INTRODUCTION
Lichen sclerosus (LS) is a chronic inflammatory skin condition of uncertain aetiology that most commonly affects the genital area in individuals of all genders, with a notably higher occurrence in postmenopausal women and young girls before puberty. Approximately 85% of LS cases are confined to the genital region, while extragenital involvement is uncommon, occurring in about 15–20% of cases [1]. The clinical manifestations of vulvar LS, especially in girls, usually include porcelain-white atrophic plaques, sometimes with a figure of eight or hourglass configuration involving both the vulva and perianal area [2]. Extragenital lichen sclerosus (EGLS) typically presents as asymptomatic, porcelain-white, atrophic plaques that may coalesce into larger areas of sclerotic skin. These lesions are frequently located on the upper trunk, neck, shoulders, and proximal extremities. Less commonly, EGLS can involve atypical sites such as the breasts, face, and acral areas, including the hands and feet [3]. LS is frequently associated with other autoimmune diseases. Studies have shown a higher prevalence of thyroid disorders (especially Hashimoto’s thyroiditis), vitiligo, alopecia areata, and pernicious anaemia in patients with LS. This comorbidity is observed in both paediatric and adult populations, suggesting a potential shared immunopathogenic mechanism [4]. This report describes the case of an 11-year-old girl diagnosed with genital and extragenital lichen sclerosus involving the palms and feet, with concurrent vitiligo.
OBJECTIVE
The aim of this case presentation is to highlight the importance of recognizing atypical presentations of LS in pediatric patients and the need for comprehensive evaluation when autoimmune skin disorders coexist. In addition to the case description, a brief review of the literature on previously reported cases of palmar LS is provided, offering comparative clinical insights and emphasizing the rarity of such manifestations in children.
CASE REPORT
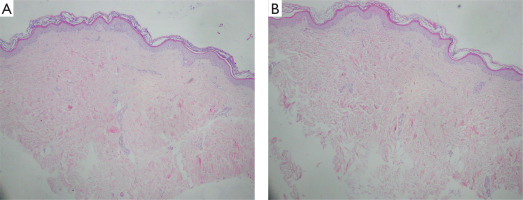
An 11-year-old girl presented to the dermatology outpatient clinic with a 4-year history of asymptomatic hypopigmented patches in the vulvar area. Approximately 2 years after the onset of the vulvar lesions, additional hypopigmented patches appeared on the palms (Figures 1 A, B), wrists, heels, and ankles, along with typical vitiligo patches on the eyelids. Small, brown macules on a depigmented background were also observed on the forearms and ankles. Based on the initial vulvar involvement, a diagnosis of LS was made. However, the subsequent development of widespread lesions raised suspicion of an alternative or coexisting dermatologic condition. A skin biopsy was taken from the lesion in the ankle area, and the histopathological findings were most consistent with LS (Figures 2 A, B). The patient was initially treated with topical clobetasol, followed by maintenance therapy with topical tacrolimus, targeting the vulvar LS. The lesions on the palms, heels, and ankles were treated with topical clobetasol and a topical calcineurin inhibitor with no significant improvement. Notably, the vitiligo patches on the eyelids showed improvement with treatment by the topical calcineurin inhibitor and eventually resolved completely.
Figure 1
A, B – Clinical presentation of hypopigmented patches on the palms with prominent acrosyryngia in a 11-year-old girl
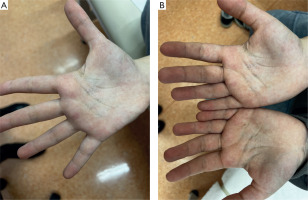
Figure 2
Magnification: 4× (A) and 10× (B). The epidermis is thin with flattened rete ridges and a thickened stratum corneum. The underlying connective tissue stroma appears subtly homogenized, with no inflammatory infiltrates. These histopathological features are not fully representative for lichen sclerosus and morphea
DISCUSSION
LS is well characterized in adults, but data on its presentation in children are still limited [5]. In paediatric cases, particularly among girls, LS most frequently affects the anogenital area, with approximately 94.6% presenting lesions in this region. Only 5.4% exhibit either isolated extragenital lesions or a combination of genital and extragenital involvement [6]. Characteristic findings of genital LS in girls encompass porcelain-white, atrophic plaques arranged in a “figure-eight” pattern involving the vulva and perianal area. Advanced cases may exhibit fissures, erosions, scarring, and architectural changes such as labial fusion or clitoral phimosis [7]. Common extragenital sites include the trunk, neck, shoulders, and upper limbs. While LS itself is already considered a rare condition, its manifestation on the palms and feet is exceptionally uncommon. To date, only a few cases involving lesions on the palms or hands have been reported in the literature, with varying clinical presentations. Among them, Purres and Krull [8] and Petrozzi et al. [9] describe cases that are clinically similar. Notably, none of these cases have involved paediatric patients (Table 1) [8–16].
Table 1
Extragenital LS cases with palmar involvement
| Authors | Age/sex | Genital involvement | Distribution | Clinical presentation |
|---|---|---|---|---|
| Steff et al. [10] | 56 F | Yes | Dorsal feet, phalanges, palms, soles, genital area | Ivory-white papules on dorsal feet and distal phalanges; keratotic, centrally depressed papules in palmar creases; erythema of soles and thenar/hypothenar eminences |
| de Viana et al. [11] | 57 F | Yes | Soles, palms, paranasal sinuses perigenital area | Grouped hypopigmented papules and plaques, later extension to perigenital area |
| Herz-Ruelas et al. [12] | 60 F | No | Palms, anterior wrists, soles, trunk, lower extremities | Sclerotic plaques with a shiny/wrinkled surface on body lesions and soles. Small coalescing bullae noticeable upon palpation’; disabling pain on standing and walking |
| Seyffert et al. [13] | 69 M | No | Palms, arms, dorsal fingers, wrists, trunk, thighs | Erythematous, ivory, hyperpigmented atrophic plaques ranging in size from 0.5 cm to 4 cm on the proximal extremities and trunk; diffuse erythema with atrophic and bullous ivory-yellow plaques on palms with dermatoglyphic and eccrine ostia accentuation extending onto the flexural wrist |
| Sánchez-Mateos et al. [14] | 71 F | Yes | Dorsal hands, palms, wrists, fingers, genital area | Pale-yellow, firm waxy plaques evolving into pseudobullous, atrophic lesions on hands; nail ridging; vulvar pain and whitening |
| Purres and Krull [8] | 71 F | Yes | Both palms, upper back, chest, shoulders, forearms, genital area | Atrophic white papules and plaques on palms with keratotic plugs; widespread white plaques on trunk and forearms, some with follicular plugs; vulvar atrophy and whitening |
| Heibel et al. [15] | 67 F | No | Palms, wrists, toe webs, sides of feet and toes | Multiple, white, hyperkeratotic papules pain and soreness while walking and during palpation; no pruritus |
| Petrozzi et al. [9] | 57 F | No | Soles and palms | Multiple white papules on soles and few depressed, slightly keratotic papules in central palms; pain when walking |
| Aggarwal et al. [16] | 58 M | No | Dorsal hands, palms, soles, dorsal feet, wrists | Hypopigmented to depigmented guttate macular lesions, polygonal, well-defined, atrophic lesions with wrinkled surface; Koebner phenomenon and erythematous halo on dorsal hands |
Although LS can be asymptomatic, common symptoms in children include pruritus, pain, dysuria, constipation, and burning sensations in the anogenital area [6]. Behavioural changes may also occur, often driven by persistent discomfort and the child’s desire for attention and support from caregivers [17, 18]. Due to its rarity and nonspecific presentation, EGLS in children can be misdiagnosed, underscoring the importance of considering LS in the differential diagnosis of paediatric patients presenting with such lesions [19, 20].
LS is commonly associated with a variety of autoimmune conditions, including vitiligo vulgaris, alopecia areata, type 1 diabetes mellitus, thyroid dysfunction (especially Graves’ disease), atopic dermatitis, psoriasis, polymyalgia rheumatica, lupus panniculitis, primary biliary cirrhosis, myositis, systemic lupus erythematosus, and pernicious anemia [21]. A recent comprehensive study involving 744 female patients with vulvar LS revealed that 24.5% of children and 34.6% of adults had concurrent autoimmune diseases. The most prevalent autoimmune conditions identified were psoriasis, Hashimoto’s thyroiditis, lichen planus, and vitiligo [4]. Genetic studies have identified associations between LS and the HLA class II locus DQ7, particularly in women, suggesting a genetic predisposition to both LS and other autoimmune disorders. A study of 30 female children with vulvar LS demonstrated that 66% were positive for HLA DQ7, a genetic marker associated with autoimmune diseases. Furthermore, 56% of their parents or grandparents had a history of autoimmune diseases, indicating a strong familial predisposition [22].
In this case, the patient also presented with vitiligo, further complicating the diagnostic process. Vulvar LS and vulvar vitiligo share similar clinical presentations, both characterized by white macules and patches, posing a diagnostic challenge for clinicians. In paediatric patients with LS, especially girls, petechiae may be common in this region [23]. However, in cases where clinical features are atypical for LS and petechiae are present, the possibility of sexual abuse should be carefully considered and appropriately evaluated as part of the differential diagnosis [24]. In uncertain cases, a biopsy is recommended. Histopathological features of LS may vary depending on the stage of the disease and can overlap with other dermatoses [25, 26].
In early LS, both clinical and histological features may be nonspecific. Histology often reveals subtle changes such as hyperkeratosis, hypergranulosis of adnexal structures, mild irregular acanthosis, focal thickening of the basement membrane, and subepithelial oedema with homogenized collagen and dilated vessels. A lymphocytic infiltrate – lichenoid or interstitial – may be present, along with epidermal exocytosis and occasional lymphocytic vasculitis. In chronic lesions, classic findings include epidermal atrophy with flattened rete ridges, vacuolar interface changes, dermal hyalinization, loss of elastic fibres, and a band-like lymphocytic infiltrate. Notably, prominent acanthosis can still appear, particularly in vulvar LS [27, 28].
Differentiating LS from vitiligo can be facilitated by histochemical and immunohistochemical staining [29]. Fontana-Masson staining assesses melanin content, while immunohistochemical markers such as Melan-A, MITF, tyrosinase, and NKI/beteb evaluate melanocyte presence and activity [30]. Among these, tyrosinase staining demonstrated the most significant difference in melanocyte activity between LS and vitiligo, highlighting its diagnostic value [30].







