Introduction
Crohn’s disease (CD) is a chronic, incurable inflammatory disease of the gastrointestinal tract, usually diagnosed during childhood or adolescence [1]. Growth failure and malnutrition are typical complications of inflammatory bowel disease (IBD) in children. Its aetiology is multifactorial, but there are two main factors contributing to that state: chronic intestinal inflammation leading to poor absorption, which finally causes malnutrition, and the direct effect of proinflammatory cytokines secreted by inflamed intestine [2, 3].
The treatment strategies for CD in children have evolved markedly in recent years. Two meta-analyses have shown that exclusive enteral nutrition has comparable effectiveness to traditional corticosteroid therapy in the treatment of children with active CD [4, 5]. It should also be emphasized that steroid therapy has many side effects, including a negative impact on bone growth and mineralization processes, which is particularly important in children. Exclusive enteral nutrition (EEN) not only contributes to growth gain but also improves the skeletal system condition [6]. What is more, the latest meta-analysis (2017) showed not only the comparable effectiveness of EEN and corticosteroids in the induction of clinical remission in children with active CD, but also a higher efficiency of EEN in the induction of mucosal healing [7]. These findings were confirmed in 2019 by Pigneur et al.; in their study clinical remission was achieved in 100% patients on EEN and in 83% patients on steroids, but the mucosal healing rate was significantly higher in the EEN group (89%) compared with the steroid group (17%) [8]. It is known that obtaining mucosal healing in patients with CD is associated with better outcomes in long-term observation [9–11].
Similar effectiveness in induction of remission, no side effects, positive effect on child’s development and better efficiency in induction of mucosal healing are factors in favour of using EEN instead of corticosteroid therapy as a first-line treatment for young patients with active CD. On the other hand, it is known that in clinical practice EEN is not a convenient and easy therapeutic method to commence. The necessity for the patient’s high motivation, especially when the nasogastric tube placement is needed, is a common reason for renouncement this kind of treatment in children and adolescents.
There are several potential mechanisms of the impact of EEN on the inflamed intestinal mucosa, including the avoidance of enteral antigens resulting in improvement in intestinal permeability or beneficial changes to the gut microbiome [12–14]. Also, the industrial diet directly affects intestinal mucosa through reduction of secretion of pro-inflammatory cytokines [15]. Numerous studies have analysed the impact of modifications in industrial diet composition, including changes in the type of fat, protein, and bioactive peptide content/addition, but there is still no consensus on the most appropriate type of formula for CD patients [5, 16]. According to current guidelines, generally the standard polymeric formula is recommended [17].
However, it seems that the addition of bioactive peptides to the industrial diet may give additional benefits in the treatment of CD due to their anti-inflammatory effect [13, 18]. Bioactive peptides are specific growth factors. Transforming growth factor-β (TGF-β) comprises multifunctional regulatory peptides produced by various cell types. It is known that TGF-β affects the immune system regulation, as well as cell growth and differentiation both through autocrine and paracrine pathways. These cytokines control the processes of cell differentiation, proliferation, and activation (including lymphocytes, macrophages, and dendritic cells). Therefore, TGF-β plays a key role in anti-inflammatory processes and in prevention of autoimmune diseases [19]. Due to the therapeutic and anti-inflammatory properties of TGF-β, it seems that its addition to the industrial diet may be effective in CD treatment [20, 21].
Currently, only a few paediatric cohort studies analysing the effect of enteral nutrition based on formulas with TGF-β (MODULEN IBD) are available [8, 15, 22–24]. In all studies, patients receiving MODULEN IBD entered clinical remission. However, differences in duration of the therapy, method of industrial diet administration (oral/through nasogastric tube), and its daily amount (exclusive/partial enteral nutrition) limit the ability to summarize the results. Moreover, there are only 3 studies on EEN with an industrial diet provided for several weeks, which according to the current guidelines is considered the most effective method of treatment. The same 3 studies are also the only ones that assessed the impact of EEN on mucosal healing [8, 22, 24].
Aim
Therefore, there is still a need for properly designed studies determining whether total enteral nutrition based on the industrial diet with TGF-β is effective in the induction of clinical remission and mucosal healing in children with active CD.
Material and methods
Twenty patients with active CD (based on clinical symptoms, The Paediatric CD activity index (PCDAI) score, faecal calprotectin (FCP) level, and endoscopic examination) in the age group 4–18 years were included in the study.
The qualification for nutritional treatment was made after the full endoscopic examination (both gastro- and colonoscopy were performed), and the SES-CD scale was used to assess macroscopic disease activity.
Exclusive enteral nutrition was provided for 6 weeks to all children. Patients received “Modulen IBD” orally or through a nasogastric tube in cases of intolerance to oral supply. In addition to the industrial diet, drinking of water was allowed.
Daily caloric intake was established based on patient’s age and nutritional status. In malnourished patients, daily caloric intake was increased to 120–150% relative to recommendations for healthy peers (based on body weight, height, and body mass index (BMI) for age and sex).
Clinical remission was defined as PCDAI < 10, and mucosal healing (MH) was established using FCP level (n < 200 mg/kg).
Nutritional state, PCDAI score, and FCP were assessed at baseline and 4 weeks after the therapy (week 10).
Results
Among 20 children included in the study, there was a predominance of boys: male/female – 14/6. The range of patients’ age was 8.6–17.1 years, and median age was 14 years. At baseline the majority of patients (65%) presented with mild course of the disease (PCDAI: 7.5–27.5). Baseline FCP > 200 mg/kg was reported in 90% of patients. Before EEN commencement, undernutrition was observed in 30% of children, which was established by BMI below the third percentile according to Polish charts. In 25% of patients, a growth deficit was observed (defined as growth below third percentile) before nutritional therapy introduction (Table I).
Table I
Patients’ baseline characteristics (n = 20)
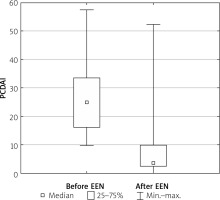
The mean decrease in PCDAI score following EEN commencement was statistically significant (from 25.6 ±12 to 5.4 ±10, p < 0.05) (Figure 1).
Full remission (defined as PCDAI < 10) was achieved in 65% of patients, clinical response in another 30% of them, and 5% of children did not respond to the treatment.
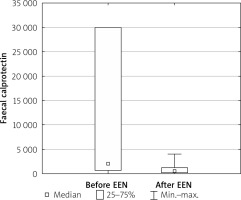
At week 10 (4 weeks after the end of EEN) a statistically significant decrease in FCP level was observed. Mean values of 3380 ±7746 at baseline declined to 1046.6 ±1219 at week 10, p < 0.05 (Figure 2). However, complete mucosal healing was stated only in 25% of patients.
Table II presents values (mean and range) of inflammatory markers (PCDAI and FCP) at baseline and week 10.
Table II
Markers of disease activity at baseline and after nutritional treatment with EEN (n = 20)
All patients, apart from 1 who was fed with a nasogastric tube, accepted oral intake of industrial formula. EEN was generally well tolerated. Initially in 20% of patients the symptoms of industrial diet intolerance were observed, but they receded within the first days of the therapy. The intolerance was manifested as stomach ache in 2 patients and nausea in 2 other cases.
The recommended daily intake of the formula was achieved in 95% of children. Only one child was unable to intake the prescribed amount of the diet due intolerance.
Improvement in BMI status was reported at the end of the treatment in all patients. The mean increase in BMI score was 0.91, and it was greater in the malnourished group compared to patients with normal nutritional status (1.19 vs. 0.62, respectively). After the treatment two-thirds of children with malnutrition had their BMI score within the normal range. The overall increase in body height was obtained in 55% of children and in 80% of those with initial growth failure. The mean growth gain was 1 cm, and it was greater in the group with initial growth deficit relative to patients with normal height values for age at baseline (1.5 cm vs. 0.8 cm, respectively) (Table III).
Table III
Patients’ anthropometric characteristics (body mass, height, and BMI) at baseline and after EEN commencement (n = 20)
Discussion
According to the current guidelines of the European Crohn’s and Colitis Organization (ECCO), EEN is recommended as first-line therapy to induce remission in children with active luminal CD [17]. Partial EN should not be used for induction of remission. The duration of EEN as induction therapy is usually 6–8 weeks. In our study all patients received EEN for 6 weeks, as recommended.
Because nutritional treatment promotes MH, restores bone mineral density, and improves growth, it should be preferred over corticosteroids for all children with inflammatory intestinal luminal disease, including colonic involvement [17, 25]. However, in clinical practice, due to the necessity for patient’s high motivation, especially when nasogastric tube placement is needed, in some cases EEN is more difficult to commence than pharmacotherapy.
At present, no placebo-controlled randomized controlled trial (RCT) of EEN with exclusive liquid formula feeds has been conducted in children with CD, but there have been several RCTs comparing EEN to standard treatment.
Three meta-analyses have demonstrated that the overall combined remission rate for EEN in paediatric CD is 73% (relative risk (RR) = 0.95, 95% confidence interval (CI): 0.67–1.34 and RR = 0.97, 95% CI: 0.7–1.4) [4, 5, 26]. The results from our study are comparable: 65% of patients entered full clinical remission following nutritional therapy with EEN.
According to published data, MH rates in children treated with EEN have ranged from 19% to 75% [27–30]. However, differences in the definition of MH in these studies limit the ability to summarize the results. The only RCT assessing MH as an outcome of EEN with Modulen IBD compared with corticosteroids demonstrated a clear superiority after 8 weeks of EEN, with rates of 89% vs. 17%, respectively [8]. In that study MH was assessed in endoscopy while in our study FCP was used to define MH because this biomarker seems to correlate best with endoscopic examination and is considered as good marker of MH [31]. At week 10 we observed a statistically significant decrease in FCP levels, which may suggest that exclusive enteral nutrition promotes healing of intestinal mucosa as well. However, only 25% of patients achieved complete mucosal healing defined as FCP below 200 mg/kg. A likely explanation for this unsatisfactory result is that the control measurement of calprotectin was done 4 weeks after the end of EEN and the start of the regular diet. Recently Levine et al. showed that after 6 weeks of EEN with “Modulen IBD” the FCP levels dropped significantly, but they increased again at week 12 (6 weeks after the reintroduction of standard food) [24]. Maybe if we performed the measurement of FCP at week 6, immediately after the end of the EEN, the results would have been better. Moreover, in the study by Levine et al., after 6 weeks of EEN, only 15% of children achieved complete MH defined in the same way as in our study, i.e. FCP below 200 mg/kg. The authors explained the poor results by the very high calprotectin levels at enrolment. The median baseline FCP level in their patients was comparable to ours, i.e. 2647 vs. 3380 µg/kg, respectively. The high FCP levels at baseline (meaning huge mucosal damage) is another potential explanation of the low rate of patients with complete MH after 6 weeks of EEN in our group.
Hartman et al. performed a retrospective study including the charts of 28 patients, to summarize the experience of several paediatric gastroenterology units in Israel using “Modulen IBD” supplementation in children and adolescents with CD [7]. They observed that a diet with “Modulen IBD” was associated with a statistically significant decrease in PCDAI, from 34.3 to 15.7 (p < 0.0001), which is consistent with our findings. Moreover, similarly to our study, the children supplemented with “Modulen IBD” also showed improvement in BMI, suggesting an additional advantage of nutritional therapy in CD patients.
A Spanish study assessing the efficacy of 6–8-week EEN as a primary therapy for children with CD (14 patients included in the study) and its impact on MH (defined by FCP, as in our study) demonstrated that after 8 weeks of EEN, 85% of patients were in clinical remission and FCP levels declined significantly without reaching normal levels [13]. These results confirm that the duration of EEN as induction therapy should be 6–8 weeks, and it takes more time to promote MH; FCP levels, although significantly reduced, were still above normal levels. In our study fewer (65%) patients were in clinical remission after EEN commencement, which may be due to shorter duration of nutritional treatment (6 vs. 8 weeks), which also suggests that the longer EEN is provided, the better the outcomes. Moreover, Levine et al. showed the remission rate, defined as in our study as PCDAI < 10 points, was lower at week 6 than at week 12 (6 weeks after the end of EEN and the start of regular diet) [24]. In our group the PCDAI was assessed 4 weeks after the end of EEN, which could be why the remission rate was lower than in the Spanish study where PCDAI was evaluated immediately after the end of EEN [13].
Although most published studies have proven the efficacy of EEN in induction of remission in paediatric CD patients, there is still an unresolved issue. Comparing our results with others we can conclude that the 6-week course of EEN led to a decline in FCP level but was too short to induce complete mucosal healing, especially in patients with high baseline FCP results. The second point is to pay attention to the lack of a strategy to reintroduce normal food after the EEN. In our patients the remission rate was lower than in studies where the assessment was done immediately after the end of nutritional therapy [13, 24]. This suggests that after EEN a special diet is needed to maintain remission. There are promising results from a study by Levine et al. – they showed that partial enteral nutrition plus a special diet (Crohn’s Disease Exclusion Diet) has the same effectiveness as EEN in induction of remission in children with Crohn’s disease. This new idea of nutritional treatment is easier to apply in patients, and it defines the next stages of re-introduction of as normal diet [24].
Nonetheless, more properly designed studies are needed to establish the optimal method of nutritional therapy in paediatric CD patients.
Conclusions
In our study including 20 children with active CD a 6-week course of EEN was an effective and well tolerated method of treatment. Nutritional therapy induced full clinical remission and led to a decline in FCP level (as marker of MH) in the majority of patients, but it was not sufficiently effective to induce complete mucosal healing. Another important benefit from that treatment was the improvement of the children’s nutritional status and growth velocity.