbiosis is associated with reduced SCFA production, leading to impaired immune regulation, enhanced immunoglobulin E-mediated allergic responses, altered ceramide and cholesterol levels, and increased transepidermal water loss (TEWL) [19].
Gut bacteria, including Clostridium, Bacteroides, Bifidobacterium, and Lactobacillus, are capable of catabolizing tryptophan via the indole pathway [15]. Tryptophan metabolites generated through protein breakdown by tryptophanase, such as indole-3-acetate and indole-3-propionic acid, can activate the aryl hydrocarbon receptor in the cytoplasm and subsequently bind to the aryl hydrocarbon receptor nuclear translocator in the nucleus of keratinocytes. Activation of the aryl hydrocarbon receptor inhibits inflammatory responses, reduces thymic stromal lymphopoietin and Th2-associated cytokine expression, and enhances skin barrier function by regulating the production of filaggrin, loricrin, involucrin, and antimicrobial peptides in keratinocytes [13, 15].
NEUROENDOCRINE PATHWAY
Differences in the composition and relative abundance of the gut microbiome are associated with the production of various neurotransmitters and neuromodulators that correlate with the severity of AD symptoms [7, 8]. The gut and skin microbiomes interact through neuroendocrine signaling via both direct and indirect mechanisms [16]. The direct mechanism involves gut microbiome-derived metabolites, such as tryptophan, which can induce pruritic sensations in the skin [7, 12]. Several neurotransmitters produced by the gut microbiome, including γ-aminobutyric acid, acetylcholine, dopamine, and serotonin, may also modulate skin function through the nervous system [11]. Lactobacillus and Bifidobacterium species are capable of producing γ-aminobutyric acid, which can suppress pruritic sensations in the skin [7, 12].
Indirect mechanisms involve neuroendocrine molecular alterations driven by cytokines produced in response to changes in gut microbiome composition. The gut microbiome indirectly regulates circulating cytokine levels, thereby influencing brain function, anxiety, and stress. These effects may lead to increased cortisol levels, which can further alter microbiome composition, epithelial permeability, and the functional integrity of both the gut and skin [8]. Stress responses modify circulating concentrations of neuroendocrine mediators, including catecholamines (norepinephrine and epinephrine) and glucocorticoids, which can suppress antimicrobial peptide synthesis in the epidermis and promote the growth and virulence of Staphylococcus aureus, ultimately contributing to skin barrier disruption and inflammation [7, 16].
GUT MICROBIOTA AND ATOPIC DERMATITIS SEVERITY
The severity of AD is significantly correlated with the degree of gut microbiota dysbiosis. Several clinical studies have shown that patients with severe AD exhibit lower gut microbiota alpha diversity compared with those with mild to moderate disease. Research by Liu et al. demonstrated that infants with AD had a significant decrease in Clostridium sensu stricto and Collinsella, along with an increased abundance of Parabacteroides (p < 0.05). Importantly, alterations in these microbial taxa (Bacteroides and Clostridium sensu stricto) were significantly associated with SCORAD scores (p < 0.05) [20].
The observed reduction in Clostridium sensu stricto and Collinsella may lead to decreased SCFA production, resulting in impaired Treg cell activity and increased susceptibility to allergic inflammatory pathway activation. This imbalance promotes elevated expression of pro-inflammatory cytokines, including IL-4, IL-5, and IL-13, which are central to AD pathogenesis. In contrast, increased abundance of Parabacteroides and Bacteroides may reflect dysbiosis-driven metabolic shifts, such as enhanced sphingolipid and glycosphingolipid pathway activity. Metabolites generated through these pathways have the potential to exacerbate skin inflammation by disrupting epidermal barrier integrity and inducing keratinocyte apoptosis and mast cell activation. Furthermore, Bacteroides-derived metabolites may stimulate pro-inflammatory adaptive immune responses, including increased Th1 and Th17 differentiation and reduced Treg cell populations, all of which contribute to greater AD severity. The correlation between increased Bacteroides abundance and higher SCORAD scores suggests that AD severity may be systemically influenced through metabolic and immunological pathways modulated by the gut microbiota [21–24].
Systemic levels of pro-inflammatory cytokines, such as IL-6, TNF-α, and IL-1β, have also been reported to be elevated in patients with severe gut microbiota dysbiosis, indicating the involvement of systemic immune activation [25]. In addition, increased total serum IgE levels and peripheral eosinophilia have been observed in association with reduced abundance of probiotic bacteria, suggesting that microbiota imbalance promotes exaggerated Th2 pathway activation. One important mechanism underlying this relationship involves TLR signaling. In patients with severe AD, TLR4 activation by Gram-negative bacterial LPS contributes to NF-κB activation, which subsequently enhances transcription of inflammatory genes. Conversely, depletion of commensal species such as Akkermansia muciniphila and Bifidobacterium longum is associated with reduced expression of anti-inflammatory cytokines, including IL-10 and TGF-β, thereby exacerbating immune dysregulation [26].
POTENTIAL USE OF PROBIOTICS IN ATOPIC DERMATITIS
The use of probiotics as an immunomodulatory therapy in AD has received considerable attention over the past two decades. Probiotics, defined as live microorganisms that confer health benefits when administered in adequate amounts, exert their effects by stabilizing the gut microbiota, strengthening the mucosal barrier, and promoting immune tolerance [24, 27]. Several species have been shown to be potentially beneficial in the context of AD, including Lactobacillus rhamnosus GG, Bifidobacterium breve, Lactobacillus casei, and Lactobacillus plantarum. A study by Yoon et al. demonstrated that oral administration of a probiotic mixture containing Lactobacillus and Bifidobacterium species for 4 weeks resulted in a significant reduction in SCORAD scores (p < 0.001) and TEWL (p = 0.028) in pre- and post-intervention assessments. Additionally, fecal calprotectin levels, a marker of gastrointestinal inflammation, were significantly reduced following the intervention (p = 0.038) [28].
The proposed mechanisms of probiotic action in AD include increased expression of anti-inflammatory cytokines (IL-10, TGFβ), expansion of TReg cell populations, and suppression of Th2-mediated immune responses. Probiotics may also enhance intestinal mucosal integrity by upregulating tight junction proteins, such as claudins and occludin, thereby limiting antigen and endotoxin translocation that could otherwise exacerbate systemic and cutaneous inflammation. Importantly, the immunomodulatory effects of probiotics are strain- and dose-dependent. Multistrain formulations, particularly combinations of Lactobacillus and Bifidobacterium species, have been reported to exert stronger synergistic effects than single-strain preparations.
Despite these promising findings, clinical outcomes remain heterogeneous due to substantial variability in study design, population characteristics, probiotic strains, dosages, and duration of intervention. Although probiotics appear to be safe and may have potential as an adjunctive therapy in selected patients with AD, particularly those with a family history of atopy, current clinical guidelines do not recommend their routine use in AD management due to insufficient and inconsistent evidence [29–32].
FECAL MICROBIOTA TRANSPLANTATION IN ATOPIC DERMATITIS
Fecal microbiota transplantation (FMT) is a therapeutic approach aimed at restoring gut microbiota balance through the transfer of fecal material from a healthy donor to an individual with dysbiosis. In AD, immune dysregulation and disruption of gut microbiota homeostasis are central elements of disease pathogenesis. Gut dysbiosis, defined as an imbalance between commensal and pathogenic microorganisms, is believed to promote systemic immune activation and exaggerated inflammatory responses, including in the skin. Under physiological conditions, the gut microbiota supports mucosal immune tolerance by promoting Treg differentiation and the secretion of anti-inflammatory cytokines such as IL-10 and TGF-β. In dysbiotic states, the production of microbiota-derived metabolites, particularly SCFAs such as butyrate, is reduced. Butyrate exerts potent immunomodulatory effects, including inhibition of Th2 cell differentiation, which is pathologically dominant in AD. Consequently, dysbiosis-associated reductions in SCFA production may enhance Th2-driven inflammation and exacerbate chronic skin disease [23, 33].
FMT aims to restore gut microbiota composition toward an eubiotic state, thereby improving mucosal and systemic immune regulation. This intervention has the potential to increase colonization by beneficial commensal taxa, including Faecalibacterium prausnitzii and Clostridia clusters IV and XIVa, which are known to support Treg differentiation and reinforce mucosal barrier integrity. In parallel, restoration of microbial balance may suppress the expansion of opportunistic pro-inflammatory taxa, such as members of the Proteobacteria phylum and the Enterobacteriaceae family, which are frequently enriched in patients with atopic disorders. Experimental studies using AD models have demonstrated that FMT from healthy donors reduces IL-4 and IL-13 expression, increases intestinal Treg populations, and leads to decreased eosinophil and mast cell infiltration in the skin, accompanied by improved epidermal integrity. These effects are associated with increased SCFA production and restoration of commensal bacteria such as Faecalibacterium and Akkermansia [34–36].
Clinical evidence remains limited but promising. A crossover clinical trial by Mashiah et al. in adults with moderate-to-severe AD, in which four FMTs were administered following two placebo treatments, demonstrated a mean SCORAD reduction of 59% (p = 0.011) and 85% at 8-week follow-up (p = 0.018). A reduction of ≥ 50% in SCORAD was achieved in 77% of patients, while 66.7% achieved a ≥ 75% reduction. No serious adverse events were reported, and microbiota analyses confirmed successful transfer of specific donor-derived microbial strains [37]. Similarly, a study by Liu et al. reported significant improvements in EASI scores in patients with moderate-to-severe AD receiving FMT compared with controls. The intervention group also demonstrated reductions in inflammatory markers, including TNF-α, total serum IgE, and Th2- and Th17-associated responses, without serious adverse events [38].
Despite these encouraging findings, FMT is not recommended for routine use in the management of AD and should be clearly discouraged outside of clinical trials, in accordance with current clinical guidelines.
Emerging evidence supports the concept of targeting the gut microbiota as a novel therapeutic avenue in AD. Future research should focus on identifying specific microbial taxa and metabolites that modulate immune responses and skin barrier function. Well-designed longitudinal and mechanistic studies are needed to clarify causal relationships between gut dysbiosis and AD pathogenesis. Moreover, randomized controlled trials evaluating microbiota-targeted strategies including precision probiotics, prebiotics, synbiotics, and next-generation microbiome-based therapies are warranted. Advances in metagenomic and metabolomic profiling may ultimately enable personalized approaches to microbiome modulation, contributing to more effective and sustainable AD management.
CONCLUSIONS
The pathophysiology of AD involves a complex interplay between immune dysregulation and impairment of the skin barrier. The gut-skin axis plays a pivotal role in maintaining immune homeostasis and preserving skin integrity in individuals with AD. Dysbiosis of the gut microbiota may disrupt cutaneous defense mechanisms and promote inflammatory responses, thereby contributing to the development and progression of AD.