Introduction
It is known that the treatment of type 1 diabetes is a challenge for both the patient and the doctor. Despite the constant struggle, most patients are unable to achieve normoglycaemia and are at risk of episodes of hypoglycaemia, diabetic ketoacidosis, or long-term vascular complications resulting from insufficient diabetes control [1]. This disease has also become a challenge for the world of technology. Advances in technology offer the opportunity to improve glycaemic control by reducing the aforementioned complications and burdens while improving quality of life. Closed-loop insulin delivery systems (also known as “artificial pancreas”) take this technology to a larger scale by integrating CGM systems with an insulin pump and an algorithm that automates insulin delivery. These systems are collectively referred to as hybrid closed loop systems (HCL systems). Currently, there are 2 systems: the commercial “auto-mode” and the “Do-it yourself” Artificial Pancreas System (DIYAPS) [2]. The use of technological progress is becoming an important and quite interesting point of care for diabetics, ranging from continuous glucose monitoring systems, through personal insulin pumps, and finally to the use of a combination of both systems to automate the insulin supply [3]. Citing evidence-based medicine, diabetological care in patients with T1D departs from the independent use of the CGM system and the insulin pump, replacing it with the HCL system option [4].
Commercial HCL systems
Closed-loop hybrid systems are characterized by automated algorithm-based insulin delivery and patient-initiated insulin delivery (e.g. post-meal boluses). There are several systems using hybrid closed loop technology currently offered on the global market: Medtronic’s System MiniMed™ 670G, and MiniMed™ 780G (SmartGuard™); Tandem’s T slim x2 Control IQ; Insulet’s Omnipod5Ò-Automated mode (HypoProtect™)[5]; and CamAPS FX DanaRS or Ypso pump [6]. Insulet’s Omnipod5Ò-Automated mode (HypoProtect™) is currently undergoing clinical trials [7]. Technology is moving forward, so advanced systems are being developed that include a developed algorithm with individualization of primary target points, an automated correction bolus function, and improved stability of the automated mode. The automated correction bolus feature is an innovation, which is why we refer to these systems as advanced hybrid closed-loop (AHCL). The AHCL systems include MiniMed™ 780G (SmartGuard™); Tandem’s T slim x2 Control IQ; Insulet’s Omnipod5Ò-Automated mode (HypoProtect™); and CamAPS FX.
The purpose of this paper is to present commercial HCL and AHCL systems devices in 2022 with a scientific perspective.
Discussion
Hybrid closed-loop systems vary; Table 1 [2, 8–39] shows the systems from each manufacturer that are present on the market (2021), allowing us to follow the intentions of the manufacturers, understand their functions, and compare the products. Each system has individual advantages and disadvantages, which we can trace in Table 2 [40, 41]. The imperfections of the systems make the technological progress faster and faster, and this guarantees that newer systems will appear on the market to meet more patients’ expectations. When we look at the advantages of HCL systems we can see many similarities, which are improvements in parameters that reflect diabetes control. The auto-mode algorithms in HCL “learn the patient” and adjust insulin delivery individually for each person. That provides better glycaemic control and safer therapy. However, the existence of different algorithms in systems causes difficulties in interpretation and the need to learn each algorithm. The aforementioned studies indicate that therapy with HCL systems reduces the time spent in hypoglycaemia, while extending the time spent in the target range, aiming to reduce glycaemic fluctuations, and improving the patient’s quality of life in all age groups [42, 43]. Therefore, HCL systems are an essential step forward for patients with diabetes, enabling them to live longer healthier lives and be safer and more comfortable. However, it is important to remember that proper patient education plays a key role in maximizing the benefits of treatment with all HCL systems [22]. For some patients, high-technology systems can add stress and burden, manifesting as switching out of the automated mode when using HCL [2]. Therefore, it is extremely important that technology strives to develop devices that are accessible for use by the common patient, regardless of their socioeconomic status.
Figure 1
Improvement of TIR (%) during therapy using the HCL and AHCL systems in studies [1, 8, 22–32]
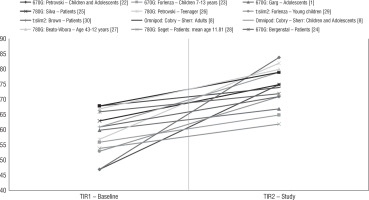
Table I
Characteristics of the commercial HCL and AHCL systems in a table of the research paper by Leelarathna et al. [2], Cobry et al.[8], and Braune et al. [9], with authors’ modification
| Name of HCL system | Medtronic Minimed™ 670G [10] | Medtronic Minimed™ 780G [11] | Tandem’s T slim x2 Control IQ [12–14] | CamAPS FX DanaRS or Ypsomed pump [6, 15] | Insulet’s Omnipod5-Automated mode (Hypoprotect™) [8, 16] |
|---|
| Name of Pump | 670G | 780G | Tandem T: Slim x2 | Dana RS pump, Ypsomed pump | Omnipod (Pod + PDM - Personal Diabetes Manager) |
| Name of Sensor | Guardian 3 | Guardian 3, Guardian 4 | Dexcom G6, Dexcom G7(coming soon [17, 18]) | Dexcom G6 | Dexcom G6, Decom G7 (coming soon [17, 18]) |
| Duration of the Sensor (days) | 7 | 7 | 10 | 10 | 10 |
| Number of finger calibrations | Minimum 2 or even 4 | Guardian 3: Minimum 2 (every 12 hours) Guardian 4: No finger calibration required | No finger calibration required | No finger calibration required | No finger calibration required |
| Pump insertion replacement time | Every 3 days | Every 3 days | 48–72 hours | 2–3 days [19] | Every 3 days |
| Working principle | 1. SmartGuard™ technology1
2. Manual mode: same as 640G [20]
3. Automatic mode:
• Base: automatically adjusted basal insulin dose every 5 minutes based on real-time CGM values
• Bolus: required to manually administer a bolus by entering carbohydrate information into the insulin pump | 1. SmartGuard™ technology
2. Manual Mode: Same as 640G [20]
3. Automatic Mode:
• Base: precise, automatically selected basal insulin dose (every 5 minutes) based on rtCGM values
• Bolus: automatic bolus correction based on rtCGM value | 1. Using CGM values in conjunction with other variables (“insulin on board”) to predict glucose levels 30 minutes in advance and adjust insulin delivery accordingly
2. If glucose levels fall < 112.5 mg/dl, basal insulin delivery is reduced
3. When glucose is predicted to fall < 70 mg/dl, basal insulin delivery is stopped
4. If glucose values > 160 mg/dl within the next 30 minutes, basal insulin increases
5. If glucose values > 180 mg/dl, Control-IQ technology calculates a correction bolus to a target of 110 mg/dL and delivers 60% of this value as needed, up to once every 1 hour | 1. Model Predictive Control (MPC) – targeted treatment
2. Automatic mode “Off” (open loop) is the mode in which the pump operates according to the programmed basic profile
3. Automatic mode “On” (closed loop) is an operating mode in which insulin delivery is directed by the application, overriding the pre-programmed basal insulin delivery
4. Automatic mode “Attempting” is a mode in which the application attempts to go into Auto mode but some condition prevents it from doing so. In “Attempting” mode, the insulin infusion will revert to the programmed basal infusion after approximately 30 minutes
5. “Boost” mode is a mode that can be used when more insulin is needed. You can set the duration (from 0 to 13 hours) and the start time of Boost mode | 1. MPC algorithm to calculate microboluses of insulin delivered every 5 minutes based on CGM glucose data and predicted glucose values over a 60-minute prediction horizon
2. The user is responsible for delivering bolus doses to meals using bolus settings programmed in PDM
3. HypoProtect function – allows temporary reduction in basal insulin delivery during exercise
4. Manual mode – only then are the basic doses used
5. Automatic mode:
• basal control of the algorithm is based on the patient's total daily insulin dose (TDDI) and does not require user intervention
• TDDI is estimated from the programmed basal doses during system startup
• the system assumes that the user needs 50% TDDI from basal insulin and 50% from bolus
• to estimate TDDI, the system calculates the total daily insulin dose resulting from the programmed basal doses and then doubles this value
• basal doses are modulated every 5 min based on TDDI
• the system tracks and updates the actual TDDI provided to the user. |
| Exercise mode | Appears | Appears | Appears | Appears | Appears (HypoProtect function for use during exercise (provides temporary reduction in basal insulin delivery) |
Options for
glucose targets | 120 mg/dl – default
setting for automatic
mode
150 mg/dl – option for
use during exercise | 100 mg/dl,
110 mg/dl,
120 mg/dl,
150 mg/dl – during
exercise | 112.5–160 mg/dl
112.5-120 mg/dl – during
sleep. 140-160 mg/dl –
during exercise | (105 mg/dl) with an adjustable
range of (80 to 200 mg/dl) | A user-programmable glucose value
between 110–150 mg/dl (110, 120, 130,
140, and 150 mg/dl) |
System
limitations | Dedicated for patients
with T1D ≥ 14 years old
System use ≥ 7 years
of age
TDDI [2] ≥ 8 units per
day. | Patients with T1D aged
7-80 years
TDDI ≥8 units per day | Intended for people aged
≥14 years
T1D in patients ≥6 years
of age.
TDDI ≥10 units of insulin
per day
Body weight ≥55 lbs | Patients with T1D aged 1 year
and over using HCL
Additional age restrictions
may apply depending on the
chosen continuous glucose
monitor and insulin pump | The system has been designed and tested
in patients with T1D aged 2 years and older |
Use by
pregnant
women | Safety has not been
studied in pregnant
women | The safety of using
the MiniMed™ 780G
system in pregnant women has not been
evaluated | Technology is not
indicated for use in
pregnant women | Can be used | Lack of data |
Table II
Advantages and disadvantages of each system
| Advantages | Disadvantages |
|---|
| Medtronic Minimed™ 670G | + An algorithm to provide automatic insulin delivery based on rtCGM data + Provides continuous insight into blood glucose + Improves TIR and reduces risk of hypoglycaemia + Alarms to inform of risks + Waterproofing of the pump for comfortable performance of various activities | – The need for multiple finger pricks – Need for multiple sensor calibrations – Still need to manually insert the post-dose bolus – Non-modifiable blood glucose targets in the Automatic mode limit options – Fewer features available in Auto mode (cannot use differential bolus settings or lower basal rate) – Large number of alarms – No ability to make manual bolus adjustments in automatic mode (introducing “fake carbs” [40]) – Pregnant women cannot operate in auto mode because the factory-set glucose target is too high to meet pregnancy goals [40] |
| Medtronic Minimed™ 780G | + An algorithm to provide automatic insulin delivery based on rtCGM data + Provides continuous insight into blood glucose + Improves TIR and reduces risk of hypoglycaemia + Alarms to inform of risks + Waterproofing of the pump for comfortable performance of various activities + Increased flexibility to set a personal blood glucose goal + System easier to use + MD-Logic [41] artificial pancreas algorithm from DreaMed Diabetes28 responsible for autocorrection boluses + The algorithm "learns the patient" + Application for people surrounding the patient (therapy partners) | – The need for multiple finger pricks – Still needing multiple sensor calibrations (this applies to the 3rdgeneration transmitter - no calibration is needed with the 4thgeneration transmitter) – Large number of alarms – Inconvenience of system upgrade for the user* – The application is only compatible with certain types of phones* – Only one user can be observed at a time from the therapy partner's application* – No ability to see blood glucose, trends on smartwatch* |
| Tandem’s T slim x2 Control IQ | + An algorithm to provide automatic insulin delivery based on rtCGM data + Provides continuous insight into blood glucose + Improves TIR and reduces risk of hypoglycaemia + Alarms to inform of risks + Easy to use + Optional sleep settings + Application for patient + No requirement for finger calibration + The algorithm “learns the patient” | – No ability to return to previous basal mode if closed loop is not adequate – Not compatible with ultrafast-acting insulins – Depends on basal values, carbohydrate ratios, and user adjustments CamAPS [2] – Watertight, not waterproof |
| CamAPS FX | + No finger calibration required + Pregnancy licence + Customizable mobile app + Downloads data from phone for privacy + Fully waterproof pump + Low age restrictions | – Limited clinical experience – App only available for Android – The need to carry a phone and the risk of it being discharged |
| Insulet’s Omnipod5-Automated mode (Hypoprotect™) | + An algorithm to provide automatic insulin delivery based on rtCGM data + Provides continuous insight into blood glucose + Improves TIR and reduces risk of hypoglycaemia + Alarms to inform of risks + Easy to use + Waterproofing of the pump for comfortable performance of various activities + Patient applications (2) + No requirement for finger calibration + The algorithm “learns the patient” + Compatible ultra-rapid-acting insulin | – System in clinical trial phase, few data – Need to control the pump from a phone (frequent need to have a second phone dedicated to the pump due to risk of rapid battery discharge) |
In addition to focusing on glycaemic control, a broader view is very important in diabetes care, in line with recommendations that include healthy lifestyle medicine, and maintenance of normal blood pressure, body weight, and lipid profile [44, 45, 46, 47]. Interesting issues requiring a separate discussion and allowing for a better therapeutic effect in the use of auto-mode are increasing trust and openness to the new technology of patients and members of the diabetic therapeutic team (percentage of sensor use and change of habits) [48].
Considering the whole, it is an undeniable fact that “auto-mode” systems represent a new stage that can be boldly called a revolution in diabetology.
Conclusions
Technology is moving forward, and with it advances in the treatment of diabetes and the prevention of its complications. The commercial HCL systems presented by the authors give patients with diabetes unique and more satisfactory tools for improving glycaemic control in the real word, but still create a challenge for the patient.









